Abstract
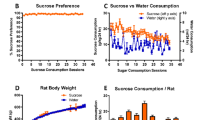
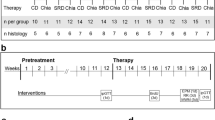
Alzheimer’s disease (AD) is the sixth leading cause of death in the United States where it is estimated that one in three seniors dies with AD or another dementia. Are modern lifestyle habits a contributing factor? Increased carbohydrate (sugar) consumption, stress and disruption of sleep patterns are quickly becoming the norm rather than the exception. Interestingly, seven months on a non-invasive high sucrose diet (20% sucrose in drinking water) has been shown to induce behavioral, metabolic and pathological changes consistent with AD in wild-type mice. As chronic stress and depression are associated with loss of locus coeruleus (LC) noradrenergic neurons and projections (source of anti-inflammatory and trophic factor control), we assessed the ability for a selective LC neurotoxin (DSP4) to accelerate and aggravate a high-sucrose mediated AD-related phenotype in wild-type mice. Male C57/Bl6 mice were divided into four groups: 1) saline injected, 2) DSP4 injected, 3) high sucrose drinking water (20%) or 4) DSP4 injected and high sucrose drinking water. We demonstrate that high sucrose consumption and DSP4 treatment promote an early-stage AD-related phenotype after only 3–4 months, as evidenced by elevated fecal corticosterone, increased despair, spatial memory deficits, increased AChE activity, elevated NO production, decreased pGSK3β and increased pTau. Combined treatment appears to accelerate and aggravate pathological processes consistent with Alzheimer disease and dementia. Developing a simple model in wild-type mice will highlight environmental and lifestyle factors that need to be addressed to slow, prevent or even reverse the rising trend in dementia patient numbers and cost.




Similar content being viewed by others
References
Alzheimer Association (2016) 2016 Alzheimer's disease facts and figures. Alzheimers Dement 12:459–509
Bekar LK, Wei HS, Nedergaard M (2012) The locus coeruleus-norepinephrine network optimizes coupling of cerebral blood volume with oxygen demand. J Cereb Blood Flow Metab 32:2135–2145. https://doi.org/10.1038/jcbfm.2012.115
Beurel E, Grieco SF, Jope RS (2015) Glycogen synthase kinase-3 (GSK3): regulation, actions, and diseases. Pharmacol Ther 148:114–131. https://doi.org/10.1016/j.pharmthera.2014.11.016
Cao D, Lu H, Lewis TL, Li L (2007) Intake of sucrose-sweetened water induces insulin resistance and exacerbates memory deficits and amyloidosis in a transgenic mouse model of Alzheimer disease. J Biol Chem 282:36275–36282. https://doi.org/10.1074/jbc.M703561200
Carvalho C, Cardoso S, Correia SC et al (2012) Metabolic alterations induced by sucrose intake and Alzheimer's disease promote similar brain mitochondrial abnormalities. Diabetes 61:1234–1242. https://doi.org/10.2337/db11-1186
Carvalho C, Machado N, Mota PC et al (2013) Type 2 diabetic and Alzheimer's disease mice present similar behavioral, cognitive, and vascular anomalies. J Alzheimers Dis 35:623–635. https://doi.org/10.3233/JAD-130005
Chen TC, Benjamin DI, Kuo T et al (2017) The glucocorticoid-Angptl4-ceramide axis induces insulin resistance through PP2A and PKCzeta. Sci Signal 10. https://doi.org/10.1126/scisignal.aai7905
Chitturi S, Abeygunasekera S, Farrell GC et al (2002) NASH and insulin resistance: Insulin hypersecretion and specific association with the insulin resistance syndrome. Hepatology 35:373–379. https://doi.org/10.1053/jhep.2002.30692
Counts SE, Mufson EJ (2010) Noradrenaline activation of neurotrophic pathways protects against neuronal amyloid toxicity. J Neurochem 113:649–660. https://doi.org/10.1111/j.1471-4159.2010.06622.x
de la Monte SM (2012) Triangulated mal-signaling in Alzheimer's disease: roles of neurotoxic ceramides, ER stress, and insulin resistance reviewed. J Alzheimers Dis 30(Suppl 2):S231–S249. https://doi.org/10.3233/jad-2012-111727
de la Monte SM, Tong M, Nguyen V, Setshedi M, Longato L, Wands JR (2010) Ceramide-mediated insulin resistance and impairment of cognitive-motor functions. J Alzheimers Dis 21:967–984. https://doi.org/10.3233/jad-2010-091726
de Mello VD, Lankinen M, Schwab U et al (2009) Link between plasma ceramides, inflammation and insulin resistance: association with serum IL-6 concentration in patients with coronary heart disease. Diabetologia 52:2612–2615. https://doi.org/10.1007/s00125-009-1482-9
Debeir T, Marien M, Ferrario J, Rizk P, Prigent A, Colpaert F, Raisman-Vozari R (2004) In vivo upregulation of endogenous NGF in the rat brain by the alpha2-adrenoreceptor antagonist dexefaroxan: potential role in the protection of the basalocortical cholinergic system during neurodegeneration. Exp Neurol 190:384–395
Dudley MW, Howard BD, Cho AK (1990) The interaction of the beta-haloethyl benzylamines, xylamine, and DSP-4 with catecholaminergic neurons. Annu Rev Pharmacol Toxicol 30:387–403
Feinstein DL, Heneka MT, Gavrilyuk V, Dello Russo C, Weinberg G, Galea E (2002) Noradrenergic regulation of inflammatory gene expression in brain. Neurochem Int 41:357–365
Fritschy JM, Grzanna R (1991) Experimentally-induced neuron loss in the locus coeruleus of adult rats. Exp Neurol 111:123–127
Fritschy JM, Geffard M, Grzanna R (1990) The response of noradrenergic axons to systemically administered DSP-4 in the rat: an immunohistochemical study using antibodies to noradrenaline and dopamine-beta-hydroxylase. J Chem Neuroanat 3:309–321
Gavrilyuk V, Dello Russo C, Heneka MT, Pelligrino D, Weinberg G, Feinstein DL (2002) Norepinephrine increases I kappa B alpha expression in astrocytes. J Biol Chem 277:29662–29668
Guix FX, Wahle T, Vennekens K et al (2012) Modification of γ-secretase by nitrosative stress links neuronal ageing to sporadic Alzheimer's disease. EMBO Mol Med 4:660–673. https://doi.org/10.1002/emmm.201200243
Harik SI, LaManna JC, Light AI, Rosenthal M (1979) Cerebral norepinephrine: influence on cortical oxidative metabolism in situ. Science 206:69–71
Harper JM, Austad SN (2000) Fecal glucocorticoids: a noninvasive method of measuring adrenal activity in wild and captive rodents. Physiol Biochem Zool 73:12–22. https://doi.org/10.1086/316721
Hauser J, Sontag TA, Tucha O, Lange KW (2012) The effects of the neurotoxin DSP4 on spatial learning and memory in Wistar rats. Atten Defic Hyperact Disord 4:93–99. https://doi.org/10.1007/s12402-012-0076-4
Heneka MT, Galea E, Gavriluyk V et al (2002) Noradrenergic depletion potentiates beta -amyloid-induced cortical inflammation: implications for Alzheimer's disease. J Neurosci 22:2434–2442
Heneka MT, Ramanathan M, Jacobs AH et al (2006) Locus ceruleus degeneration promotes Alzheimer pathogenesis in amyloid precursor protein 23 transgenic mice. J Neurosci 26:1343–1354
Hernandez F, Lucas JJ, Avila J (2013) GSK3 and tau: two convergence points in Alzheimer's disease. J Alzheimers Dis 33(Suppl 1):S141–S144. https://doi.org/10.3233/JAD-2012-129025
Hooper C, Killick R, Lovestone S (2008) The GSK3 hypothesis of Alzheimer's disease. J Neurochem 104:1433–1439. https://doi.org/10.1111/j.1471-4159.2007.05194.x
Jardanhazi-Kurutz D, Kummer MP, Terwel D, Vogel K, Dyrks T, Thiele A, Heneka MT (2010) Induced LC degeneration in APP/PS1 transgenic mice accelerates early cerebral amyloidosis and cognitive deficits. Neurochem Int 57:375–382. https://doi.org/10.1016/j.neuint.2010.02.001
Kalinin S, Gavrilyuk V, Polak PE, Vasser R, Zhao J, Heneka MT, Feinstein DL (2007) Noradrenaline deficiency in brain increases beta-amyloid plaque burden in an animal model of Alzheimer's disease. Neurobiol Aging 28:1206–1214. https://doi.org/10.1016/j.neurobiolaging.2006.06.003
Krishna M (2013) Role of special stains in diagnostic liver pathology. Clin Liver Dis 2. https://doi.org/10.1002/cld.148
Kwak YD, Wang R, Li JJ, Zhang YW, Xu H, Liao FF (2011) Differential regulation of BACE1 expression by oxidative and nitrosative signals. Mol Neurodegener 6:17. https://doi.org/10.1186/1750-1326-6-17
Lemos C, Rial D, Goncalves FQ et al (2016) High sucrose consumption induces memory impairment in rats associated with electrophysiological modifications but not with metabolic changes in the hippocampus. Neuroscience 315:196–205. https://doi.org/10.1016/j.neuroscience.2015.12.018
Lyn-Cook LE, Lawton M, Tong M et al (2009) Hepatic ceramide may mediate brain insulin resistance and neurodegeneration in type 2 diabetes and non-alcoholic steatohepatitis. J Alzheimers Dis 16:715–729. https://doi.org/10.3233/JAD-2009-0984
Marien MR, Colpaert FC, Rosenquist AC (2004) Noradrenergic mechanisms in neurodegenerative diseases: a theory. Brain Res Brain Res Rev 45:38–78
McIntosh LJ, Hong KE, Sapolsky RM (1998) Glucocorticoids may alter antioxidant enzyme capacity in the brain: baseline studies. Brain Res 791:209–214
Moreira PI (2013) High-sugar diets, type 2 diabetes and Alzheimer's disease. Curr Opin Clin Nutr Metab Care 16:440–445. https://doi.org/10.1097/MCO.0b013e328361c7d1
Osmanovic J, Plaschke K, Salkovic-Petrisic M, Grünblatt E, Riederer P, Hoyer S (2010) Chronic exogenous corticosterone administration generates an insulin-resistant brain state in rats. Stress 13:123–131. https://doi.org/10.3109/10253890903080379
Parr C, Mirzaei N, Christian M, Sastre M (2015) Activation of the Wnt/β-catenin pathway represses the transcription of the β-amyloid precursor protein cleaving enzyme (BACE1) via binding of T-cell factor-4 to BACE1 promoter. FASEB J 29:623–635. https://doi.org/10.1096/fj.14-253211
Peppiatt CM, Howarth C, Mobbs P, Attwell D (2006) Bidirectional control of CNS capillary diameter by pericytes. Nature 443:700–704
Raichle ME, Hartman BK, Eichling JO, Sharpe LG (1975) Central noradrenergic regulation of cerebral blood flow and vascular permeability. Proc Natl Acad Sci U S A 72:3726–3730
Salkovic-Petrisic M, Hoyer S (2007) Central insulin resistance as a trigger for sporadic Alzheimer-like pathology: an experimental approach. J Neural Transm Suppl:217–233
Salkovic-Petrisic M, Osmanovic J, Grünblatt E, Riederer P, Hoyer S (2009) Modeling sporadic Alzheimer's disease: the insulin resistant brain state generates multiple long-term morphobiological abnormalities including hyperphosphorylated tau protein and amyloid-beta. J Alzheimers Dis 18:729–750. https://doi.org/10.3233/JAD-2009-1184
Scheiblich H, Schlutter A, Golenbock DT, Latz E, Martinez-Martinez P, Heneka MT (2017) Activation of the NLRP3 inflammasome in microglia: The role of ceramide. J Neurochem. https://doi.org/10.1111/jnc.14225
Seol GH, Ziburkus J, Huang S et al (2007) Neuromodulators control the polarity of spike-timing-dependent synaptic plasticity. Neuron 55:919–929
Stranahan AM, Arumugam TV, Cutler RG, Lee K, Egan JM, Mattson MP (2008a) Diabetes impairs hippocampal function through glucocorticoid-mediated effects on new and mature neurons. Nat Neurosci 11:309–317. https://doi.org/10.1038/nn2055
Stranahan AM, Lee K, Pistell PJ et al (2008b) Accelerated cognitive aging in diabetic rats is prevented by lowering corticosterone levels. Neurobiol Learn Mem 90:479–483. https://doi.org/10.1016/j.nlm.2008.05.005
Szot P, Franklin A, Miguelez C et al (2016) Depressive-like behavior observed with a minimal loss of locus coeruleus (LC) neurons following administration of 6-hydroxydopamine is associated with electrophysiological changes and reversed with precursors of norepinephrine. Neuropharmacology 101:76–86. https://doi.org/10.1016/j.neuropharm.2015.09.003
Tong M, de la Monte SM (2009) Mechanisms of ceramide-mediated neurodegeneration. J Alzheimers Dis 16:705–714. https://doi.org/10.3233/JAD-2009-0983
Wang ZJ, Zhang XQ, Cui XY et al (2015) Glucocorticoid receptors in the locus coeruleus mediate sleep disorders caused by repeated corticosterone treatment. Sci Rep 5:9442. https://doi.org/10.1038/srep09442
Zarow C, Lyness SA, Mortimer JA, Chui HC (2003) Neuronal loss is greater in the locus coeruleus than nucleus basalis and substantia nigra in Alzheimer and Parkinson diseases. Arch Neurol 60:337–341
Zhang J, Zhu Y, Zhan G et al (2014) Extended wakefulness: compromised metabolics in and degeneration of locus ceruleus neurons. J Neurosci 34:4418–4431. https://doi.org/10.1523/JNEUROSCI.5025-12.2014
Acknowledgements
This work was supported by the Saskatchewan Health Research Foundation (SHRF grant number 3075.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None of the authors have any conflicts, financial or otherwise, to disclose. All authors approved the final manuscript.
Rights and permissions
About this article
Cite this article
Choudhary, P., Pacholko, A.G., Palaschuk, J. et al. The locus coeruleus neurotoxin, DSP4, and/or a high sugar diet induce behavioral and biochemical alterations in wild-type mice consistent with Alzheimers related pathology. Metab Brain Dis 33, 1563–1571 (2018). https://doi.org/10.1007/s11011-018-0263-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-018-0263-x