Abstract
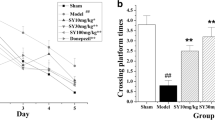
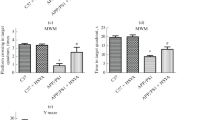
Alzheimer’s disease (AD), the most common cause of dementia worldwide, is mainly characterized by the aggregated β-amyloid (Aβ) and hyperphosphorylated tau. Safflower yellow (SY) is a novel water extract of natural safflower and has been suggested to ameliorate memory deficits in several animal models of dementia. In this study, we aimed to investigate the effect and mechanism of SY on deficits of learning and memory and hyperphosphorylation of tau in APP/PS1 double transgenic mice. APP/PS1 mice were administered with SY (10, 30, 100 mg/kg) by oral gavage for three months at the age of six months. The ability of learning and memory was investigated using the step-down test and Morris water maze test, and protein level in the brain was evaluated using western blot. Here, we found that SY treatment can improve spatial learning and memory ability, and reduce tau hyperphosphorylation at Ser199, Thr205, Ser396, Ser404 sites in APP/PS1 mice. In addition, the activity the of cyclin-dependent kinase 5 (CDK-5) and glycogen synthase kinase 3β (GSK-3β), major kinases involved in tau phosphorylation, was siginificantly decreased in APP/PS1 mice by SY treatment. These results support SY can serve as a promising multitarget neuronal therapeutic agent for the treatment of AD.




Similar content being viewed by others
References
Ballatore C, Lee VM, Trojanowski JQ (2007) Tau-mediated neurodegeneration in Alzheimer’s disease and related disorders. Nat Rev Neurosci 8:663–672
Chen L, Ding L, Zhang H, Li J, Wang Y, Wang X, Qu C, Zhang H (2006) Dynamic microwave-assisted extraction coupled with on-line spectrophotometric determination of safflower yellow in Flos Carthami. Anal Chim Acta 580:75–82
Chin PC, Majdzadeh N, D’Mello SR (2005) Inhibition of GSK3beta is a common event in neuroprotection by different survival factors. Brain Res Mol Brain Res 137:193–201
Cohen P, Frame S (2001) The renaissance of GSK3. Nat Rev Mol Cell Biol 2:769–776
Dhariwala FA, Rajadhyaksha MS (2008) An unusual member of the Cdk family: Cdk5. Cell Mol Neurobiol 28:351–369
Dolan PJ, Johnson GV (2010) The role of tau kinases in Alzheimer’s disease. Curr Opin Drug Discov Devel 13:595–603
Gong CX, Lidsky T, Wegiel J, Zuck L, Grundke-Iqbal I, Iqbal K (2000) Phosphorylation of microtubule-associated protein tau is regulated by protein phosphatase 2 A in mammalian brain. Implications for neurofibrillary degeneration in Alzheimer’s disease. J Biol Chem 275:5535–5544
Hanger DP, Byers HL, Wray S, Leung KY, Saxton MJ, Seereeram A, Reynolds CH, Ward MA, Anderton BH (2007) Novel phosphorylation sites in tau from Alzheimer brain support a role for casein kinase 1 in disease pathogenesis. J Biol Chem 282:23645–23654
Hanger DP, Anderton BH, Noble W (2009) Tau phosphorylation: the therapeutic challenge for neurodegenerative disease. Trends Mol Med 15:112–119
Herreman A, Serneels L, Annaert W, Collen D, Schoonjans L, De Strooper B (2000) Total inactivation of gamma-secretase activity in presenilin-deficient embryonic stem cells. Nat Cell Biol 2:461–462
Hu YL, Wang PL, Zhang Y, Liu M (2012) Effects of safflower yellow on learning and memory impairments on scopolamine-induced dementia in mice. Chin J Gerontol 19:4197–4198
Huang HC, Jiang ZF (2009) Accumulated amyloid-beta peptide and hyperphosphorylated tau protein: relationship and links in Alzheimer’s disease. J Alzheimers Dis 16:15–27
Iqbal K, Alonso AC, Chen S, Chohan MO, El-Akkad E, Gong CX, Khatoon S, Li B, Liu F, Rahman A, Tanimukai H, Grundke-Iqbal I (2005) Tau pathology in Alzheimer disease and other tauopathies. Biochim Biophys Acta 1739:198–210
Iqbal K, Liu F, Gong CX, Grundke-Iqbal I (2010) Tau in Alzheimer disease and related tauopathies. Curr Alzheimer Res 7:656–664
Jayapalan S, Natarajan J (2013) The role of CDK5 and GSK3B kinases in hyperphosphorylation of microtubule associated protein tau (MAPT) in Alzheimer’s disease. Bioinformation 9:1023–1030
Jessberger S, Aigner S, Clemenson GJ, Toni N, Lie DC, Karalay O, Overall R, Kempermann G, Gage FH (2008) Cdk5 regulates accurate maturation of newborn granule cells in the adult hippocampus. PLoS Biol 6:e272
Kurt MA, Davies DC, Kidd M, Duff K, Howlett DR (2003) Hyperphosphorylated tau and paired helical filament-like structures in the brains of mice carrying mutant amyloid precursor protein and mutant presenilin-1 transgenes. Neurobiol Dis 14:89–97
Li HX, Han SY, Wang XW, Ma X, Zhang K, Wang L, Ma ZZ, Tu PF (2009) Effect of the carthamins yellow from Carthamus tinctorius L. On hemorheological disorders of blood stasis in rats. Food Chem Toxicol 47(8):1797–1802
Li CC, Yang CZ, Li XM, Zhao XM, Zou Y, Fan L, Zhou L, Liu JC, Niu YC (2012) Hydroxysafflor yellow a induces apoptosis in activated hepaticstellate cells through ERK1/2 pathway in vitro. Eur J Pharm Sci 46(5):397–404
Liu F, Grundke-Iqbal I, Iqbal K, Gong CX (2005) Contributions of protein phosphatases PP1, PP2A, PP2B and PP5 to the regulation of tau phosphorylation. Eur J Neurosci 22:1942–1950
Liu Y, Lian Z, Zhu H, Wang Y, Yu S, Chen T, Qu J, Li J, Ma S, Chen X (2013) A systematic, integrated study on the neuroprotective effects of hydroxysafflor yellow a revealed by (1)H NMR-based Metabonomics and the NF-kappaB pathway. Evid Based Complement Alternat Med 2013:147362
Lucas JJ, Hernandez F, Gomez-Ramos P, Moran MA, Hen R, Avila J (2001) Decreased nuclear beta-catenin, tau hyperphosphorylation and neurodegeneration in GSK-3beta conditional transgenic mice. EMBO J 20:27–39
Ma Q, Ruan YY, Xu H, Shi XM, Wang ZX, Hu YL (2015) Safflower yellow reduces lipid peroxidation, neuropathology, tau phosphorylation and ameliorates amyloid β-induced impairment of learning and memory in rats. Biomed Pharmacother 76:153–164
Maas T, Eidenmuller J, Brandt R (2000) Interaction of tau with the neural membrane cortex is regulated by phosphorylation at sites that are modified in paired helical filaments. J Biol Chem 275:15733–15740
Meselhy MR, Kadota S, Momose Y, Hatakeyama N, Kusai A, Hattori M, et al. (1993) Two new quinochalcone yellow pigments from Carthamus tinctorius and Ca2 þantagonistic activity of tinctormine. Chem Pharm Bull 41(10):1796–1802
Rodriguez-Martin T, Cuchillo-Ibanez I, Noble W, Nyenya F, Anderton BH, Hanger DP (2013) Tau phosphorylation affects its axonal transport and degradation. Neurobiol Aging 34:2146–2157
Schindowski K, Bretteville A, Leroy K, Begard S, Brion JP, Hamdane M, Buee L (2006) Alzheimer’s disease-like tau neuropathology leads to memory deficits and loss of functional synapses in a novel mutated tau transgenic mouse without any motor deficits. Am J Pathol 169:599–616
Selkoe DJ (2001) Alzheimer’s disease results from the cerebral accumulation and cytotoxicity of amyloid beta-protein. J Alzheimers Dis 3:75–80
Sengupta A, Novak M, Grundke-Iqbal I, Iqbal K (2006) Regulation of phosphorylation of tau by cyclin-dependent kinase 5 and glycogen synthase kinase-3 at substrate level. FEBS Lett 580:5925–5933
Shankar GM, Li S, Mehta TH, Garcia-Munoz A, Shepardson NE, Smith I, Brett FM, Farrell MA, Rowan MJ, Lemere CA, Regan CM, Walsh DM, Sabatini BL, Selkoe DJ (2008) Amyloid-beta protein dimers isolated directly from Alzheimer’s brains impair synaptic plasticity and memory. Nat Med 14:837–842
Shelton SB, Johnson GV (2004) Cyclin-dependent kinase-5 in neurodegeneration. J Neurochem 88:1313–1326
Sontag E, Luangpirom A, Hladik C, Mudrak I, Ogris E, Speciale S, White CR (2004) Altered expression levels of the protein phosphatase 2 A ABalphaC enzyme are associated with Alzheimer disease pathology. J Neuropathol Exp Neurol 63:287–301
Takahashi Y, Miyasaka N, Tasaka S, Miura I, Urano S, Ikura M, et al. (1982) Constitution of two coloring matters in the flower petals of Carthamus tinctorius L. Tetrahedron Lett 23:51–63
Wang JZ, Wu Q, Smith A, Grundke-Iqbal I, Iqbal K (1998) Tau is phosphorylated by GSK-3 at several sites found in Alzheimer disease and its biological activity markedly inhibited only after it is prephosphorylated by A-kinase. FEBS Lett 436:28–34
Wang CC, Choy CS, Liu YH, Cheah KP, Li JS, Wang JT, Yu WY, Lin CW, Cheng HW, Hu CM (2011) Protective effect of dried safflower petal aqueous extract and its main constituent, carthamus yellow, againstlipopolysaccharide-induced inflammation in RAW264.7 macrophages. J Sci Food Agric 91(2):218–225
Wen Y, Planel E, Herman M, Figueroa HY, Wang L, Liu L, Lau LF, Yu WH, Duff KE (2008) Interplay between cyclin-dependent kinase 5 and glycogen synthase kinase 3 beta mediated by neuregulin signaling leads to differential effects on tau phosphorylation and amyloid precursor protein processing. J Neurosci 28:2624–2632
Xu H, Ma Q, Wang ZX, Hu YL (2013) Effects of safflower yellow on D-galactose/sodium nitrite induced learning and memory disorders in mice. Chin J Gerontol 29:59–61
Yang Z, Yang J, Jia Y, Tian Y, Wen A (2009) Pharmacokinetic properties of hydroxysafflor yellow a in healthy Chinese female volunteers. J Ethnopharmacol 124:635–638
Zhang Z, Nadeau P, Song W, Donoviel D, Yuan M, Bernstein A, Yankner BA (2000) Presenilins are required for gamma-secretase cleavage of beta-APP and transmembrane cleavage of notch-1. Nat Cell Biol 2:463–465
Acknowledgments
This research was supported by Natural Science Foundation of China (No.81360495).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Ruan, Yy., Zhai, W., Shi, Xm. et al. Safflower yellow ameliorates cognition deficits and reduces tau phosphorylation in APP/PS1 transgenic mice. Metab Brain Dis 31, 1133–1142 (2016). https://doi.org/10.1007/s11011-016-9857-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-016-9857-3