Abstract
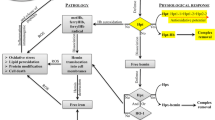
Basic science investigations and clinical observations in recent years indicate that hemoglobins (Hbs) may have important roles in the pathogenesis of multiple sclerosis (MS). These findings can be summarized as follows: 1- Erythrocyte fragility is higher in MS patients, the released free Hb damages blood-brain barrier, myelin basic protein and also triggers iron overload and inflammation. 2- Free Hb may further activate the inflammatory responses through Toll-like receptor 4 (TLR4), present on microglia and other innate immunocytes. 3- Hbs are expressed in neural cells including dopaminergic neurons. Also, several studies have demonstrated that Hbs are expressed in astrocytes and oligodendroglia. 4- Hb overexpression in neural cells upregulate mitochondrial complex I–V subunits. The comparison of the mitochondrial proteome between healthy and patients with MS revealed only four differentially expressed proteins including Hb β-chain. 5- Microarray analysis of 8300 genes in monocytes of twins with and without MS showed a difference in 25 genes that include genes encoding α- and β-globins as well. 6- β- and α-globin gene clusters reside at chromosomal regions 11p15.5 and 16p13.3, respectively. Whole genome screen (WGS) in Sardinian MS families using 327 markers revealed linkage in 3 regions including 11p15.5 loci. Further, 11p15.5 and 16p13.3 were part of the 17 regions identified in the WGS study of 136 sibling-pairs in Nordic countries analyzing 399 microsatellite markers. In the light of these findings, we propose that free Hb released from dying erythrocytes is detrimental. On the contrary, intracellular Hbs in neural cells are protective in MS. The genomic linkage findings can be explained by common haematologically-silent Hb variants that may lower the protective function of intracellular Hbs, and therefore, enhance the risk for MS. In the absence of such variants, aberrations in the translational and post-translational mechanisms controlling synthesis of neural Hbs may also enhance the vulnerability to MS. Alternatively, such genetic variants may perturb the metabolism of anti-inflammatory hemorphins produced via cleavage of Hbs.
Similar content being viewed by others
References
Aboul-Enein F, Lassmann H (2005) Mitochondrial damage and histotoxic hypoxia: a pathway of tissue injury in inflammatory brain disease? Acta Neuropathol 109:49–55
Akesson E, Oturai A, Berg J, Fredrikson S, Andersen O, Harbo HF, Laaksonen M, Myhr KM, Nyland HI, Ryder LP, Sandberg-Wollheim M, Sorensen PS, Spurkland A, Svejgaard A, Holmans P, Compston A, Hillert J, Sawcer S (2002) A genome-wide screen for linkage in Nordic sib-pairs with multiple sclerosis. Genes Immun 3:279–285
Alanoglu G, Kilbas S, Arslan C, Senol A, Kutluhan S (2007) Autoimmune hemolytic anemia during interferon-beta-I b treatment for multiple sclerosis. Mult Scler 13(5):683–685
Alperin JB, Dow PA, Petteway MB (1977) Hemoglobin A2 levels in health and various hematologic disorders. Am J Clin Pathol 67:219–226
Bambo MP, Garcia-Martin E, Perez-Olivan S, Sigut J, Fumero F, Fuentes JL, Ara JR, Martin J, Larrosa JM, Gonzalez-De la Rosa M (2013) Diagnostic ability of a new method for measuring haemoglobin levels in the optic nerve head in multiple sclerosis patients. Br J Ophthalmol 97:1543–1548
Bamm VV, Harauz G (2014) Hemoglobin as a source of iron overload in multiple sclerosis: does multiple sclerosis share risk factors with vascular disorders? Cell Mol Life Sci 71:1789–1798
Bamm VV, Lanthier DK, Stephenson EL, Smith GS, Harauz G (2015) In vitro study of the direct effect of extracellular hemoglobin on myelin components. Biochim Biophys Acta 1852:92–103
Baum H (1995) Mitochondrial antigens, molecular mimicry and autoimmune disease. Biochim Biophys Acta 1271:111–121
Biagioli M, Pinto M, Cesselli D, Zaninello M, Lazarevic D, Roncaglia P, Simone R, Vlachouli C, Plessy C, Bertin N, Beltrami A, Kobayashi K, Gallo V, Santoro C, Ferrer I, Rivella S, Beltrami CA, Carninci P, Raviola E, Gustincich S (2009) Unexpected expression of alpha- and beta-globin in mesencephalic dopaminergic neurons and glial cells. Proc Natl Acad Sci U S A 106:15454–15459
Breymann C, Fibach E, Visca E, Huettner C, Huch A, Huch R (1999) Induction of fetal hemoglobin synthesis with recombinant human erythropoietin in anemic patients with heterozygous beta-thalassemia during pregnancy. J Matern Fetal Med 8(1):1–7
Brines M, Cerami A (2012) The receptor that tames the innate immune response. Mol Med 18:486–496
Broadwater L, Pandit A, Clements R, Azzam S, Vadnal J, Sulak M, Yong VW, Freeman EJ, Gregory RB, McDonough J (2011) Analysis of the mitochondrial proteome in multiple sclerosis cortex. Biochim Biophys Acta 1812:630–641
Brown N (2014) Localization of hemoglobin in MS cortex and its relevance to MS neuropathology. Thesis Submitted to the Kent State University Honors College Advisor: Jennifer McDonough. Available at: https://etd.ohiolink.edu/pg_10?0::NO:10:P10_ACCESSION_NUM:ksuhonors1399400834. Accessed May 24, 2016
Brown N, Alkhayer K, Clements R, Singhal N, Gregory R, Azzam S, Li S, Freeman E, McDonough J (2016) Neuronal hemoglobin expression and its relevance to multiple sclerosis neuropathology. J Mol Neurosci [Epub ahead of print]
Butt OI, Buehler PW, D'Agnillo F (2011) Blood-brain barrier disruption and oxidative stress in Guinea pig after systemic exposure to modified cell-free hemoglobin. Am J Pathol 178:1316–1328
Caspary EA, Sewell F, Field EJ (1967) Red blood cell fragility in multiple sclerosis. Br Med J 2:610–611
Cervellini I, Annenkov A, Brenton T, Chernajovsky Y, Ghezzi P, Mengozzi M (2013a) Erythropoietin (EPO) increases myelin gene expression in CG4 oligodendrocyte cells through the classical EPO receptor. Mol Med 19:223–229
Cervellini I, Ghezzi P, Mengozzi M (2013b) Therapeutic efficacy of erythropoietin in experimental autoimmune encephalomyelitis in mice, a model of multiple sclerosis. Methods Mol Biol 982:163–173
Chmielewski H, Kedziora J, Zagórski T, Dudek I, Berkan L, Mazurek M (1987) Effect of the treatment with adrenocorticotropic hormone on the 2,3-diphosphoglycerate level in the erythrocytes of patients with multiple sclerosis. Neurol Neurochir Pol 21:309–314
Coraddu F, Sawcer S, D'Alfonso S, Lai M, Hensiek A, Solla E, Broadley S, Mancosu C, Pugliatti M, Marrosu MG, Compston A (2001) A genome screen for multiple sclerosis in Sardinian multiplex families. Eur J Hum Genet 9:621–626
Cousens NE, Gaff CL, Metcalfe SA, Delatycki MB (2010) Carrier screening for beta-thalassaemia: a review of international practice. Eur J Hum Genet 18:1077–1083
De Keyser J, Steen C, Mostert JP, Koch MW (2008) Hypoperfusion of the cerebral white matter in multiple sclerosis: possible mechanisms and pathophysiological significance. J Cereb Blood Flow Metab 28:1645–1651
Dehghan S, Hesaraki M, Soleimani M, Mirnajafi-Zadeh J, Fathollahi Y, Javan M (2016) Oct4 transcription factor in conjunction with valproic acid accelerates myelin repair in demyelinated optic chiasm in mice. Neuroscience 318:178–189
Dyall SD, Brown MT, Johnson PJ (2004) Ancient invasions: from endosymbionts to organelles. Science 304(5668):253–257
Farber SD, Farber MO, Brewer G, Magnes CJ, Peterson RG (1991) Oxygen affinity of hemoglobin and peripheral nerve degeneration in experimental diabetes. J Neurol Sci 101:204–207
Giardine B, Borg J, Viennas E, et al. (2014) Updates of the HbVar database of human hemoglobin variants and thalassemia mutations. Nucleic Acids Res 42(Database issue):D1063–D1069
Giunti D, Parodi B, Cordano C, Uccelli A, Kerlero de Rosbo N (2014) Can we switch microglia's phenotype to foster neuroprotection? Focus on multiple sclerosis. Immunology 141:328–339
Graham J, Hobson D, Ponnampalam A (2014) High affinity hemoglobin and Parkinson's disease. Med Hypotheses 83:819–821
Hon GM, Hassan MS, van Rensburg SJ, Abel S, Van Jaarsveld P, Erasmus RT, Matsha T (2009) Red blood cell membrane fluidity in the etiology of multiple sclerosis. J Membr Biol 232:25–34
Hon GM, Hassan MS, van Rensburg SJ, Erasmus RT, Matsha T (2012) The haematological profile of patients with multiple sclerosis. Open J Mod Neurosurg 2:36–44
Horng LY, Hsu PL, Chen LW, Tseng WZ, Hsu KT, Wu CL, Wu RT (2015) Activating mitochondrial function and haemoglobin expression with EH-201, an inducer of erythropoietin in neuronal cells, reverses memory impairment. Br J Pharmacol 172(19):4741–4756
Jilani T, Iqbal MP (2011) Does vitamin E have a role in treatment and prevention of anemia? Pak J Pharm Sci 24(2):237–242
Kang JH, Chen YH, Lin, HC (2010) Comorbidities amongst patients with multiple sclerosis: a population-based controlled study. Eur J Neuroln.d. 17: 1215–1219
Khatri I, Alexander C, Brandenburg K, Fournier K, Mach JP, Rietschel ET, Ulmer AJ, Terzioglu E, Waelli T, Gorczynski RM (2009) Induction of tolerogenic vs immunogenic dendritic cells (DCs) in the presence of GM-CSF is regulated by the strength of signaling from monophosphoryl lipid a (MPLA) in association with glutathione and fetal hemoglobin gamma-chain. Immunol Lett 124:44–49
Kocer B, Engur S, Ak F, Yilmaz M (2009) Serum vitamin B12, folate, and homocysteine levels and their association with clinical and electrophysiological parameters in multiple sclerosis. J Clin Neurosci 16:399–403
Koudriavtseva T, Renna R, Plantone D, Mandoj C, Piattella MC, Giannarelli D (2015) Association between anemia and multiple sclerosis. Eur Neurol 73:233–237
Lagace DC, Eisch AJ (2005) Mood-stabilizing drugs: are their neuroprotective aspects clinically relevant? Psychiatr Clin North Am 28(2):399–414
Lang E, Lang F (2015) Triggers, inhibitors, mechanisms, and significance of eryptosis: the suicidal erythrocyte death. Biomed Res Int 2015:513518. doi:10.1155/2015/513518
Lassmann H (2003) Hypoxia-like tissue injury as a component of multiple sclerosis lesions. J Neurol Sci 206:187–191
Lee SK, Ding JL (2013) A perspective on the role of extracellular hemoglobin on the innate immune system. DNA Cell Biol 32:36–40
Liepke C, Baxmann S, Heine C, Breithaupt N, Ständker L, Forssmann WG (2003) Human hemoglobin-derived peptides exhibit antimicrobial activity: a class of host defense peptides. J Chromatogr B Anal Technol Biomed Life Sci 791:345–356
Lysandropoulos AP, Benghiat F (2013) Severe auto-immune hemolytic anemia in a fingolimod-treated multiple sclerosis patient. Mult Scler 19(11):1551–1552
Marcel GA, George C (1983) Pentoxifylline and cerebrovascular diseases. Eur Neurol 22(Suppl 1):89–97
Marrie RA, Reider N, Cohen J, Stuve O, Sorensen PS, Cutter G, Reingold SC, Trojano M (2015) A systematic review of the incidence and prevalence of autoimmune disease in multiple sclerosis. Mult Scler 21:282–289
Modell B, Darlison M. (2015)Global epidemiology of haemoglobin disorders and derived service indicators. Available at: http://www.who.int/bulletin/volumes/86/6/06-036673/en/. Accessed April 23, 2015
Monti B, Polazzi E, Contestabile A (2009) Biochemical, molecular and epigenetic mechanisms of valproic acid neuroprotection. Curr Mol Pharmacol 2(1):95–109
Najmi Varzaneh F, Najmi Varzaneh F, Azimi AR, Rezaei N, Sahraian MA (2014) Efficacy of combination therapy with erythropoietin and methylprednisolone in clinical recovery of severe relapse in multiple sclerosis. Acta Neurol Belg 114(4):273–278
Ntaios G, Chatzinikolaou A, Saouli Z, et al. (2007) Discrimination indices as screening tests for beta-thalassemic trait. Ann Hematol 86:487–491
O'Brien K, Fitzgerald DC, Naiken K, Alugupalli KR, Rostami AM, Gran B (2008) Role of the innate immune system in autoimmune inflammatory demyelination. Curr Med Chem 15:1105–1115
Papayannopoulou T, Finch CA (1972) On the in vivo action of erythropoietin: a quantitative analysis. J Clin Invest 51(5):1179–1185
Pazhoohan S, Satarian L, Asghari AA, Salimi M, Kiani S, Mani AR, Javan M (2014) Valproic acid attenuates disease symptoms and increases endogenous myelin repair by recruiting neural stem cells and oligodendrocyte progenitors in experimental autoimmune encephalomyelitis. Neurodegener Dis 13(1):45–52
Poljakovic Z, Zurak N, Brinar V, Korsic M, Basic S, Hajnsek S (2006) Growth hormone and insulin growth factor-I levels in plasma and cerebrospinal fluid of patients with multiple sclerosis. Clin Neurol Neurosurg 108:255–258
Prineas J (1968) Red blood cell size in multiple sclerosis. Acta Neurol Scand 44:81–90
Reddi YR, Sudhakar Rao V, Reddi GD, Niranjan Rao P, Nair R, Ahuja YR (1976) Elevated hemoglobin-A2 levels in rheumatic fever and rheumatic heart disease: a new finding. Indian Pediatr 13:153–154
Reynolds EH, Linnell JC, Faludy JE (1991) Multiple sclerosis associated with vitamin B12 deficiency. Arch Neurol 48:808–811
Reynolds EH, Bottiglieri T, Laundy M, Crellin RF, Kirker SG (1992) Vitamin B12 metabolism in multiple sclerosis. Arch Neurol 49(6):649–645
Richter F, Meurers BH, Zhu C, Medvedeva VP, Chesselet MF (2009) Neurons express hemoglobin alpha- and beta-chains in rat and human brains. J Comp Neurol 515:538–547
Rieckmann P, O'Connor P, Francis GS, Wetherill G, Alteri E (2004) Haematological effects of interferon-beta-1a (Rebif) therapy in multiple sclerosis. Drug Saf 27:745–756
Saeedi M, Forughipour M, Sasannezhad P, Shoeibi A (2011) Interferon-Beta-1b induced autoimmune hemolytic anemia in a patient with MS: a case report. Iran Red Crescent Med J 13(3):210–212
Sanderson K, Nyberg F, Khalil Z (1998) Modulation of peripheral inflammation by locally administered hemorphin-7. Inflamm Res 47:49–55
Sandyk R, Awerbuch GI (1993) Vitamin B12 and its relationship to age of onset of multiple sclerosis. Int J Neurosci 71(1–4):93–99
Särkijärvi S, Kuusisto H, Paalavuo R, Levula M, Airla N, Lehtimäki T, Kaprio J, Koskenvuo M, Elovaara I (2006) Gene expression profiles in Finnish twins with multiple sclerosis. BMC Med Genet 7:11
Sheshadri P, Abraham J (2012) Antimicrobial properties of hemoglobin. Immunopharmacol Immunotoxicol 34:896–900
Song CZ, Wang QW, Song CC (2012) Diminution of hemoglobin-derived hemorphin: an underlying risk factor for cognitive deficit in diabetes. J Neurol Sci 317:157–158
Terova G, Cattaneo AG, Preziosa E, Bernardini G, Saroglia M (2011) Impact of acute stress on antimicrobial polypeptides mRNA copy number in several tissues of marine sea bass (Dicentrarchus labrax). BMC Immunol 12:69. doi:10.1186/1471-2172-12-69
Trapp BD, Stys PK (2009) Virtual hypoxia and chronic necrosis of demyelinated axons in multiple sclerosis. Lancet Neurol 8:280–291
Ullal AJ, Corrales J, Fernandes JM (2011) Application of antimicrobial polypeptide host defenses to aquaculture: exploitation of downregulation and upregulation responses. Comp Biochem Physiol Part D Genomics Proteomics 6:44–54
Usman M, Moinuddin M, Ahmed SA (2011) Role of iron deficiency anemia in the propagation of beta thalssemia gene. Kor J Hepatol 46:41–44
Ventura P, Panini R, Tremosini S, Salvioli G (2004) A role for homocysteine increase in haemolysis of megaloblastic anaemias due to vitamin B(12) and folate deficiency: results from an in vitro experience. Biochim Biophys Acta 1739(3):3–42
Walser M, Schiöler L, Oscarsson J, Aberg MA, Svensson J, Aberg ND, Isgaard J (2014) Different modes of GH administration influence gene expression in the male rat brain. J Endocrinol 222:181–190
Witte ME, Mahad DJ, Lassmann H, van Horssen J (2014) Mitochondrial dysfunction contributes to neurodegeneration in multiple sclerosis. Trends Mol Med 20:179–187
Yamamoto Y, Ono H, Ueda A, Shimamura M, Nishimura K, Hazato T (2002) Spinorphin as an endogenous inhibitor of enkephalin-degrading enzymes: roles in pain and inflammation. Curr Protein Pept Sci 3:587–599
Zlatkov N, Damianova L, Prodanov K, Kiriakova N (1990) Hemoglobin A2 and F in patients with psoriasis. Dermatol Monatsschr 176:403–406
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors report no conflict of interest.
Rights and permissions
About this article
Cite this article
Altinoz, M.A., Ozcan, E.M., Ince, B. et al. Hemoglobins as new players in multiple sclerosis: metabolic and immune aspects. Metab Brain Dis 31, 983–992 (2016). https://doi.org/10.1007/s11011-016-9845-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-016-9845-7