Abstract
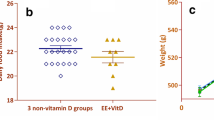
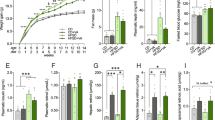
Type 1 diabetes mellitus (T1DM) has been associated with long-term complications in the central nervous system, causing brain cellular dysfunctions and cognitive deficits. On the other hand, enriched environment (EE) induces experience-dependent plasticity, especially in the hippocampus, improving the performance of animals in learning and memory tasks. Thus, our objective was to investigate the influence of the EE on memory deficits, locomotion, corticosterone levels, synaptophysin (SYP) protein immunoreactivity, cell survival and microglial activation in the dentate gyrus (DG) of T1DM rat hippocampus. Male Wistar rats (21-day-old) were exposed to EE or maintained in standard housing (controls, C) for 3 months. At adulthood, the C and EE animals were randomly divided and diabetes was induced in half of them. All the animals received 4 doses of BrdU, 24 h apart. Hippocampus-dependent spatial memory, general locomotion and serum corticosterone levels were evaluated at the end of the experiment. The animals were transcardially perfused 30 days post-BrdU administration. Our results showed that EE was able to prevent/delay the development of memory deficits caused by diabetes in rats, however it did not revert the motor impairment observed in the diabetic group. SYP immunoreactivity was increased in the enriched healthy group. The EE decreased the serum corticosterone levels in diabetic adult rats and attenuated the injurious microglial activation, though without altering the decrease of the survival cell. Thus, EE was shown to help to ameliorate cognitive comorbidities associated with T1DM, possibly by reducing hyperactivity in the hypothalamic–pituitary–adrenal axis and microglial activation in diabetic animals.






Similar content being viewed by others
References
Alvarez EO, Beauquis J, Revsin Y, Banzan AM, Roig P, Nicola AF, Saravia F (2009) Cognitive dysfunction and hippocampal changes in experimental type 1 diabetes. Behav Brain Res 198:224–230
Ayoub AE, Salm AK (2003) Increased morphological diversity of microglia in the activated hypothalamic supraoptic nucleus. J Neurosci 23:7759–7766
Beauquis J, Saravia F, Coulaud J, Roig P, Dardenne M, Homo-Delarche F, De Nicola A (2008) Prominently decreased hippocampal neurogenesis in a spontaneous model of type 1 diabetes, the nonobese diabetic mouse. Exp Neurol 210:359–367
Beauquis J, Roig P, Nicola AF, Saravia F (2010) Short-term environmental enrichment enhances adult neurogenesis, vascular network and dendritic complexity in the hippocampus of type 1 diabetic mice. PLoS One 5:1–12
Benaroya-Milshtein N, Hollander N, Apter A, Kukulansky T, Raz N, Wilf A, Yaniv I, Pick CG (2004) Environmental enrichment in mice decreases anxiety, attenuates stress responses and enhances natural killer cell activity. Eur J Neurosci 20:1341–1347
Biessels GJ, Kamal A, Ramakers GM, Urban IJ, Spruijt BM, Erkelens DW, Gispen WH (1996) Place learning and Hippocampal synaptic plasticity in Streptozotocin-induced diabetic rats. Diabetes 45:1259–1266
Biscaro B, Lindvall O, Tesco G, Ekdahl CT, Nitsch RM (2012) Inhibition of microglial activation protects Hippocampal neurogenesis and improves cognitive deficits in a transgenic mouse model for Alzheimer’s disease. Neurodegener Dis 9:187–198
Brands AMA, Biessels GJ, Haan EHF, Kappelle LJ, Kessels RPC (2005) The effects of type 1 diabetes on cognitive performance: A meta-analysis. Diabetes Care 28:726–735
Brownlee M (2005) The pathobiology of diabetic complications. A unifying mechanism Diabetes 54:1615–1625
Bruel-Jungerman E, Laroche S, Rampon C (2005) New neurons in the dentate gyrus are involved in the expression of enhanced long-term memory following environmental enrichment. Eur J Neurosci 21:513–521
Centenaro LA, Jaeger MC, Ilha J, Souza MA, Kalil-Gaspar PI, Cunha NB, Marcuzzo S, Achaval M (2011) Olfactory and respiratory lamina propria transplantation after spinal cord transection in rats: Effects on functional recovery and axonal regeneration. Brain Res 1426:54–72
Coleman ES, Judd RL, Hoel J, Dennis J, Posner P (2004) Effects of diabetes mellitus on astrocyte GFAP and glutamate transporters in the CNS. Glia 48:166–178
Coleman ES, Dennis JC, Braden TD, Judd RL, Posner P (2010) Insulin treatment prevents diabetes-induced alterations in astrocyte glutamate uptake and GFAP content in rats at 4 and 8 weeks of diabetes duration. Brain Res 1306:131–141
de Senna PN, Ilha J, Baptista PPA, do Nascimento PS, Leite MC, Paim MF, Gonçalves CA, Achaval M, Xavier LL (2011) Effects of physical exercise on spatial memory and astroglial alterations in the hippocampus of diabetic rats. Metab Brain Dis 26:269–279
Desrocher M, Rovet J (2004) Neurocognitive correlates of type 1 diabetes mellitus in childhood. Child Neuropsychol 10:36–52
Ekdahl CT, Kokaia Z, Lindvall O (2009) Brain inflammation and adult neurogenesis: The dual role of microglia. Neuroscience 158:1021–1029
Estrada FS, Hernández VS, López-Hernández E, Corona-Morales AA, Solís H, Escobar A, Zhang L (2012) Glial activation in a pilocarpine rat model for epileptogenesis: A morphometric and quantitative analysis. Neurosci Lett 514:51–56
Ferreira AFB, Real CC, Rodrigues AC, Alves AS, Britto LRG (2011) Short-term, moderate exercise is capable of inducing structural, bdnf-independent Hippocampal plasticity. Brain Res 1425:111–122
Fox C, Merali Z, Harrison C (2006) Therapeutic and protective effect of environmental enrichment against psychogenic and neurogenic stress. Behav Brain Res 175:1–8
Frick KM, Fernandez SM (2003) Enrichment enhances spatial memory and increases synaptophysin levels in aged female mice. Neurobiol Aging 24:615–626
Gemma C, Bachstetter AD, Bickford PC (2010) Neuron-microglia dialogue and Hippocampal neurogenesis in the aged brain. Aging Dis 1:232–244
Gispen WH, Biessels GJ (2000) Cognition and synaptic plasticity in diabetes mellitus. Trends Neurosci 23:542–549
Grillo CA, Piroli GG, Wood GE, Reznikov LR, McEwen BS, Reagan LP (2005) Immunocytochemical analysis of synaptic proteins provides new insights into diabetes-mediated plasticity in the rat hippocampus. Neuroscience 136:477–486
Grote HE, Hannan AJ (2007) Regulators of adult neurogenesis in the healthy and diseased brain. Clin Exp Pharmacol Physiol 34:533–545
Jackson-Guilford J, Leander JD, Nisenbaum LK (2000) The effect of Streptozotocin-induced diabetes on cell proliferation in the rat dentate gyrus. Neurosci Lett 293:91–94
Kamal A, Biessels GJ, Urban IJA, Gispen WH (1999) Hippocampal synaptic plasticity in streptozotocindiabetic rats: Impairment of long-term Potentiation and facilitation of long-term depression. Neuroscience 90:737–745
Kim SU, Vellis J (2005) Microglia in health and disease. J Neurosci Res 81:302–313
Kohman RA, Rhodes JS (2013) Neurogenesis, inflammation and behavior. Brain Behav Immun 27:22–32
Ladeby R, Wirenfeldt M, Garcia-Ovejero D, Fenger C, Dissing-Olesen L, Dalmau I, Finsen B (2005) Microglial cell population dynamics in the injured adult central nervous system. Brain Res Rev 48:196–206
Leggio MG, Mandolesi L, Federico F, Spirito F, Ricci B, Gelfo F, Petrosini L (2005) Environmental enrichment promotes improved spatial abilities and enhanced dendritic growth in the rat. Behav Brain Res 163:78–90
Malberg JE, Eisch AJ, Nestler EJ, Duman RS (2000) Chronic antidepressant treatment increases neurogenesis in adult rat hippocampus. J Neurosci 20:9104–9110
Messier C (2005) Impact of impaired glucose tolerance and type 2 diabetes on cognitive aging. Neurobiol Aging 26:26–30
Morley-Fletcher S, Rea M, Maccari S, Laviola G (2003) Environmental enrichment during adolescence reverses the effects of prenatal stress on play behaviour and HPA axis reactivity in rats. Eur J Neurosci 18:3367–3374
Napoli I, Neumann H (2009) Microglial clearance function in health and disease. Neuroscience 158:1030–1038
Nascimento PS, Lovatel GA, Barbosa S, Ilha J, Centenaro LA, Malysz T, Xavier LL, Schaan BD, Achaval M (2011) Treadmill training improves motor skills and increases tyrosine hydroxylase immunoreactivity in the substantia nigra pars compacta in diabetic rats. Brain Res 1382:173–180
Nithianantharajah J, Hannan AJ (2006) Enriched environments, experience dependent plasticity and disorders of the nervous system. Nat Rev Neurosci 7:697–709
Nithianantharajah J, Levis H, Murphy M (2004) Environmental enrichment results in cortical and subcortical changes in levels of synaptophysin and PSD-95 proteins. Neurobiol Learn Mem 81:200–210
Nitta A, Murai R, Suzuki N, Ito H, Nomoto H, Katoh G, Furukawa Y, Furukawa S (2002) Diabetic neuropathies in brain are induced by deficiency of BDNF. Neurotoxicol Teratol 24:695–701
Nowakowski RS, Lewin SB, Miller MW (1989) Bromodeoxyuridine immunohistochemical determination of the lengths of the cell cycle and the DNA- synthetic phase for an anatomically defined population. J Neurocytol 18:311–318
Paxinos G, Watson C (1982) The rat brain in stereotaxic coordinates. Academic, Sydney
Pereira LO, Strapasson ACP, Nabinger PM, Achaval M, Netto CA (2008) Early enriched housing results in partial recovery of memory deficits in female, but not in male, rats after neonatal hypoxia–ischemia. Brain Res 1218:257–266
Piazza FV, Pinto GV, Trott G, Marcuzzo S, Gomez R, Fernandes MC (2011) Enriched environment prevents memory deficits in type 1 diabetic rats. Behav Brain Res 217:16–20
Revsin Y, Rekers NV, Louwe MC, Saravia FE, Nicola AF, Kloet ER, Oitz MS (2009) Glucocorticoid receptor blockade normalizes Hippocampal alterations and cognitive impairment in Streptozotocin-induced type 1 diabetes mice. Neuropsychopharmacology 34:747–758
Rhoden EL, Rhoden CR (2006) Princípios e técnicas em experimentação animal. UFRGS, Porto Alegre
Sima AAF (2010) Encephalopathies: The emerging diabetic complications. Acta Diabetol 47:279–293
Sima AAF, Zhang W, Kreipke CW, Rafols JA, Hoffman WH (2009a) Inflammation in diabetic encephalopathy is prevented by C-peptide. Rev Diabet Stud 6:37–42
Sima AAF, Zhang W, Muzik O, Kreipke CW, Rafols JA, Hoffman WH (2009b) Sequential abnormalities in type 1 diabetic encephalopathy and the effects of C-peptide. Rev Diabet Stud 6:211–222
Stranahan AM, Arumugam TV, Cutler RG, Lee K, Egan JM, Mattson MP (2008) Diabetes impairs hippocampal function through glucocorticoid-mediated effects on new and mature neurons. Nat Neurosci 11:309–317
Streit WJ, Walter SA, Pennell NA (1999) Reactive microgliosis. Prog Neurobiol 57:563–581
Taupin P (2007) Brdu immunohistochemistry for studying adult neurogenesis: Paradigms, pitfalls, limitations, and validation. Brain Res Rev 53:198–214
Valente T, Gella A, Fernàndez-Busquets X, Unzeta M, Durany N (2010) Immunohistochemical analysis of human brain suggests pathological synergism of Alzheimer’s disease and diabetes mellitus. Neurobiol Dis 37:67–76
Veena J, Srikumar BN, Mahati K, Bhagya V, Raju TR, Rao BSS (2009a) Enriched environment restores hippocampal cell proliferation and ameliorates cognitive deficits in chronically stressed rats. J Neurosci Res 87:831–843
Veena J, Srikumar BN, Raju TR, Rao BSS (2009b) Exposure to enriched environment restores the survival and differentiation of new born cells in the hippocampus and ameliorates depressive symptoms in chronically stressed rats. Neurosci Lett 455:178–182
Wessels AM, Scheltens P, Barkhof F, Heine RJ (2008) Hyperglycaemia as a determinant of cognitive decline in patients with type 1 diabetes. Eur J Pharmacol 585:88–96
Will B, Galani R, Kelche C, Rosenzweig MR (2004) Recovery from brain injury in animals: Relative efficacy of environmental enrichment, physical exercise or formal training (1990–2002). Prog Neurobiol 72:167–182
Xavier LL, Viola GG, Ferraz AC, Da Cunha C, Deonizio JMD, Netto CA, Achaval M (2005) A simple and fast densitometric method for the analysis of tyrosine hydroxylase immunoreactivity in the substantia nigra pars compacta and in the ventral tegmental area. Brain Res Protoc 16:58–64
Yirmiya R, Goshen I (2011) Immune modulation of learning, memory, neural plasticity and neurogenesis. Brain Behav Immun 25:181–213
Ziv Y, Ron N, Butovsky O, Landa G, Sudai E, Greenberg N, Cohen H, Kipnis J, Schwartz M (2006) Immune cells contribute to the maintenance of neurogenesis and spatial learning abilities in adulthood. Nat Neurosci 9:268–275
Acknowledgments
The authors would like to thank the members of Laboratory of Comparative Histophysiology, the Electron Microscopy Center of the Federal University of Rio Grande do Sul for the microscopy analyzes and Henrique B. Biehl for his technical assistance. The authors are also grateful for the financial support provided by CNPq, CAPES, FAPERGS 10/0304-2, UFRGS and Matilde Achaval, MD, PhD, CNPq1A researcher.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Piazza, F.V., Segabinazi, E., Centenaro, L.A. et al. Enriched environment induces beneficial effects on memory deficits and microglial activation in the hippocampus of type 1 diabetic rats. Metab Brain Dis 29, 93–104 (2014). https://doi.org/10.1007/s11011-013-9467-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-013-9467-2