Abstract
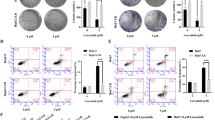
Tanshinone IIA (TSIIA), a multi-pharmaceutical compound, has been demonstrated to have anti-tumor properties. This study explores the potential regulatory mechanism of TSIIA on non-small cell lung cancer (NSCLC) progression. The cytotoxicity of TSIIA was evaluated by MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide) and LDH (lactate dehydrogenase) assays. Expression levels of circ_0020123 (hsa_circ_0020123) and microRNA-1299 (miR-1299) were assessed by quantitative real-time polymerase chain reaction (qRT-PCR). Cell proliferation, migration, invasion, and apoptosis were analyzed by MTT, colony formation, transwell, wound-healing, or flow cytometry assays. The relationship between miR-1299 and circ_0020123 or HMGB3 (high mobility group box 3) was verified by the dual-luciferase reporter and/or RNA immunoprecipitation (RIP) assays. Protein level of HMGB3 was measured by western blotting. The relationship between TSIIA and circ_0020123 was confirmed by xenograft assay. TSIIA reduced xenograft tumor growth in vivo and repressed proliferation, migration, invasion, and facilitated apoptosis of NSCLC cells in vitro. TSIIA reduced circ_0020123 and HMGB3 expression, whereas elevated miR-1299 expression in NSCLC cells. Circ_0020123 knockdown enhanced the repressive influence of TSIIA treatment on the malignancy of NSCLC cells in vitro and in vivo. Circ_0020123 sponged miR-1299 to regulate HMGB3 expression under TSIIA treatment. MiR-1299 inhibitor reversed circ_0020123 knockdown-mediated influence on malignant behaviors of NSCLC cells under TSIIA treatment. HMGB3 elevation offset the suppressive impact of miR-1299 mimic on the malignancy of NSCLC cells under TSIIA treatment. TSIIA curbed NSCLC progression by the circ_0020123/miR-1299/HMGB3 axis, manifesting that the TSIIA/circ_0020123/miR-1299/HMG regulatory network might be a potential treatment strategy for NSCLC.








Similar content being viewed by others
Data availability
The analyzed data sets generated during the present study are available from the corresponding author on reasonable request.
Abbreviations
- TSIIA:
-
Tanshinone IIA
- NSCLC:
-
Non-small cell lung cancer
- hsa_circ_0020123:
-
Circ_0020123
- qRT-PCR:
-
Quantitative real-time polymerase chain reaction
- miR-1299:
-
MicroRNA-1299
- RIP:
-
RNA immunoprecipitation
- TSIIA:
-
Tanshinone IIA
- PI:
-
Propidium iodide
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424
Miller KD, Nogueira L, Mariotto AB, Rowland JH, Yabroff KR, Alfano CM, Jemal A, Kramer JL, Siegel RL (2019) Cancer treatment and survivorship statistics, 2019. CA Cancer J Clin 69(5):363–385
Heigener DF, Reck M (2018) Lung cancer in 2017: giant steps and stumbling blocks. Nat Rev Clin Oncol 15(2):71–72
Ernani V, Steuer CE, Jahanzeb M (2017) The end of nihilism: systemic therapy of advanced non-small cell lung cancer. Annu Rev Med 68:153–168
Wang Y, Zhang Q, Chen Y, Liang C-L, Liu H, Qiu F, Dai Z (2020) Antitumor effects of immunity-enhancing traditional Chinese medicine. Biomed Pharmacother 121:109570
Xiang Y, Guo Z, Zhu P, Chen J, Huang Y (2019) Traditional Chinese medicine as a cancer treatment: modern perspectives of ancient but advanced science. Cancer Med 8(5):1958–1975
Ren J, Fu L, Nile SH, Zhang J, Kai G (2019) Salvia miltiorrhiza in treating cardiovascular diseases: a review on its pharmacological and clinical applications. Front Pharmacol 10:753
Zhou ZY, Zhao WR, Zhang J, Chen XL, Tang JY (2019) Sodium tanshinone IIA sulfonate: a review of pharmacological activity and pharmacokinetics. Biomed Pharmacother 118:109362
Liao XZ, Gao Y, Huang S, Chen ZZ, Sun LL, Liu JH, Chen HR, Yu L, Zhang JX, Lin LZ (2019) Tanshinone IIA combined with cisplatin synergistically inhibits non-small-cell lung cancer in vitro and in vivo via down-regulating the phosphatidylinositol 3-kinase/Akt signalling pathway. Phytother Res 33(9):2298–2309
Wang R, Luo Z, Zhang H, Wang T (2019) Tanshinone IIA reverses gefitinib-resistance in human non-small-cell lung cancer via regulation Of VEGFR/Akt pathway. Onco Targets Ther 12:9355–9365
Zhang HS, Zhang FJ, Li H, Liu Y, Du GY, Huang YH (2016) Tanshinone IIA inhibits human esophageal cancer cell growth through miR-122-mediated PKM2 down-regulation. Arch Biochem Biophys 598:50–56
Chiu TL, Su CC (2017) Tanshinone IIA increases protein expression levels of PERK, ATF6, IRE1α, CHOP, caspase-3 and caspase-12 in pancreatic cancer BxPC-3 cell-derived xenograft tumors. Mol Med Rep 15(5):3259–3263
Yu J, Wang X, Li Y, Tang B (2017) Tanshinone IIA suppresses gastric cancer cell proliferation and migration by downregulation of FOXM1. Oncol Rep 37(3):1394–1400
Kristensen LS, Andersen MS, Stagsted LVW, Ebbesen KK, Hansen TB, Kjems J (2019) The biogenesis, biology and characterization of circular RNAs. Nat Rev Genet 20(11):675–691
Li Y, Ge Y-Z, Xu L, Jia R (2020) Circular RNA ITCH: a novel tumor suppressor in multiple cancers. Life Sci 254:117176
Lei B, Tian Z, Fan W, Ni B (2019) Circular RNA: a novel biomarker and therapeutic target for human cancers. Int J Med Sci 16(2):292–301
Chi G, Yang F, Xu D, Liu W (2020) Silencing hsa_circ_PVT1 (circPVT1) suppresses the growth and metastasis of glioblastoma multiforme cells by up-regulation of miR-199a-5p. Artif Cells Nanomed Biotechnol 48(1):188–196
Peng L, Sang H, Wei S, Li Y, Jin D, Zhu X, Li X, Dang Y, Zhang G (2020) circCUL2 regulates gastric cancer malignant transformation and cisplatin resistance by modulating autophagy activation via miR-142-3p/ROCK2. Mol Cancer 19(1):156
Qu D, Yan B, Xin R, Ma T (2018) A novel circular RNA hsa_circ_0020123 exerts oncogenic properties through suppression of miR-144 in non-small cell lung cancer. Am J Cancer Res 8(8):1387–1402
Bi R, Wei W, Lu Y, Hu F, Yang X, Zhong Y, Meng L, Wang M, Jiang L, Xie X (2020) High hsa_circ_0020123 expression indicates poor progression to non-small cell lung cancer by regulating the miR-495/HOXC9 axis. Aging (Albany NY) 12(17):17343–17352
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25(4):402–408
Fu L, Han B, Zhou Y, Ren J, Cao W, Patel G, Kai G, Zhang J (2020) The anticancer properties of tanshinones and the pharmacological effects of their active ingredients. Front Pharmacol 11:193
Lin CY, Chang TW, Hsieh WH, Hung MC, Lin IH, Lai SC, Tzeng YJ (2016) Simultaneous induction of apoptosis and necroptosis by Tanshinone IIA in human hepatocellular carcinoma HepG2 cells. Cell Death Discov 2(1):16065
Jieensinue S, Zhu H, Li G, Dong K, Liang M, Li Y (2018) Tanshinone IIA reduces SW837 colorectal cancer cell viability via the promotion of mitochondrial fission by activating JNK-Mff signaling pathways. BMC Cell Biol 19(1):21
Lin C-Y, Tam Ly M, Yang SH, Lai S-C, Chang T-W, Lin I-H, Tzeng Y-J (2021) Tanshinone IIA shows higher antiproliferative activities than sinapic acid in 4 cancer cell lines and simultaneously induces apoptosis and necroptosis in human lung cancer A549 cells. Nat Prod Commun 16(10):1934578X211050521
Chang CC, Lai JS, Tsai CS, Ma SW, Lin JY, Huang LR, Lu CH, Liao EC, Ho TF (2013) Proapoptotic and TRAIL-sensitizing constituents isolated from Salvia militiorrhiza (Danshen). J Biosci Bioeng 116(4):516–523
Liao X-Z, Gao Y, Huang S, Chen Z-Z, Sun L-L, Liu J-H, Chen H-R, Yu L, Zhang J-X, Lin L-Z (2019) Tanshinone IIA combined with cisplatin synergistically inhibits non-small-cell lung cancer in vitro and in vivo via down-regulating the phosphatidylinositol 3-kinase/Akt signalling pathway. Phytother res : PTR 33(9):2298–2309
Gao F, Li M, Liu W, Li W (2020) Inhibition of EGFR signaling and activation of mitochondrial apoptosis contribute to tanshinone IIA-mediated tumor suppression in non-small cell lung cancer cells. Onco Targets Ther 13:2757–2769
Wan J, Hao L, Zheng X, Li Z (2019) Circular RNA circ_0020123 promotes non-small cell lung cancer progression by acting as a ceRNA for miR-488-3p to regulate ADAM9 expression. Biochem Biophys Res Commun 515(2):303–309
Wang L, Zhao L, Wang Y (2020) Circular RNA circ_0020123 promotes non-small cell lung cancer progression by sponging miR-590-5p to regulate THBS2. Cancer Cell Int 20:387
Liang Z-Z, Guo C, Zou M-M, Meng P, Zhang T-T (2020) circRNA-miRNA-mRNA regulatory network in human lung cancer: an update. Cancer Cell Int 20:173
Zhang FB, Du Y, Tian Y, Ji ZG, Yang PQ (2019) MiR-1299 functions as a tumor suppressor to inhibit the proliferation and metastasis of prostate cancer by targeting NEK2. Eur Rev Med Pharmacol Sci 23(2):530–538
Liu LH, Tian QQ, Liu J (2019) Upregulation of hsa_circ_0136666 contributes to breast cancer progression by sponging miR-1299 and targeting CDK6. J Cell Biochem 120(8):12684–12693
Zhou Y, Yong H, Pei Y, Li K, Lou X, Wu Y, Dong X, Wang W, Li N, Zhang D et al (2020) miR-1299/NOTCH3/TUG1 feedback loop contributes to the malignant proliferation of ovarian cancer. J Cell Biochem 44(2):438–448
Cao S, Li L, Li J, Zhao H (2020) MiR-1299 impedes the progression of non-small-cell lung cancer through EGFR/PI3K/AKT signaling pathway. Onco Targets Ther 13:7493–7502
Agresti A, Bianchi ME (2003) HMGB proteins and gene expression. Curr Opin Genet Dev 13(2):170–178
Zhang Z, Chang Y, Zhang J, Lu Y, Zheng L, Hu Y, Zhang F, Li X, Zhang W, Li X (2017) HMGB3 promotes growth and migration in colorectal cancer by regulating WNT/β-catenin pathway. PLoS ONE 12(7):e0179741
Mukherjee A, Huynh V, Gaines K, Reh WA, Vasquez KM (2019) Targeting the high-mobility group box 3 protein sensitizes chemoresistant ovarian cancer cells to cisplatin. PLoS ONE 79(13):3185–3191
Zhuang S, Yu X (2020) High mobility group box 3 promotes cervical cancer proliferation by regulating Wnt/β-catenin pathway. J Gynecol Oncol 31(6):e91
Li Y, Ma Y, Zhang T, Feng C, Liu Y (2020) High-mobility group box 3 (HMGB3) silencing inhibits non-small cell lung cancer development through regulating Wnt/β-catenin pathway. Biol Chem 401(10):1191–1198
Wang J, Sheng Z, Cai Y (2019) Effects of microRNA-513b on cell proliferation, apoptosis, invasion, and migration by targeting HMGB3 through regulation of mTOR signaling pathway in non-small-cell lung cancer. J Cell Physiol 234(7):10934–10941
Acknowledgements
Not applicable
Funding
This work was not received funds.
Author information
Authors and Affiliations
Contributions
Conceptualization and Methodology: XY and WS; Formal analysis and Data curation: CX, NY and QL; Validation and Investigation: FS and XY; Writing—original draft preparation and Writing—review and editing: FS, XY, WS and CX; Approval of final manuscript: all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethical approval
The present study was approved by the ethical review committee of Yantai Hospital of Traditional Chinese Medicine.
Consent to participate
Written informed consent was obtained from all enrolled patients.
Consent for publication
Patients agree to participate in this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, F., Yang, X., Song, W. et al. Tanshinone IIA (TSIIA) represses the progression of non-small cell lung cancer by the circ_0020123/miR-1299/HMGB3 pathway. Mol Cell Biochem 478, 1973–1986 (2023). https://doi.org/10.1007/s11010-022-04646-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-022-04646-3