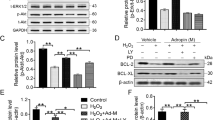
Abstract
Myocardial infarction (MI) damages cardiomyocytes permanently and compromises cardiac function. Mesenchymal stem cells (MSCs) with the potential to differentiate into multiple lineages are considered as one of the best options for the treatment of MI. However, aging affects their regeneration capability. With age, reactive oxygen species (ROS) accumulate in cells ultimately causing cell death. To successfully utilize these stem cells in clinic, novel strategies to improve their functional capability should be explored. In this study, we aimed to enhance the cardiac regeneration potential of bone marrow MSCs derived from aging rats by treating them with antioxidants, rutin or quercetagetin in separate in vivo experiments. Oxidative stress was induced by treating MSCs of young and aging rats with different concentrations of H2O2 which resulted in an increase in the ROS level. MSCs were treated with rutin or quercetagetin at varying concentrations and exposed to H2O2. It was observed that both antioxidants significantly (P < 0.001) suppressed H2O2-induced intracellular ROS accumulation in a dose-dependent manner. An optimized concentration of 10 µM rutin or quercetagetin was used for the in vivo experiments. MI models were developed in aging rats by ligation of left anterior descending artery and treated MSCs were transplanted in the MI models. Echocardiography was performed after 2 and 4 weeks of cell transplantation to evaluate the functional status of the infarcted heart and histological analysis was performed after 4 weeks to assess cardiac regeneration. Significant improvement was observed in cardiac parameters including LVEF% (P < 0.001), LVFS% (P < 0.01 and P < 0.001), LVIDd (P < 0.01 and P < 0.001), LVIDs (P < 0.001), LVEDV (P < 0.001) and LVESV (P < 0.001) in the treated young as well as aging MSCs. It is concluded from these findings that rutin and quercetagetin treatment enhance the regeneration efficiency of young and aging MSCs in vivo. These antioxidants can be effectively utilized to improve cellular therapy for myocardial infarction by suppressing ROS production.





Similar content being viewed by others
Data availability
Enquiries about data availability should be directed to the authors.
References
WHO (2018) The top 10 causes of death. World Health Organization; 24 Maggio. http://www.who.int/en/news-room/fact-sheets/detail/the-top-10-causes-of-death. Accessed May 2022
Katwa LC, Mendoza C, Clements M (2022) CVD and COVID-19: emerging roles of cardiac fibroblasts and myofibroblasts. Cells 11:1316
Lu L, Liu M, Sun R, Zheng Y, Zhang P (2015) Myocardial infarction: symptoms and treatments. Cell Biochem Biophys 72:865–867
Spadaccio C, Benedetto U (2018) Coronary artery bypass grafting (CABG) vs percutaneous coronary intervention (PCI) in the treatment of multivessel coronary disease: quo vadis? a review of the evidences on coronary artery disease. Ann Cardiothorac Surg 7(4):506–515
Kim YH, Her AY, Jeong MH, Kim BK, Lee SY, Hong SJ, Shin DH, Kim JS, Ko YG, Choi D, Hong MK (2019) Comparison between beta-blockers with angiotensin-converting enzyme inhibitors and beta-blockers with angiotensin II type I receptor blockers in ST-segment elevation myocardial infarction after successful percutaneous coronary intervention with drug-eluting stents. Cardiovas Drugs Ther 33:55–67
Fuchs M, Schibilsky D, Zeh W, Berchtold-Herz M, Beyersdorf F, Siepe M (2019) Does the heart transplant have a future? Eur J of Cardiothorac Surg 55:38–48
Kypridemos C, Collins B, McHale P, Bromley H, Parvulescu P, Capewell S, O’Flaherty M (2018) Future cost-effectiveness and equity of the NHS health check cardiovascular disease prevention programme: microsimulation modelling using data from Liverpool UK. PLoS med 15:e1002573
Trounson A, McDonald C (2015) Stem cell therapies in clinical trials: progress and challenges. Cell Stem Cell 17:11–22
Ma S, Xie N, Li W, Yuan B, Shi Y, Wang Y (2014) Immunobiology of mesenchymal stem cells. Cell Death Differ 21:216–225
Gudleviciene Z, Kundrotas G, Liudkeviciene R, Rascon J, Jurga M (2015) Quick and effective method of bone marrow mesenchymal stem cell extraction. Open Med 10:44–49
Gattazzo F, Urciuolo A, Bonaldo P (2014) Extracellular matrix: a dynamic microenvironment for stem cell niche. Biochim Biophys Acta 1840:2506–2519
El-Sawah SG, Althobaiti F, Aldhahrani A, Fayad E, Abdel-Dayem MA, Amen RM, Shabana ES, El-Hallous EI, Rashwan HM (2021) Investigation of the antioxidant defensive role of both AD-MSCs and BM-MSCs in modulating the alteration in the oxidative stress status in various STZ-diabetic rats’ tissues. Biocell 45:1561–1568
El-Sawah SG, Althobaiti F, Rashwan HM, Aldhahrani A, Abdel-Dayem MA et al (2022) Anti-inflammatory and antioxidant potential capacities of AD-MSCs and BM-MSCs in suppressing pancreatic β-cells auto-immunity and apoptosis in rats with T1DM induced model. Biocell 46:745–757
Cai M, Shen R, Song L, Lu M, Wang J, Zhao S, Tang Y, Meng X, Li Z, He ZX (2016) Bone marrow mesenchymal stem cells (BM-MSCs) improve heart function in swine myocardial infarction model through paracrine effects. Sci Rep 6:28250
Gallina C, Turinetto V, Giachino C (2015) A new paradigm in cardiac regeneration: the mesenchymal stem cell secretome. Stem Cells Int 2015:765846
Silva WA Jr, Covas DT, Panepucci RA, Proto-Siqueira R, Siufi JL, Zanette DL, Santos AR, Zago MA (2003) The profile of gene expression of human marrow mesenchymal stem cells. Stem Cells 21:661–669
Orlic D, Kajstura J, Chimenti S, Jakoniuk I, Anderson SM, Li B, Pickel J, McKay R, Nadal-Ginard B, Bodine DM, Leri A (2001) Bone marrow cells regenerate infarcted myocardium. Nature 410:701–705
Amado LC, Saliaris AP, Schuleri KH, John MS, Xie JS, Cattaneo S, Durand DJ, Fitton T, Kuang JQ, Stewart G, Lehrke S et al (2005) Cardiac repair with intramyocardial injection of allogeneic mesenchymal stem cells after myocardial infarction. Proc Natl Acad Sci USA 102:11474–11479
Teo AK, Vallier L (2010) Emerging use of stem cells in regenerative medicine. Biochem J 428:11–23
Xu J, Qian J, Xie X, Lin L, Zou Y, Fu M, Huang Z, Zhang G, Su Y, Ge J (2012) High density lipoprotein protects mesenchymal stem cells from oxidative stress-induced apoptosis via activation of the PI3K/Akt pathway and suppression of reactive oxygen species. Int J Mol Sci 13:17104–17120
Wei H, Li Z, Hu S, Chen X, Cong X (2010) Apoptosis of mesenchymal stem cells induced by hydrogen peroxide concerns both endoplasmic reticulum stress and mitochondrial death pathway through regulation of caspases, p38 and JNK. J Cell Biochem 111:967–978
Asumda FZ, Chase PB (2011) Age-related changes in rat bone-marrow mesenchymal stem cell plasticity. BMC Cell Biol 12(1):44
Raggi C, Berardi AC (2012) Mesenchymal stem cells, aging and regenerative medicine. Muscles, Ligaments Tendons J 2(3):239–242
Linton PJ, Dorshkind K (2004) Age-related changes in lymphocyte development and function. Nat Immunol 5(2):133–139
Pang WW, Price EA, Sahoo D, Beerman I, Maloney WJ, Rossi DJ, Schrier SL, Weissman IL (2011) Human bone marrow hematopoietic stem cells are increased in frequency and myeloid-biased with age. Proc Natl Acad Sci USA 108(50):20012–20017
Domingues CC, Kundu N, Kropotova Y, Ahmadi N, Sen S (2019) Antioxidant-upregulated mesenchymal stem cells reduce inflammation and improve fatty liver disease in diet-induced obesity. Stem Cell Res Ther 10:280
Dal-Ros S, Bronner C, Auger C, Schini-Kerth VB (2012) Red wine polyphenols improve an established aging-related endothelial dysfunction in the mesenteric artery of middle-aged rats: role of oxidative stress. Biochem Biophys Res Commun 419(2):381–387
Khodja NI, Chataigneau T, Auger C, Schini-Kerth VB (2012) Grape-derived polyphenols improve aging-related endothelial dysfunction in rat mesenteric artery: role of oxidative stress and the angiotensin system. PLoS ONE 7(2):e32039
Du G, Sun L, Zhao R, Du L, Song J, Zhang L, He G, Zhang Y, Zhang J (2016) Polyphenols: potential source of drugs for the treatment of ischaemic heart disease. Pharmacol Ther 162:23–34
Janbaz KH, Saeed SA, Gilani AH (2002) Protective effect of rutin on paracetamol- and CCl4-induced hepatotoxicity in rodents. Fitoterapia 73:557–563
Ahmed OM, Moneim AA, Yazid IA, Mahmoud AM (2010) Antihyperglycemic, antihyperlipidemic and antioxidant effects and the probable mechanisms of action of ruta graveolens infusion and rutin in nicotinamide-streptozotocin-induced diabetic rats. Diabetol Croat 39(1):15–35
Kamalakkannan N, Prince PS (2006) Antihyperglycaemic and antioxidant effect of rutin, a polyphenolic flavonoid, in streptozotocin-induced diabetic wistar rats. Basic Clin Pharmacol Toxicol 98(1):97–103
Srivastava D, Ieda M (2012) Critical factors for cardiac reprogramming. Circ Res 111:5–8
La Casa C, Villegas I, De La Lastra CA, Motilva V, Calero MM (2000) Evidence for protective and antioxidant properties of rutin, a natural flavone, against ethanol induced gastric lesions. J Ethnopharmacol 71(1):45–53
Kamel KM, El-Raouf A, Ola M, Metwally SA, El-Latif A, Hekma A, El-sayed ME (2014) Hesperidin and Rutin, antioxidant citrus flavonoids, attenuate cisplatin-induced nephrotoxicity in rats. J Biochem Mol Toxicol 28(7):312–319
Sun J, Wang H, Liu B, Shi W, Shi J, Zhang Z, Xing J (2017) Rutin attenuates H2O2-induced oxidation damage and apoptosis in Leydig cells by activating PI3K/Akt signal pathways. Biomed Pharmacother 88:500–506
Yang X, Kang SM, Jeon BT, Kim YD, Ha JH, Kim YT, Jeon YJ (2011) Isolation and identification of an antioxidant flavonoid compound from citrus-processing by-product. J Sci Food Agric 91(10):1925–1927
Kang GJ, Han SC, Ock JW, Kang HK, Yoo ES (2013) Anti-inflammatory effect of quercetagetin, an active component of immature citrus unshiu HaCaT human keratinocytes. Biomol Ther 21(2):138–145
Haneef K, Naeem N, Khan I, Iqbal H, Kabir N, Jamall S, Zahid M, Salim A (2014) Conditioned medium enhances the fusion capability of rat bone marrow mesenchymal stem cells and cardiomyocytes. Mol Biol Rep 4:3099–3112
Hafer K, Iwamoto KS, Schiestl RH (2008) Refinement of the dichlorofluorescein assay for flow cytometric measurement of reactive oxygen species in irradiated and bystander cell populations. Radiation Res 169:460–468
Zhou L, Jing Y, Styblo M, Chen Z, Waxman S (2005) Glutathione-S-transferase π inhibits As2O3-induced apoptosis in lymphoma cells: involvement of hydrogen peroxide catabolism. Blood 105:1198–1203
Khan I, Ali A, Akhter MA, Naeem N, Chotani MA, Mustafa T, Salim A (2016) Preconditioning of mesenchymal stem cells with 2,4-dinitrophenol improves cardiac function in infarcted rats. Life Sci 162:60–69
Bruna F, Contador D, Conget P, Erranz B, Sossa CL, Arango-Rodríguez ML (2016) Regenerative potential of mesenchymal stromal cells: age-related changes. Stem Cells Int 2016:1461648
Zeng W, Xiao J, Zheng G, Xing F, Tipoe GL, Wang X, He C, Chen ZY, Liu Y (2015) Antioxidant treatment enhances human mesenchymal stem cell anti-stress ability and therapeutic efficacy in an acute liver failure model. Sci Rep 5:11100
Sethe S, Scutt A, Stolzing A (2006) Aging of mesenchymal stem cells. Ageing Res Rev 5(1):91–116
Atashi F, Modarressi A, Pepper MS (2015) The role of reactive oxygen species in mesenchymal stem cell adipogenic and osteogenic differentiation: a review. Stem Cells Dev 24:1150–1163
Ma C, Pi C, Yang Y, Lin L, Shi Y, Li Y, Li Y, He X (2017) Nampt expression decreases age-related senescence in rat bone marrow mesenchymal stem cells by targeting sirt1. PLoS ONE 12:e0170930
Fafian-Labora J, Fernández-Pernas P, Fuentes I, De Toro J, Oreiro N, Sangiao-Alvarellos S, Mateos J, Arufe MC (2015) Influence of age on rat bone-marrow mesenchymal stem cells potential. Sci Rep 5:16765
Klotz B, Mentrup B, Regensburger M, Zeck S, Schneidereit J, Schupp N, Linden C, Merz C, Ebert R, Jakob F (2012) 1, 25-dihydroxyvitamin D3 treatment delays cellular aging in human mesenchymal stem cells while maintaining their multipotent capacity. PLoS ONE 7:e29959
Denu RA, Hematti P (2016) Effects of oxidative stress on mesenchymal stem cell biology. Oxid Med Cell Longev 2016:2989076
Shuai Y, Liao L, Su X, Yu Y, Shao B, Jing H (2016) Melatonin treatment improves mesenchymal stem cells therapy by preserving stemness during long-term in vitro expansion. Theranostics 6:1899–1917
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
Each author contributed significantly and meets the criteria for authorship and assumes the corresponding responsibility. TM conducted all the experiments and wrote the first draft, with technical and analytical assistance from IK in the in vivo work, HI assisted in in vitro studies. Both IK and HI contributed in manuscript writing. SU conducted some of the experiments. NN assisted in the in vivo work, especially echocardiography. SF provided the expertise related to the chemical compounds. AS conceived the idea, critically analyzed the results and finalized the manuscript. All authors are in agreement with the content of the manuscript and reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mustafa, T., Khan, I., Iqbal, H. et al. Rutin and quercetagetin enhance the regeneration potential of young and aging bone marrow-derived mesenchymal stem cells in the rat infarcted myocardium. Mol Cell Biochem 478, 1759–1770 (2023). https://doi.org/10.1007/s11010-022-04628-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-022-04628-5