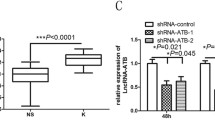
Abstract
Keloid is a common dermis tumor, occurring repeatedly, affecting the quality of patients’ life. Long non-coding RNAs (lncRNAs) have crucial regulatory capacities in skin scarring formation and subsequent scar carcinogenesis. The intention of this study was to investigate the mechanism and function of GNAS antisense-1 (GNAS-AS1) in keloids. Clinical samples were collected to evaluate the expression of GNAS-AS1, RUNX2, and miR-188-5p by qRT-PCR. The proliferation, migration, and invasion of HKF cells were detected by CCK-8, wound healing, and Transwell assays. The expression levels of mRNA and protein were examined through qRT-PCR and Western blot assay. Luciferase reporter assay was used to identify the binding relationship among GNAS-AS1, miR-188-5p, and Runt-related transcription factor 2 (RUNX2). GNAS-AS1 and RUNX2 expressions were remarkably enhanced, and miR-188-5p expression was decreased in keloid clinical tissues and HKF cells. GNAS-AS1 overexpression promoted cells proliferation, migration, and invasion, while GNAS-AS1 knockdown had the opposite trend. Furthermore, overexpression of GNAS-AS1 reversed the inhibitory effect of 5-FU on cell proliferation, migration, and invasion. MiR-188-5p inhibition or RUNX2 overexpression could enhance the proliferation, migration, and invasion of HKF cells. GNAS-AS1 targeted miR-188-5p to regulate RUNX2 expression. In addition, the inhibition effects of GNAS-AS1 knockdown on HKF cells could be reversed by inhibition of miR-188-5p or overexpression of RUNX2, while RUNX2 overexpression eliminated the suppressive efficaciousness of miR-188-5p mimics on HKF cells growth. GNAS-AS1 knockdown could regulate the miR-188-5p/RUNX2 signaling axis to inhibit the growth and migration in keloid cells. It is suggested that GNAS-AS1 may become a new target for the prevention and treatment of keloid.







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Huang Y, Wang Y, Lin L, Wang P, Jiang L, Liu J, Wang X (2020) Overexpression of miR-133a-3p inhibits fibrosis and proliferation of keloid fibroblasts by regulating IRF5 to inhibit the TGF-beta/Smad2 pathway. Mol Cell Probes 52:101563. https://doi.org/10.1016/j.mcp.2020.101563
Cui J, Li Z, Jin C, Jin Z (2020) Knockdown of fibronectin extra domain B suppresses TGF-beta1-mediated cell proliferation and collagen deposition in keloid fibroblasts via AKT/ERK signaling pathway. Biochem Biophys Res Commun 526:1131–1137. https://doi.org/10.1016/j.bbrc.2020.04.021
Naylor MC, Brissett AE (2012) Current concepts in the etiology and treatment of keloids. Facial Plast Surg 28:504–512. https://doi.org/10.1055/s-0032-1325644
Andrews JP, Marttala J, Macarak E, Rosenbloom J, Uitto J (2016) Keloids: the paradigm of skin fibrosis—pathomechanisms and treatment. Matrix Biol 51:37–46. https://doi.org/10.1016/j.matbio.2016.01.013
Marttala J, Andrews JP, Rosenbloom J, Uitto J (2016) Keloids: animal models and pathologic equivalents to study tissue fibrosis. Matrix Biol 51:47–54. https://doi.org/10.1016/j.matbio.2016.01.014
Bock O, Schmid-Ott G, Malewski P, Mrowietz U (2006) Quality of life of patients with keloid and hypertrophic scarring. Arch Dermatol Res 297:433–438. https://doi.org/10.1007/s00403-006-0651-7
Wang KC, Chang HY (2011) Molecular mechanisms of long noncoding RNAs. Mol Cell 43:904–914. https://doi.org/10.1016/j.molcel.2011.08.018
Kretz M, Webster DE, Flockhart RJ, Lee CS, Zehnder A, Lopez-Pajares V, Qu K, Zheng GX, Chow J, Kim GE, Rinn JL, Chang HY, Siprashvili Z, Khavari PA (2012) Suppression of progenitor differentiation requires the long noncoding RNA ANCR. Genes Dev 26:338–343. https://doi.org/10.1101/gad.182121.111
Bernard JJ, Cowing-Zitron C, Nakatsuji T, Muehleisen B, Muto J, Borkowski AW, Martinez L, Greidinger EL, Yu BD, Gallo RL (2012) Ultraviolet radiation damages self noncoding RNA and is detected by TLR3. Nat Med 18:1286–1290. https://doi.org/10.1038/nm.2861
Khaitan D, Dinger ME, Mazar J, Crawford J, Smith MA, Mattick JS, Perera RJ (2011) The melanoma-upregulated long noncoding RNA SPRY4-IT1 modulates apoptosis and invasion. Cancer Res 71:3852–3862. https://doi.org/10.1158/0008-5472.CAN-10-4460
Weinstein LS, Liu J, Sakamoto A, Xie T, Chen M (2004) Minireview: GNAS: normal and abnormal functions. Endocrinology 145:5459–5464. https://doi.org/10.1210/en.2004-0865
Liu J, Yu S, Litman D, Chen W, Weinstein LS (2000) Identification of a methylation imprint mark within the mouse Gnas locus. Mol Cell Biol 20:5808–5817. https://doi.org/10.1128/MCB.20.16.5808-5817.2000
Williamson CM, Ball ST, Dawson C, Mehta S, Beechey CV, Fray M, Teboul L, Dear TN, Kelsey G, Peters J (2011) Uncoupling antisense-mediated silencing and DNA methylation in the imprinted Gnas cluster. PLoS Genet 7:e1001347. https://doi.org/10.1371/journal.pgen.1001347
Regard JB, Cherman N, Palmer D, Kuznetsov SA, Celi FS, Guettier JM, Chen M, Bhattacharyya N, Wess J, Coughlin SR, Weinstein LS, Collins MT, Robey PG, Yang Y (2011) Wnt/beta-catenin signaling is differentially regulated by Galpha proteins and contributes to fibrous dysplasia. Proc Natl Acad Sci USA 108:20101–20106. https://doi.org/10.1073/pnas.1114656108
Regard JB, Malhotra D, Gvozdenovic-Jeremic J, Josey M, Chen M, Weinstein LS, Lu J, Shore EM, Kaplan FS, Yang Y (2013) Activation of Hedgehog signaling by loss of GNAS causes heterotopic ossification. Nat Med 19:1505–1512. https://doi.org/10.1038/nm.3314
Liu SQ, Zhou ZY, Dong X, Guo L, Zhang KJ (2020) LncRNA GNAS-AS1 facilitates ER+ breast cancer cells progression by promoting M2 macrophage polarization via regulating miR-433-3p/GATA3 axis. Biosci Rep. https://doi.org/10.1042/BSR20200626
Li Z, Feng C, Guo J, Hu X, Xie D (2020) GNAS-AS1/miR-4319/NECAB3 axis promotes migration and invasion of non-small cell lung cancer cells by altering macrophage polarization. Funct Integr Genomics 20:17–28. https://doi.org/10.1007/s10142-019-00696-x
Wang XQ, Xu H, Wang CH, Xie H (2020) Long non-coding RNA GNAS-AS1 promotes cell migration and invasion via regulating Wnt/beta-catenin pathway in nasopharyngeal carcinoma. Eur Rev Med Pharmacol Sci 24:3077–3084. https://doi.org/10.26355/eurrev_202003_20672
Liang X, Ma L, Long X, Wang X (2015) LncRNA expression profiles and validation in keloid and normal skin tissue. Int J Oncol 47:1829–1838. https://doi.org/10.3892/ijo.2015.3177
Hessam S, Sand M, Skrygan M, Gambichler T, Bechara FG (2017) Expression of miRNA-155, miRNA-223, miRNA-31, miRNA-21, miRNA-125b, and miRNA-146a in the Inflammatory pathway of Hidradenitis suppurativa. Inflammation 40:464–472. https://doi.org/10.1007/s10753-016-0492-2
Rupaimoole R, Slack FJ (2017) MicroRNA therapeutics: towards a new era for the management of cancer and other diseases. Nat Rev Drug Discov 16:203–222. https://doi.org/10.1038/nrd.2016.246
An G, Liang S, Sheng C, Liu Y, Yao W (2017) Upregulation of microRNA-205 suppresses vascular endothelial growth factor expression-mediated PI3K/Akt signaling transduction in human keloid fibroblasts. Exp Biol Med (Maywood) 242:275–285. https://doi.org/10.1177/1535370216669839
Yao X, Cui X, Wu X, Xu P, Zhu W, Chen X, Zhao T (2018) Tumor suppressive role of miR-1224-5p in keloid proliferation, apoptosis and invasion via the TGF-beta1/Smad3 signaling pathway. Biochem Biophys Res Commun 495:713–720. https://doi.org/10.1016/j.bbrc.2017.10.070
Wang M, Qiu R, Gong Z, Zhao X, Wang T, Zhou L, Lu W, Shen B, Zhu W, Xu W (2019) miR-188-5p emerges as an oncomiRNA to promote gastric cancer cell proliferation and migration via upregulation of SALL4. J Cell Biochem 120:15027–15037. https://doi.org/10.1002/jcb.28764
Wang M, Zhang H, Yang F, Qiu R, Zhao X, Gong Z, Yu W, Zhou B, Shen B, Zhu W (2020) miR-188-5p suppresses cellular proliferation and migration via IL6ST: a potential noninvasive diagnostic biomarker for breast cancer. J Cell Physiol 235:4890–4901. https://doi.org/10.1002/jcp.29367
Yang X, Wang P (2019) MiR-188-5p and MiR-141-3p influence prognosis of bladder cancer and promote bladder cancer synergistically. Pathol Res Pract 215:152598. https://doi.org/10.1016/j.prp.2019.152598
Zhang H, Qi S, Zhang T, Wang A, Liu R, Guo J, Wang Y, Xu Y (2015) miR-188-5p inhibits tumour growth and metastasis in prostate cancer by repressing LAPTM4B expression. Oncotarget 6:6092–6104. https://doi.org/10.18632/oncotarget.3341
Peng Y, Shen X, Jiang H, Chen Z, Wu J, Zhu Y, Zhou Y, Li J (2018) miR-188-5p suppresses gastric cancer cell proliferation and invasion via targeting ZFP91. Oncol Res 27:65–71. https://doi.org/10.3727/096504018X15191223015016
Zhu W, Wu X, Yang B, Yao X, Cui X, Xu P, Chen X (2019) Corrigendum to: miR-188-5p regulates proliferation and invasion via PI3K/Akt/MMP-2/9 signaling in keloids. Acta Biochim Biophys Sin (Shanghai) 51:980. https://doi.org/10.1093/abbs/gmz060
Xu Z, Guo B, Chang P, Hui Q, Li W, Tao K (2019) The differential expression of miRNAs and a preliminary study on the mechanism of miR-194-3p in keloids. Biomed Res Int 2019:8214923. https://doi.org/10.1155/2019/8214923
Li JH, Liu S, Zhou H, Qu LH, Yang JH (2014) starBase v2.0: decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA interaction networks from large-scale CLIP-Seq data. Nucleic Acids Res 42:D92–D97. https://doi.org/10.1093/nar/gkt1248
Macarak EJ, Wermuth PJ, Rosenbloom J, Uitto J (2021) Keloid disorder: fibroblast differentiation and gene expression profile in fibrotic skin diseases. Exp Dermatol 30:132–145. https://doi.org/10.1111/exd.14243
Shih B, Bayat A (2010) Genetics of keloid scarring. Arch Dermatol Res 302:319–339. https://doi.org/10.1007/s00403-009-1014-y
Sun XJ, Wang Q, Guo B, Liu XY, Wang B (2017) Identification of skin-related lncRNAs as potential biomarkers that involved in Wnt pathways in keloids. Oncotarget 8:34236–34244. https://doi.org/10.18632/oncotarget.15880
Si L, Zhang M, Guan E, Han Q, Liu Y, Long X, Long F, Zhao RC, Huang J, Liu Z, Zhao R, Zhang H, Wang X (2020) Resveratrol inhibits proliferation and promotes apoptosis of keloid fibroblasts by targeting HIF-1alpha. J Plast Surg Hand Surg 54:290–296. https://doi.org/10.1080/2000656X.2020.1771719
Shi J, Yao S, Chen P, Yang Y, Qian M, Han Y, Wang N, Zhao Y, He Y, Lyu L, Lu D (2020) The integrative regulatory network of circRNA and microRNA in keloid scarring. Mol Biol Rep 47:201–209. https://doi.org/10.1007/s11033-019-05120-y
Zhu HY, Bai WD, Li C, Zheng Z, Guan H, Liu JQ, Yang XK, Han SC, Gao JX, Wang HT, Hu DH (2016) Knockdown of lncRNA-ATB suppresses autocrine secretion of TGF-beta2 by targeting ZNF217 via miR-200c in keloid fibroblasts. Sci Rep 6:24728. https://doi.org/10.1038/srep24728
Pang Q, Wang Y, Xu M, Xu J, Xu S, Shen Y, Xu J, Lei R (2019) MicroRNA-152-5p inhibits proliferation and migration and promotes apoptosis by regulating expression of Smad3 in human keloid fibroblasts. BMB Rep 52:202–207
Liu Y, Yang D, Xiao Z, Zhang M (2012) miRNA expression profiles in keloid tissue and corresponding normal skin tissue. Aesthetic Plast Surg 36:193–201. https://doi.org/10.1007/s00266-011-9773-1
Kashiyama K, Mitsutake N, Matsuse M, Ogi T, Saenko VA, Ujifuku K, Utani A, Hirano A, Yamashita S (2012) miR-196a downregulation increases the expression of type I and III collagens in keloid fibroblasts. J Investig Dermatol 132:1597–1604. https://doi.org/10.1038/jid.2012.22
Duan X, Wu Y, Zhang Z, Lu Z (2020) Identification and analysis of dysregulated lncRNA and associated ceRNA in the pathogenesis of keloid. Ann Transl Med 8:222. https://doi.org/10.21037/atm.2020.01.07
Cheng N, Wu J, Yin M, Xu J, Wang Y, Chen X, Nie Z, Yin J (2019) LncRNA CASC11 promotes cancer cell proliferation in hepatocellular carcinoma by inhibiting miRNA-188-5p. Biosci Rep. https://doi.org/10.1042/BSR20190251
Yan S, Yue Y, Wang J, Li W, Sun M, Gu C, Zeng L (2019) LINC00668 promotes tumorigenesis and progression through sponging miR-188-5p and regulating USP47 in colorectal cancer. Eur J Pharmacol 858:172464. https://doi.org/10.1016/j.ejphar.2019.172464
Kaptan E, Sancar Bas S, Sancakli A, Aktas HG, Bayrak BB, Yanardag R, Bolkent S (2017) Runt-related transcription factor 2 (Runx2) is responsible for galectin-3 overexpression in human thyroid carcinoma. J Cell Biochem 118:3911–3919. https://doi.org/10.1002/jcb.26043
Hsu CK, Lin HH, Harn HI, Ogawa R, Wang YK, Ho YT, Chen WR, Lee YC, Lee JY, Shieh SJ, Cheng CM, McGrath JA, Tang MJ (2018) Caveolin-1 controls hyperresponsiveness to mechanical stimuli and fibrogenesis-associated RUNX2 activation in keloid fibroblasts. J Investig Dermatol 138:208–218. https://doi.org/10.1016/j.jid.2017.05.041
Lv W, Wu M, Ren Y, Luo X, Hu W, Zhang Q, Wu Y (2021) Treatment of keloids through Runx2 siRNAinduced inhibition of the PI3K/AKT signaling pathway. Mol Med Rep. https://doi.org/10.3892/mmr.2020.11693
Acknowledgements
We would like to give our sincere gratitude to the reviewers for their constructive comments and the support of Hainan Natural Science Foundation for this project.
Funding
This work was supported by Hainan Provincial Natural Science Foundation of China (822MS173).
Author information
Authors and Affiliations
Contributions
YL: conceptualization; funding acquisition; LL: writing—original draft; JYW: data curation; resources; FG: methodology; XL: formal analysis; SSL: investigation; software; visualization; ZYQ: project administration; supervision; ZHL: validation; writing—review AND editing. All authors have read and approved the final version of this manuscript to be published.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical approval
All patients and their families had informed consent. These experiments in our research were countenanced by the Ethics Committee of Hainan General Hospital, Hainan Affiliated Hospital of Hainan Medical University.
Consent to participate
All patients and their families had informed consent.
Consent for publication
All patients and their families had informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Y., Li, L., Wang, JY. et al. LncRNA GNAS-AS1 knockdown inhibits keloid cells growth by mediating the miR-188-5p/RUNX2 axis. Mol Cell Biochem 478, 707–719 (2023). https://doi.org/10.1007/s11010-022-04538-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-022-04538-6