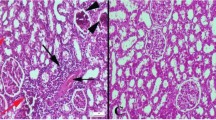
Abstract
Pancreatic inflammation and oxidative damage remain major concerns in type 1 diabetes mellitus (T1DM). Punicalagin, a major polyphenol in pomegranates, exhibited antioxidant and protective effects on several organs in case of T1DM; however, no study has yet explored the protective effects of punicalagin on the pancreas and islets of Langerhans. T1DM was induced by injecting 40 mg/kg streptozotocin (STZ) intraperitoneally. Punicalagin (1 mg/kg ip) was injected daily for 15 days after T1DM induction. In diabetic rats, punicalagin treatment lowered the levels of inflammatory biomarkers (monocyte chemoattractant protein-1 and C-reactive protein) and adhesion molecules (E-selectin, intercellular adhesion molecule, and vascular cell adhesion molecule) while activating myeloperoxidase activity. Treatment of diabetic rats with punicalagin improved glutathione content and superoxide dismutase, catalase, and glutathione peroxidase activities; upregulated serum paraoxonase-1 activity; and prevented the elevation lipid peroxidation and protein oxidation products in the pancreas. Furthermore, punicalagin protected the pancreas against STZ-induced histopathological alterations and increased immune-reactive β-cells while reducing leucocyte infiltration into the islets of Langerhans, leading to normalized blood glucose and insulin levels. These findings indicated that punicalagin might protect against the development of insulitis in T1DM. In conclusion, punicalagin exerts a strong protective effect on the pancreas against oxidative injury and inflammation in STZ-induced experimental T1DM. The present results recommend punicalagin as a potential adjuvant for reducing diabetes-associated insulitis.









Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Roder PV, Wu B, Liu Y, Han W (2016) Pancreatic regulation of glucose homeostasis. Exp Mol Med 48:e219. https://doi.org/10.1038/emm.2016.6
Newsholme P, Cruzat VF, Keane KN, Carlessi R, de Bittencourt PI (2016) Molecular mechanisms of ROS production and oxidative stress in diabetes. Biochem J 473:4527–4550. https://doi.org/10.1042/BCJ20160503C
Pugliese A (2016) Insulitis in the pathogenesis of type 1 diabetes. Pediatr Diabetes 17:31–36. https://doi.org/10.1111/pedi.12388
Fraga CG, Croft KD, Kennedy DO, Tomás-Barberán FA (2019) The effects of polyphenols and other bioactives on human health. Food Funct 10:514–528. https://doi.org/10.1039/c8fo01997e
Stiller A, Garrison K, Gurdyumov K, Kenner J, Yasmin F, Yates P et al (2021) From fighting critters to saving lives: polyphenols in plant defense and human health. Int J Mol Sci 22:8995. https://doi.org/10.3390/ijms22168995
Faddladdeen KAJ (2021) Ameliorating effect of pomegranate peel extract supplement against type 1 diabetes-induced hepatic changes in the rat: biochemical, morphological and ultrastructural microscopic studies. Folia Morphol (Warsz) 80:149–157. https://doi.org/10.5603/FM.a2020.0034
Subkorn P, Norkaew C, Deesrisak K, Tanyong D (2021) Punicalagin, a pomegranate compound, induces apoptosis and autophagy in acute leukemia. PeerJ 9:e12303. https://doi.org/10.7717/peerj.12303
Venusova E, Kolesarova A, Horky P, Slama P (2021) Physiological and Immune Functions of Punicalagin. Nutrients 13:2150. https://doi.org/10.3390/nu13072150
Taheri Rouhi SZ, Sarker MMR, Rahmat A, Alkahtani SA, Othman F (2017) The effect of pomegranate fresh juice versus pomegranate seed powder on metabolic indices, lipid profile, inflammatory biomarkers, and the histopathology of pancreatic islets of Langerhans in streptozotocin-nicotinamide induced type 2 diabetic Sprague-Dawley rats. BMC Complement Altern Med 17:156. https://doi.org/10.1186/s12906-017-1667-6
Venusova E, Kolesarova A, Horky P, Slama P (2021) Physiological and immune functions of punicalagin. Nutrients 13:2150. https://doi.org/10.3390/nu13072150
Yu LM, Dong X, Xue XD, Zhang J, Li Z, Wu HJ et al (2019) Protection of the myocardium against ischemia/reperfusion injury by punicalagin through an SIRT1-NRF-2-HO-1-dependent mechanism. Chem Biol Interact 306:152–162. https://doi.org/10.1016/j.cbi.2019.05.003
El-Missiry MA, Amer MA, Hemieda FA, Othman AI, Sakr DA, Abdulhadi HL (2015) Cardioameliorative effect of punicalagin against streptozotocin-induced apoptosis, redox imbalance, metabolic changes and inflammation. Egypt J Basic Appl Sci 2:247–260. https://doi.org/10.1016/j.ejbas.2015.09.004
An X, Zhang Y, Cao Y, Chen J, Qin H, Yang L (2020) Punicalagin protects diabetic nephropathy by inhibiting pyroptosis based on TXNIP/NLRP3 pathway. Nutrients 12:1516. https://doi.org/10.3390/nu12051516
Zhong J, Reece EA, Yang P (2015) Punicalagin exerts protective effect against high glucose-induced cellular stress and neural tube defects. Biochem Biophys Res Commun 467:179–184. https://doi.org/10.1016/j.bbrc.2015.10.024
Jin D, Zhang B, Li Q, Tu J, Zhou B (2020) Effect of punicalagin on multiple targets in streptozotocin/high-fat diet-induced diabetic mice. Food Funct 11:10617–10634. https://doi.org/10.1039/d0fo01275k
El-Missiry MA, ElKomy MA, Othman AI, AbouEl-Ezz AM (2018) Punicalagin ameliorates the elevation of plasma homocysteine, amyloid-beta, TNF-alpha and apoptosis by advocating antioxidants and modulating apoptotic mediator proteins in brain. Biomed Pharmacother 102:472–480. https://doi.org/10.1016/j.biopha.2018.03.096
Furman BL (2015) Streptozotocin-induced diabetic models in mice and rats. Curr Protoc Pharmacol. https://doi.org/10.1002/0471141755.ph0547s70
Othman AI, El-Sawi MR, El-Missiry MA, Abukhalil MH (2017) Epigallocatechin-3-gallate protects against diabetic cardiomyopathy through modulating the cardiometabolic risk factors, oxidative stress, inflammation, cell death and fibrosis in streptozotocin-nicotinamide-induced diabetic rats. Biomed Pharmacother 94:362–373. https://doi.org/10.1016/j.biopha.2017.07.129
Janardhan KS, Jensen H, Clayton NP, Herbert RA (2018) Immunohistochemistry in investigative and toxicologic pathology. Toxicol Pathol 46:488–510. https://doi.org/10.1177/0192623318776907
Anggorowati N, Kurniasari CR, Damayanti K, Cahyanti T, Widodo I, Ghozali A et al (2017) Histochemical and immunohistochemical study of α-SMA, collagen, and PCNA in epithelial ovarian neoplasm. Asian Pac J Cancer Prev: APJCP 18:667. https://doi.org/10.22034/APJCP.2017.18.3.667
Zheng P, Li Z, Zhou Z (2018) Gut microbiome in type 1 diabetes: a comprehensive review. Diabetes Metab Res Rev 34:e3043. https://doi.org/10.1002/dmrr.3043
Lindsay RS, Corbin K, Mahne A, Levitt BE, Gebert MJ, Wigton EJ et al (2015) Antigen recognition in the islets changes with progression of autoimmune islet infiltration. J Immunol 194:522–530. https://doi.org/10.4049/jimmunol.1400626
Rhee KJ, Lee CG, Kim SW, Gim DH, Kim HC, Jung BD (2015) Extract of Ginkgo Biloba Ameliorates streptozotocin-induced type 1 diabetes mellitus and high-fat diet-induced type 2 diabetes mellitus in mice. Int J Med Sci 12:987–994. https://doi.org/10.7150/ijms.13339
Jialal I, Kaur H, Devaraj S (2013) Human C-reactive protein accentuates macrophage activity in biobreeding diabetic rats. J Diabetes Complications 27:23–28. https://doi.org/10.1016/j.jdiacomp.2012.03.020
Cvetkovic I, Al-Abed Y, Miljkovic D, Maksimovic-Ivanic D, Roth J, Bacher M et al (2005) Critical role of macrophage migration inhibitory factor activity in experimental autoimmune diabetes. Endocrinol 146:2942–2951. https://doi.org/10.1210/en.2004-1393
Cao Y, Chen J, Ren G, Zhang Y, Tan X, Yang L (2019) Punicalagin prevents inflammation in LPS-induced RAW264.7 macrophages by inhibiting FoxO3a/autophagy signaling pathway. Nutrients 11:2794. https://doi.org/10.3390/nu11112794
BenSaad LA, Kim KH, Quah CC, Kim WR, Shahimi M (2017) Anti-inflammatory potential of Ellagic acid, Gallic acid and punicalagin A&B isolated from Punica granatum. BMC Complement Altern Med 17:47. https://doi.org/10.1186/s12906-017-1555-0
Nguyen-Ngo C, Willcox JC, Lappas M (2020) Anti-inflammatory effects of phenolic acids punicalagin and curcumin in human placenta and adipose tissue. Placenta 100:1–12. https://doi.org/10.1016/j.placenta.2020.08.002
Liu L, Liu Y, Qi B, Wu Q, Li Y, Wang Z (2014) Nicorandil attenuates endothelial VCAM-1 expression via thioredoxin production in diabetic rats induced by streptozotocin. Mol Med Rep 9:2227–2232. https://doi.org/10.3892/mmr.2014.2066
Gustavsson C, Agardh C-D, Zetterqvist AV, Nilsson J, Agardh E, Gomez MF (2010) Vascular cellular adhesion molecule-1 (VCAM-1) expression in mice retinal vessels is affected by both hyperglycemia and hyperlipidemia. PLoS ONE 5:e12699. https://doi.org/10.1371/journal.pone.0012699
Asni E, Romus I (2019) The effect of alpha-lipoic acid on expression of VCAM-1 in type 2 diabetic rat. Anat Cell Biol 52:176–182. https://doi.org/10.5115/acb.2019.52.2.176
Asgary S, Sahebkar A, Afshani MR, Keshvari M, Haghjooyjavanmard S, Rafieian-Kopaei M (2014) Clinical evaluation of blood pressure lowering, endothelial function improving, hypolipidemic and anti-inflammatory effects of pomegranate juice in hypertensive subjects. Phytother Res 28:193–199. https://doi.org/10.1002/ptr.4977
Sohrab G, Nasrollahzadeh J, Zand H, Amiri Z, Tohidi M, Kimiagar M (2014) Effects of pomegranate juice consumption on inflammatory markers in patients with type 2 diabetes: a randomized, placebo-controlled trial. J Res Med Sci 19:215–220
Sohrab G, Nasrollahzadeh J, Tohidi M, Zand H, Nikpayam O (2018) Pomegranate juice increases sirtuin1 protein in peripheral blood mononuclear cell from patients with type 2 diabetes: a randomized placebo controlled clinical trial. Metab Syndr Relat Disord 16:446–451. https://doi.org/10.1089/met.2017.0146
Habtemariam S (2019) Medicinal foods as potential therapies for type-2 Diabetes and associated diseases: the chemical and pharmacological basis of their action, 1st edn. Academic Press
Bradley PP, Priebat DA, Christensen RD, Rothstein G (1982) Measurement of cutaneous inflammation: estimation of neutrophil content with an enzyme marker. J Invest Dermatol 78:206–209. https://doi.org/10.1111/1523-1747.ep12506462
Vanessa Fiorentino T, Prioletta A, Zuo P, Folli F (2013) Hyperglycemia-induced oxidative stress and its role in diabetes mellitus related cardiovascular diseases. Curr Pharm Des 19:5695–5703. https://doi.org/10.2174/1381612811319320005
Mabhida SE, Johnson R, Ndlovu M, Sangweni NF, Louw J, Opoku A et al (2018) A Lanosteryl triterpene from Protorhus longifolia augments insulin signaling in type 1 diabetic rats. BMC Complement Altern Med 18:265. https://doi.org/10.1186/s12906-018-2337-z
Kulkarni R, Acharya J, Ghaskadbi S, Goel P (2014) Oxidative stress as a covariate of recovery in diabetes therapy. Front Endocrinol (Lausanne) 5:89. https://doi.org/10.3389/fendo.2014.00089
Tang D, Liu L, Ajiakber D, Ye J, Xu J, Xin X et al (2018) Anti-diabetic effect of Punica granatum flower polyphenols extract in type 2 diabetic rats: activation of Akt/GSK-3beta and inhibition of IRE1alpha-XBP1 pathways. Front Endocrinol (Lausanne) 9:586. https://doi.org/10.3389/fendo.2018.00586
Davies MJ, Hawkins CL (2020) The role of myeloperoxidase in biomolecule modification, chronic inflammation, and disease. Antioxid Redox Signal 32:957–981. https://doi.org/10.1089/ars.2020.8030
Nishat S, Klinke A, Baldus S, Khan LA, Basir SF (2014) Increased A3AR-dependent vasoconstriction in diabetic mice is promoted by myeloperoxidase. J Cardiovasc Pharmacol 64:465–472. https://doi.org/10.1097/FJC.0000000000000139
Ojha S, Alkaabi J, Amir N, Sheikh A, Agil A, Fahim MA et al (2014) Withania coagulans fruit extract reduces oxidative stress and inflammation in kidneys of streptozotocin-induced diabetic rats. Oxid Med Cell Longev 2014:201436. https://doi.org/10.1155/2014/201436
Stocker P, Cassien M, Vidal N, Thétiot-Laurent S, Pietri S (2017) A fluorescent homogeneous assay for myeloperoxidase measurement in biological samples. A positive correlation between myeloperoxidase-generated HOCl level and oxidative status in STZ-diabetic rats. Talanta 170:119–127. https://doi.org/10.1016/j.talanta.2017.03.102
Ndrepepa G (2019) Myeloperoxidase—a bridge linking inflammation and oxidative stress with cardiovascular disease. Clin Chim Acta 493:36–51. https://doi.org/10.1016/j.cca.2019.02.022
Kuchta A, Strzelecki A, Cwiklinska A, Toton M, Gruchala M, Zdrojewski Z et al (2015) PON-1 activity and plasma 8-isoprostane concentration in patients with angiographically proven coronary artery disease. Oxid Med Cell Longev 2015:5136937. https://doi.org/10.1155/2016/5136937
Tas S, Sarandol E, Ziyanok S, Aslan K, Dirican M (2005) Effects of green tea on serum paraoxonase/arylesterase activities in streptozotocin-induced diabetic rats. Nutr Res 25:1061–1074. https://doi.org/10.1016/j.nutres.2005.10.001
Taş S, Sarandöl E, Dirican M (2014) Vitamin B6 supplementation improves oxidative stress and enhances serum paraoxonase/arylesterase activities in streptozotocin-induced diabetic rats. Sci World J 2014:351598. https://doi.org/10.1155/2014/351598
Hedrick CC, Thorpe SR, Fu MX, Harper CM, Yoo J, Kim SM et al (2000) Glycation impairs high-density lipoprotein function. Diabetologia 43:312–320. https://doi.org/10.1007/s001250050049
Rosenblat M, Hayek T, Aviram M (2006) Anti-oxidative effects of pomegranate juice (PJ) consumption by diabetic patients on serum and on macrophages. Atherosclerosis 187:363–371. https://doi.org/10.1016/j.atherosclerosis.2005.09.006
Betanzos-Cabrera G, Guerrero-Solano A, Cruz-Mayorga D, Calderon Z, Villanueva-Sanchez J, Irecta AP et al (2009) Effect of pomegranate juice on paraoxonase 1 (pon1) gene expression and enzymatic activity in a streptozotocin-induced diabetes and diet-induced obese mode. Can J Diabetes 33:266–267. https://doi.org/10.1016/S1499-2671(09)33210-4
Betanzos-Cabrera G, Guerrero-Solano J, Martínez-Pérez M, Calderón-Ramos Z, Belefant-Miller H, Cancino-Diaz JC (2011) Pomegranate juice increases levels of paraoxonase1 (PON1) expression and enzymatic activity in streptozotocin-induced diabetic mice fed with a high-fat diet. Food Res Int 44:1381–1385. https://doi.org/10.1016/j.foodres.2011.01.042
BinMowyna MN, Binobead MA, Al Badr NA, AlSedairy SA, Elredh IAR, Al-Qahtani WS (2019) Effect of Saudi and Egyptian pomegranate polyphenols in regulating the activity of PON1, PON2 and lipid profile for preventing coronary heart disease. BioRxiv 2019:570838. https://doi.org/10.1101/570838
Rojas J, Bermudez V, Palmar J, Martinez MS, Olivar LC, Nava M et al (2018) Pancreatic beta cell death: novel potential mechanisms in diabetes therapy. J Diabetes Res 2018:9601801. https://doi.org/10.1155/2018/9601801
Willcox A, Richardson SJ, Bone AJ, Foulis AK, Morgan NG (2009) Analysis of islet inflammation in human type 1 diabetes. Clin Exp Immunol 155:173–181. https://doi.org/10.1111/j.1365-2249.2008.03860.x
Zhao B, Wu F, Han X, Zhou W, Shi Q, Wang H (2020) Protective effects of acarbose against insulitis in multiple low-dose streptozotocin-induced diabetic mice. Life Sci 263:118490. https://doi.org/10.1016/j.lfs.2020.118490
Shalaby MF, Zaki AA, Shabana S, Osman NM (2015) Effects of Punica granatum peels extract on the Intestinal α-glucosidase activity and the histopathology of the pancreas of Alloxan–induced diabetic rats. Egypt J Med Sci 36:255–272
Wang JY, Zhu C, Qian TW, Guo H, Wang DD, Zhang F et al (2015) Extracts of black bean peel and pomegranate peel ameliorate oxidative stress-induced hyperglycemia in mice. Exp Ther Med 9:43–48. https://doi.org/10.3892/etm.2014.2040
Acknowledgements
Facilities provided by Mansoura University are greatly acknowledged.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by HLA, BRD, LHA, AIO, MAEM, and MEA. The first draft of the manuscript was written and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interests
None. The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in this study were conducted in accordance with the regulations approved by the Ethics Committee at Faculty of Science, Mansoura University, Egypt.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Abdulhadi, H.L., Dabdoub, B.R., Ali, L.H. et al. Punicalagin protects against the development of pancreatic injury and insulitis in rats with induced T1DM by reducing inflammation and oxidative stress. Mol Cell Biochem 477, 2817–2828 (2022). https://doi.org/10.1007/s11010-022-04478-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-022-04478-1