Abstract
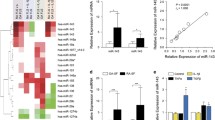
Aberrant proliferation and migration of fibroblast-like synoviocytes (FLS) are major characteristics of rheumatoid arthritis (RA). MicroRNA-133 (miR-133) is a tumor-suppressive miRNA that targets various genes responsive for cell proliferation and migration. The aim of this study was to examine the effect of miR-133 on RA FLS. A high throughput miRNA microarray was performed in synovium from mice with collagen-induced arthritis (CIA). Expression levels of miR-133 and the putative targets were determined in synovium and FLS from patients with RA and mice with CIA. Overexpression of miR-133 in RA FLS was performed by lentiviral vector-mediated transfer of precursor miRNA (pre-miR). The expression of miR-133a/b was decreased in the joint tissue and FLS of CIA mice, as determined by miRNA array and qRT-PCR. Down-regulation of miR-133a/b expression could also be observed in synovium and FLS from patients with RA. Overexpression of miR-133 reduced cell viability and migration of RA FLS, with decreased levels of FSCN1, EGFR, and MET. Our findings demonstrated the inhibitory effects of miR-133 on FLS viability and migration, and might contribute to the pharmacologic development of miR-133 therapeutics in patients with RA.





Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- RA:
-
Rheumatoid arthritis
- CIA:
-
Collagen-induced arthritis
- FLS:
-
Fibroblast-like synoviocytes
- MiRNA:
-
MicroRNA
- MicroRNA-133:
-
MiR-133
- Precursor miRNA:
-
Pre-miR
- EMT:
-
Epithelial-mesenchymal transition
- UTR:
-
Untranslated region
- ESCC:
-
Esophageal squamous cell carcinoma
- GEO:
-
Gene Expression Omnibus
- CIA FLS:
-
FLS from mice with CIA
- RA FLS:
-
FLS from patients with RA
- N FLS:
-
FLS from naïve control mice
- H FLS:
-
FLS from healthy human FLS
- TNF:
-
Tumor necrosis factor
- MOI:
-
Multiplicity of infection
References
Bustamante MF, Garcia-Carbonell R, Whisenant KD, Guma M (2017) Fibroblast-like synoviocyte metabolism in the pathogenesis of rheumatoid arthritis. Arthritis Res Ther 19:110. https://doi.org/10.1186/s13075-017-1303-3
Lafyatis R, Remmers EF, Roberts AB, Yocum DE, Sporn MB, Wilder RL (1989) Anchorage-independent growth of synoviocytes from arthritic and normal joints. Stimulation by exogenous platelet-derived growth factor and inhibition by transforming growth factor-beta and retinoids. J Clin Investig 83:1267–1276
Müller-Ladner U, Kriegsmann J, Franklin BN, Matsumoto S, Geiler T, Gay RE, Gay S (1996) Synovial fibroblasts of patients with rheumatoid arthritis attach to and invade normal human cartilage when engrafted into SCID mice. Am J Pathol 149:1607
Chen SY (2014) Does epithelial-mesenchymal transition happen in rheumatoid joints? Eur J Rheumatol 1:86
Chen SY, Shiau AL, Li YT, Lin CC, Jou IM, Liu MF, Wu CL, Wang CR (2015) Transcription factor snail regulates tumor necrosis factor alpha-mediated synovial fibroblast activation in the rheumatoid joint. Arthritis Rheumatol 67:39–50. https://doi.org/10.1002/art.38899
Lauzier A, Lavoie RR, Charbonneau M, Gouin-Boisvert B, Harper K, Dubois CM (2016) Snail is a critical mediator of invadosome formation and joint degradation in arthritis. Am J Pathol 186:359–374. https://doi.org/10.1016/j.ajpath.2015.10.021
Shen P-C, Chang P-C, Hsieh J-L (2021) Snail regulation in fibroblast-like synoviocytes by a histone deacetylase or glycogen synthase kinase inhibitor affects cell proliferation and gene expression. PLoS ONE 16:e0257839
de Launay D, Vreijling J, Hartkamp LM, Karpus ON, Abreu JR, van Maanen MA, Sanders ME, Grabiec AM, Hamann J, Ørum H (2010) Silencing the expression of Ras family GTPase homologues decreases inflammation and joint destruction in experimental arthritis. Am J Pathol 177:3010–3024
Lee S-Y, Kwok S-K, Son H-J, Ryu J-G, Kim E-K, Oh H-J, Cho M-L, Ju JH, Park S-H, Kim H-Y (2013) IL-17-mediated Bcl-2 expression regulates survival of fibroblast-like synoviocytes in rheumatoid arthritis through STAT3 activation. Arthritis Res Ther 15:R31
Matsumoto S, Müller-Ladner U, Gay R, Nishioka K, Gay S (1996) Ultrastructural demonstration of apoptosis, Fas and Bcl-2 expression of rheumatoid synovial fibroblasts. J Rheumatol 23:1345–1352
Zhang HG, Wang Y, Xie JF, Liang X, Liu D, Yang P, Hsu HC, Ray R, Mountz JD (2001) Regulation of tumor necrosis factor α–mediated apoptosis of rheumatoid arthritis synovial fibroblasts by the protein kinase Akt. Arthritis Rheum 44:1555–1567
Cha HS, Rosengren S, Boyle DL, Firestein GS (2006) PUMA regulation and proapoptotic effects in fibroblast-like synoviocytes. Arthritis Rheum 54:587–592
You X, Boyle DL, Hammaker D, Firestein GS (2006) PUMA-mediated apoptosis in fibroblast-like synoviocytes does not require p53. Arthritis Res Ther 8:R157
Krol J, Loedige I, Filipowicz W (2010) The widespread regulation of microRNA biogenesis, function and decay. Nat Rev Genet 11:597–610
Peng JS, Chen SY, Wu CL, Chong HE, Ding YC, Shiau AL, Wang CR (2016) Amelioration of experimental autoimmune arthritis through targeting of synovial fibroblasts by intraarticular delivery of MicroRNAs 140–3p and 140–5p. Arthritis Rheumatol 68:370–381. https://doi.org/10.1002/art.39446
Chen J-F, Mandel EM, Thomson JM, Wu Q, Callis TE, Hammond SM, Conlon FL, Wang D-Z (2006) The role of microRNA-1 and microRNA-133 in skeletal muscle proliferation and differentiation. Nat Genet 38:228–233
Xu F, Li F, Zhang W, Jia P (2015) Growth of glioblastoma is inhibited by miR-133-mediated EGFR suppression. Tumour Biol 36:9553–9558. https://doi.org/10.1007/s13277-015-3724-4
Zhao H, Li M, Li L, Yang X, Lan G, Zhang Y (2013) MiR-133b is down-regulated in human osteosarcoma and inhibits osteosarcoma cells proliferation, migration and invasion, and promotes apoptosis. PLoS ONE 8:e83571. https://doi.org/10.1371/journal.pone.0083571
Kano M, Seki N, Kikkawa N, Fujimura L, Hoshino I, Akutsu Y, Chiyomaru T, Enokida H, Nakagawa M, Matsubara H (2010) miR-145, miR-133a and miR-133b: tumor-suppressive miRNAs target FSCN1 in esophageal squamous cell carcinoma. Int J Cancer 127:2804–2814. https://doi.org/10.1002/ijc.25284
Li J, Zhang S, Pei M, Wu L, Liu Y, Li H, Lu J, Li X (2018) FSCN1 promotes epithelial-mesenchymal transition through increasing snail1 in ovarian cancer cells. Cell Physiol Biochem 49:1766–1777
Li YT, Chen SY, Wang CR, Liu MF, Lin CC, Jou IM, Shiau AL, Wu CL (2012) Brief Report: Amelioration of collagen-induced arthritis in mice by lentivirus-mediated silencing of microRNA-223. Arthritis Rheum 64:3240–3245
Pohlers D, Beyer A, Koczan D, Wilhelm T, Thiesen HJ, Kinne RW (2007) Constitutive upregulation of the transforming growth factor-beta pathway in rheumatoid arthritis synovial fibroblasts. Arthritis Res Ther 9:R59. https://doi.org/10.1186/ar2217
Machesky LM, Li A (2010) Fascin: Invasive filopodia promoting metastasis. Commun Integr Biol 3:263–270
Yang S, Huang F-K, Huang J, Chen S, Jakoncic J, Leo-Macias A, Diaz-Avalos R, Chen L, Zhang JJ, Huang X-Y (2013) Molecular mechanism of fascin function in filopodial formation. J Biol Chem 288:274–284
Darnel AD, Behmoaram E, Vollmer RT, Corcos J, Bijian K, Sircar K, Su J, Jiao J, Alaoui-Jamali MA, Bismar TA (2009) Fascin regulates prostate cancer cell invasion and is associated with metastasis and biochemical failure in prostate cancer. Clin Cancer Res 15:1376–1383
Hashimoto Y, Ito T, Inoue H, Okumura T, Tanaka E, Tsunoda S, Higashiyama M, Watanabe G, Imamura M, Shimada Y (2005) Prognostic significance of fascin overexpression in human esophageal squamous cell carcinoma. Clin Cancer Res 11:2597–2605
Tan VY, Lewis SJ, Adams JC, Martin RM (2013) Association of fascin-1 with mortality, disease progression and metastasis in carcinomas: a systematic review and meta-analysis. BMC Med 11:52
Ma K, Zhang C, Li W (2021) Fascin1 mediated release of pro-inflammatory cytokines and invasion/migration in rheumatoid arthritis via the STAT3 pathway. Cell Cycle 20:2210–2220
Chen SY, Shiau AL, Shieh GS, Su CH, Lee CH, Lee HL, Wang CR, Wu CL (2009) Amelioration of experimental arthritis by a telomerase-dependent conditionally replicating adenovirus that targets synovial fibroblasts. Arthritis Rheum 60:3290–3302. https://doi.org/10.1002/art.24940
Zhu S, Pan W, Song X, Liu Y, Shao X, Tang Y, Liang D, He D, Wang H, Liu W, Shi Y, Harley JB, Shen N, Qian Y (2012) The microRNA miR-23b suppresses IL-17-associated autoimmune inflammation by targeting TAB2, TAB3 and IKK-alpha. Nat Med 18:1077–1086. https://doi.org/10.1038/nm.2815
Beg MS, Brenner AJ, Sachdev J, Borad M, Kang YK, Stoudemire J, Smith S, Bader AG, Kim S, Hong DS (2017) Phase I study of MRX34, a liposomal miR-34a mimic, administered twice weekly in patients with advanced solid tumors. Invest New Drugs 35:180–188. https://doi.org/10.1007/s10637-016-0407-y
Zhang W, Wu Y, Shiozaki Y, Sugimoto Y, Takigawa T, Tanaka M, Matsukawa A, Ozaki T (2018) miRNA-133a-5p inhibits the expression of osteoblast differentiation-associated markers by targeting the 3’ UTR of RUNX2. DNA Cell Biol 37:199–209. https://doi.org/10.1089/dna.2017.3936
Acknowledgements
We thank Dr. D. Trono (Ecole Polytechnique Federale de Lausanne, Lausanne, Switzerland) for generously providing psPAX2 and pMD2.G plasmids for the production of lentiviral vectors and the Laboratory Animal Center, College of Medicine, National Cheng Kung University and Taiwan Animal Consortium (AAALAC International Full Accreditation) for the animal care. We also thank professor Chi-Chien Lin helps us collect synovial tissue samples at Taichung Veterans General Hospital, Taichung, Taiwan.
Funding
This work was supported by MOST 108-2314-B-006-037-MY3, and 108-2314-B-273-005-MY3 from the Ministry of Science and Technology, Taiwan.
Author information
Authors and Affiliations
Contributions
All authors were involved in drafting the article or revising it critically for important intellectual content, and approved the final version to be published. Drs. CLWu and Chen have full access to all of the data in the study, and take responsibility for the integrity of data and the accuracy of data analysis. Conceptualization: SYC, JLH, PTW, CLW. Data curation: SYC, JLH, PTW, ALS, CLW. Formal Analysis: SYC, JLH, PTW, ALS, CLW. Funding acquisition: SYC, JLH. Investigation: SYC, JLH, PTW, CLW. Methodology: SYC, JLH, PTW, ALS, CLW. Project administration: SYC, JLH. Resources: SYC, JLH, PTW, CLW. Supervision: SYC, JLH, PTW, CLW. Validation: SYC, JLH, PTW, CLW. Visualization: SYC, JLH, CLW. Writing-original draft: SYC, JLH, CLW. Writing-review and editing: SYC, JLH, PTW, ALS, CLW.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, SY., Hsieh, JL., Wu, PT. et al. MicroRNA-133 suppresses cell viability and migration of rheumatoid arthritis fibroblast-like synoviocytes by down-regulation of MET, EGFR, and FSCN1 expression. Mol Cell Biochem 477, 2529–2537 (2022). https://doi.org/10.1007/s11010-022-04457-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-022-04457-6