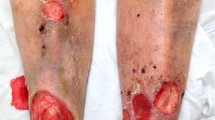
Abstract
Chronic wounds are a substantial clinical problem in diabetes and nearly 6% of diabetics suffer from foot disease including ulceration, infection, and tissue necrosis. Wound healing in diabetes is impaired and delayed and is augmented by diabetic complications. Wound healing involves complex cellular, molecular, and biochemical processes and animal models are the most suitable prototype to investigate and understand the underlying pathological changes in the process of wound healing. Animal models are also useful in evaluating the safety and efficacy of newer therapeutic agents and improving the clinical approaches for human patients with chronic ulcers. The wound healing strategies get more complicated in the presence of diabetes and its associated complication. Despite the advancement in methods of wound healing, the healing of the chronic diabetic foot ulcer (DFU) remains an important clinical problem resulting in costly and prolonged treatment and poses a risk for major amputation. Saying that it is important to elucidate the newer therapeutic targets and strategies via an in-depth understanding of the complicated cascade of the chronic DFU. A major challenge in translating lab findings to clinics is the lack of an optimal preclinical model capable of properly recapitulating human wounds. Both small and large animal models of wound healing involving rodents, rabbits, and pigs have been discussed. Mouse and rats as small animal models and pig as large animal models have been discussed in association with the diabetic wound but there are advantages and limitations for each model. In this review, we critically reviewed the pros and cons of experimental models of diabetic wound healing with a focus on type II diabetes rodent models.

Similar content being viewed by others
Data availability
Not applicable.
References
Sen CK (2019) Human wounds and its burden: an updated compendium of estimates. Adv Wound Care (New Rochelle). 8(2):39–48. https://doi.org/10.1089/wound.2019.0946
Raghav A, Khan ZA, Labala RK, Ahmad J, Noor S, Mishra BK (2018) Financial burden of diabetic foot ulcers to world: a progressive topic to discuss always. Ther Adv Endocrinol Metab. 9(1):29–31. https://doi.org/10.1177/2042018817744513
Frykberg RG, Banks J (2015) Challenges in the treatment of chronic wounds. Adv Wound Care (New Rochelle). 4(9):560–82. https://doi.org/10.1089/wound.2015.0635
Elliot S, Wikramanayake TC, Jozic I, Tomic-Canic M (2018) A modeling conundrum: murine models for cutaneous wound healing. J Investig Dermatol 138(4):736–740
Grada A, Mervis J, Falanga V (2018) Research techniques made simple: animal models of wound healing. J Investig Dermatol 138(10):2095-2105.e1
Mustoe TA, O’shaughnessy K, Kloeters O (2006) Chronic wound pathogenesis and current treatment strategies: a unifying hypothesis. Plast Reconstr Surg 117(7S):35S-41S
Mirzaei M, Rahmaninan M, Mirzaei M, Nadjarzadeh A, Dehghani Tafti AA (2020) Epidemiology of diabetes mellitus, pre-diabetes, undiagnosed and uncontrolled diabetes in Central Iran: results from Yazd health study. BMC Public Health 20(1):166. https://doi.org/10.1186/s12889-020-8267-y
Mekala KC, Bertoni AG (2020) Epidemiology of diabetes mellitus. Transplantation, bioengineering, and regeneration of the endocrine pancreas. Elsevier, New York, pp 49–58
Deshpande AD, Harris-Hayes M, Schootman M (2008) (2008) Epidemiology of diabetes and diabetes-related complications. Phys Ther 88(11):1254–64. https://doi.org/10.2522/ptj.20080020
Oliver TI, Mutluoglu M. (2019) Diabetic foot ulcer
Shofler D, Rai V, Mansager S, Cramer K, Agrawal DK (2021) Impact of resolving mediators in the immunopathology of diabetes and wound healing. Expert Rev Clin Immunol 17(6):681–90. https://doi.org/10.1080/1744666X.2021.1912598
Guo S, Dipietro LA (2010) Factors affecting wound healing. J Dent Res 89(3):219–29. https://doi.org/10.1177/0022034509359125
Suckow MA, Gobbett TA (2017) Peterson RG (2017) Wound healing delay in the ZDSD rat. In Vivo 31(1):55–60. https://doi.org/10.21873/invivo.11025
Chen L, Mirza R, Kwon Y, DiPietro LA, Koh TJ (2015) The murine excisional wound model: contraction revisited. Wound Repair Regen 23(6):874–877. https://doi.org/10.1111/wrr.12338
Doeing DC, Borowicz JL, Crockett ET (2003) Gender dimorphism in differential peripheral blood leukocyte counts in mice using cardiac, tail, foot, and saphenous vein puncture methods. BMC Clin Pathol 3(1):3. https://doi.org/10.1186/1472-6890-3-3
Mestas J, Hughes CC (2004) Of mice and not men: differences between mouse and human immunology. J Immunol 172(5):2731–8. https://doi.org/10.4049/jimmunol.172.5.2731
Tao L, Reese TA (2017) Making mouse models that reflect human immune responses. Trends Immunol 38(3):181–93. https://doi.org/10.1016/j.it.2016.12.007
Zschaler J, Schlorke D, Arnhold J (2014) Differences in innate immune response between man and mouse. Crit Rev Immunol 34(5):433–454
Seaton M, Hocking A, Gibran NS (2015) Porcine models of cutaneous wound healing. ILAR J 56(1):127–38. https://doi.org/10.1093/ilar/ilv016
Pastar I, Liang L, Sawaya AP, Wikramanayake TC, Glinos GD, Drakulich S et al (2018) Preclinical models for wound-healing studies. In: Marques A, Reis R, Pirraco R, Cerqueria M (eds) Skin tissue models. Elsevier, New York, pp 223–53. https://doi.org/10.1016/B978-0-12-810545-0.00010-3
Masson-Meyers DS, Andrade TA, Caetano GF, Guimaraes FR, Leite MN, Leite SN et al (2020) Experimental models and methods for cutaneous wound healing assessment. Int J Exp Pathol 101(1–2):21–37. https://doi.org/10.1111/iep.12346
Rodier PM (1976) (1976) Critical periods for behavioral anomalies in mice. Environ Health Perspect 18:79–83. https://doi.org/10.1289/ehp.761879
Trøstrup H, Thomsen K, Calum H, Høiby N, Moser C. (2016) Animal models of chronic wound care: the application of biofilms in clinical research. Chronic Wound Care Manage Res. 3:123–32. http://creativecommons.org/licenses/by-nc/3.0/
Wong VW, Sorkin M, Glotzbach JP, Longaker MT, Gurtner GC (2011) Surgical approaches to create murine models of human wound healing. J Biomed Biotechnol. https://doi.org/10.1155/2011/969618
Sami DG, Heiba HH, Abdellatif A (2019) Wound healing models: a systematic review of animal and non-animal models. Wound Med 24(1):8–17. https://doi.org/10.1016/j.wndm.2018.12.001
Dorsett-Martin WA (2004) Rat models of skin wound healing: a review. Wound Repair Regen 12(6):591–599. https://doi.org/10.1111/j.1067-1927.2004.12601.x
Kim DJ, Mustoe T, Clark RA (2015) Cutaneous wound healing in aging small mammals: a systematic review. Wound Repair Regen. 23(3):318–39. https://doi.org/10.1111/wrr.12290
Slavkovsky R, Kohlerova R, Tkacova V, Jiroutova A, Tahmazoglu B, Velebny V et al (2011) Zucker diabetic fatty rat: a new model of impaired cutaneous wound repair with type II diabetes mellitus and obesity. Wound Repair Regen 19(4):515–525. https://doi.org/10.1111/j.1524-475X.2011.00703.x
Nascimento AP, Costa AM (2006) Overweight induced by high-fat diet delays rat cutaneous wound healing. Br J Nutr 96(6):1069–1077. https://doi.org/10.1017/bjn20061955
Huynh P, Phie J, Krishna SM, Golledge J (2020) Systematic review and meta-analysis of mouse models of diabetes-associated ulcers. BMJ Open Diabetes Res Care. https://doi.org/10.1136/bmjdrc-2019-000982
King AJ (2012) (2012) The use of animal models in diabetes research. Br J Pharmacol 166(3):877–894. https://doi.org/10.1111/j.1476-5381.2012.01911.x
Fang RC, Kryger ZB, Buck DW II, De La Garza M, Galiano RD, Mustoe TA (2010) Limitations of the db/db mouse in translational wound healing research: Is the NONcNZO10 polygenic mouse model superior? Wound Repair Regen 18(6):605–13. https://doi.org/10.1111/j.1524-475X.2010.00634.x
Reinwald S, Peterson RG, Allen MR, Burr DB (2009) Skeletal changes associated with the onset of type 2 diabetes in the ZDF and ZDSD rodent models. Am J Physiol Endocrinol Metab 296(4):E765–E774. https://doi.org/10.1152/ajpendo.90937.2008
Velander P, Theopold C, Hirsch T, Bleiziffer O, Zuhaili B, Fossum M et al (2008) Impaired wound healing in an acute diabetic pig model and the effects of local hyperglycemia. Wound Repair Regen 16(2):288–93. https://doi.org/10.1111/j.1524-475X.2008.00367.x
Wang B, Charukeshi Chandrasekera P, Pippin J, J. (2014) Leptin-and leptin receptor-deficient rodent models: relevance for human type 2 diabetes. Curr Diabetes Rev 10(2):131–45. https://doi.org/10.2174/1573399810666140508121012
Surwit RS, Kuhn CM, Cochrane C, McCubbin JA, Feinglos MN (1988) Diet-induced type II diabetes in C57BL/6J mice. Diabetes 37(9):1163–7. https://doi.org/10.2337/diab.37.9.1163
Cheng KY, Lin ZH, Cheng YP, Chiu HY, Yeh NL, Wu TK et al (2018) Wound healing in streptozotocin-induced diabetic rats using atmospheric-pressure argon plasma jet. Sci Rep 8(1):12214. https://doi.org/10.1038/s41598-018-30597-1
Yu CO-L, Leung K-S, Fung K-P, Lam FF-Y, Ng ES-K, Lau K-M et al (2017) The characterization of a full-thickness excision open foot wound model in n5-streptozotocin (STZ)-induced type 2 diabetic rats that mimics diabetic foot ulcer in terms of reduced blood circulation, higher C-reactive protein, elevated inflammation, and reduced cell proliferation. Exp Anim 66(3):259–69. https://doi.org/10.1538/expanim.17-0016
Zhang M, Lv X-Y, Li J, Xu Z-G, Chen L (2008) The characterization of high-fat diet and multiple low-dose streptozotocin induced type 2 diabetes rat model. Exp Diabetes Res. https://doi.org/10.1155/2008/704045
Liu Z, Li W, Li X, Zhang M, Chen L, Zheng Y-N et al (2013) Antidiabetic effects of malonyl ginsenosides from Panax ginseng on type 2 diabetic rats induced by high-fat diet and streptozotocin. J Ethnopharmacol 145(1):233–40. https://doi.org/10.1016/j.jep.2012.10.058
Qian C, Zhu C, Yu W, Jiang X, Zhang F (2015) High-fat diet/low-dose streptozotocin-induced type 2 diabetes in rats impacts osteogenesis and Wnt signaling in bone marrow stromal cells. PLoS ONE. https://doi.org/10.1371/journal.pone.0136390
Premilovac D, Gasperini RJ, Sawyer S, West A, Keske MA, Taylor BV et al (2017) A new method for targeted and sustained induction of type 2 diabetes in rodents. Sci Rep 7(1):14158. https://doi.org/10.1038/s41598-017-14114-4
Barrière DA, Noll C, Roussy G, Lizotte F, Kessai A, Kirby K et al (2018) Combination of high-fat/high-fructose diet and low-dose streptozotocin to model long-term type-2 diabetes complications. Sci. Rep 8(1):424. https://doi.org/10.1038/s41598-017-18896-5
Greenhalgh DG, Warden GD, Tissue V (2001) Wound care models. Surgical research. Academic Press, London, pp 379–91. International Standard Book Number 0-12-655330-0
Falanga V, Schrayer D, Cha J, Butmarc J, Carson P, Roberts AB et al (2004) Full-thickness wounding of the mouse tail as a model for delayed wound healing: accelerated wound closure in Smad3 knock-out mice. Wound Repair Regen 12(3):320–6. https://doi.org/10.1111/j.1067-1927.2004.012316.x
Wang X, Ge J, Tredget EE, Wu Y (2013) The mouse excisional wound splinting model, including applications for stem cell transplantation. Nat Protoc 8(2):302–9. https://doi.org/10.1038/nprot.2013.002
Galiano RD, Michaels JT, DObryansky MV, Levine JP, Gurtner GC (2004) Quantitative and reproducible murine model of excisional wound healing. Wound Repair Regen 12(4):485–92. https://doi.org/10.1111/j.1067-1927.2004.12404.x
Enoch S, Leaper DJ (2008) Basic science of wound healing. Surg Infect (Larchmt) 26(2):31–37. https://doi.org/10.1016/j.mpsur.2007.11.005
Wallace HA, Basehore BM, Zito PM (2017) Wound healing phases
Sorg H, Tilkorn DJ, Hager S, Hauser J, Mirastschijski U (2017) Skin wound healing: an update on the current knowledge and concepts. Eur Surg Res 58(1–2):81–94. https://doi.org/10.1159/000454919
Ellis S, Lin EJ, Tartar D (2018) Immunology of wound healing. Curr Dermatol Rep 7(4):350–8. https://doi.org/10.1007/s13671-018-0234-9
Barrientos S, Stojadinovic O, Golinko MS, Brem H, Tomic-Canic M (2008) Growth factors and cytokines in wound healing. Wound Repair Regen 16(5):585–601. https://doi.org/10.1111/j.1524-475X.2008.00410.x
Ridiandries A, Tan JTM, Bursill CA (2018) The role of chemokines in wound healing. Int J Mol Sci. https://doi.org/10.3390/ijms19103217
Behm B, Babilas P, Landthaler M, Schreml S (2012) Cytokines, chemokines and growth factors in wound healing. J Eur Acad Dermatol Venereol 26(7):812–20. https://doi.org/10.1111/j.1468-3083.2011.04415.x
Xiao T, Yan Z, Xiao S, Xia Y (2020) Proinflammatory cytokines regulate epidermal stem cells in wound epithelialization. Stem Cell Res Ther 11(1):232. https://doi.org/10.1186/s13287-020-01755-y
Kyaw B, Jaerbrink K, Martinengo L, Car J, Harding K, Schmidtchen A (2017) Need for improved definition of “chronic wounds” in clinical studies. Acta Dermato-Venereol 98(1):157–8. https://doi.org/10.2340/00015555-2786
Iqbal A, Jan A, Wajid MA, Tariq S (2017) Management of chronic non-healing wounds by hirudotherapy. World J Plast Surg 6(1):9–17
Boulton AJ, Kirsner RS, Vileikyte L (2004) Neuropathic diabetic foot ulcers. N Engl J Med 351(1):48–55. https://doi.org/10.1056/NEJMcp032966
Zhang Z, Liu P, Yang B, Li J, Wang W, Yang H et al (2021) Necrotizing fasciitis caused by diabetic foot. Int J Infect Dis 103:3–5. https://doi.org/10.1016/j.ijid.2020.11.132
Lyder CH, Ayello EA (2008) Pressure ulcers: a patient safety issue. Patient safety and quality: an evidence-based handbook for nurses. https://www.ncbi.nlm.nih.gov/books/NBK2631/?report=reader
Yarkony GM (1994) Pressure ulcers: a review. Arch Phys Med Rehabil 75(8):908–917. https://doi.org/10.1016/0003-9993(94)90117-1
Acknowledgements
As the corresponding author, I declare that this manuscript is original; that the article does not infringe upon any copyright or other proprietary rights of any third party; that neither the text nor the data have been reported or published previously.
Funding
VR is supported by an intramural Grant IMR Rai 12397B from the Western University of Health Sciences, Pomona, California. RM is supported by an intramural seed Grant IMR 12402F from the Western University of Health Sciences, Pomona, California. The research work of DKA is supported by the R01 HL144125 and R01 HL147662 grants from the National Institutes of Health, USA. The content of this critical review is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
VR wrote the initial draft; RM and DKA critically edited and reviewed the manuscript; VR, RM, DKA finalized the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All the authors have no conflict of interest and have read the journal’s authorship statement.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rai, V., Moellmer, R. & Agrawal, D.K. Clinically relevant experimental rodent models of diabetic foot ulcer. Mol Cell Biochem 477, 1239–1247 (2022). https://doi.org/10.1007/s11010-022-04372-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-022-04372-w