Abstract
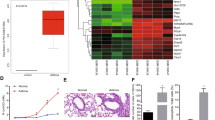
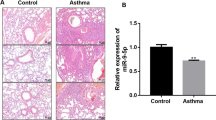
This study investigated the regulatory effects of microRNA-1278 (miR-1278) on airway inflammation, airway reconstruction, and the proliferation and apoptosis of airway smooth muscle cells (ASMCs) induced by transforming growth factor β1 (TGF-β1). The results showed that miR-1278 was upregulated in the blood and lung tissues (LTs) of patients with asthma compared with that in healthy volunteers; miR-1278 expression was also upregulated in asthmatic mice, and miR-1278 inhibition improved the LTs of asthmatic mice. Moreover, miR-1278 inhibited inflammation in asthmatic mice and counteracted the effect of TGF-β1 of induced proliferation and reduced apoptosis in ASMCs. DLRA indicated that miR-1278 targeted the 3′-UTR of Src-homology 2-containing phosphatase 1 (SHP-1). Furthermore, miR-1278 promoted ASMC proliferation, in which TGF-β1 played an important role by regulating the SHP-1/STAT3 signaling pathway. In conclusion, this study showed that miR-1278 played a critical role in the processes of airway remodeling and reduction of apoptosis by targeting SHP-1.







Similar content being viewed by others
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
References
Masoli M, Fabian D, Holt S et al (2004) The global burden of asthma: executive summary of the GINA Dissemination Committee report. Allergy 59:469–478
Thomsen AML, Ehrenstein V, Riis AH et al (2018) The potential impact of paternal age on risk of asthma in childhood: a study within the Danish National Birth Cohort. Respir Med 137:30–34
Holloway JW, Yang IA, Holgate ST (2010) Genetics of allergic disease. J Allergy Clin Immunol 125:S81–S94
Staab EB, Sanderson SD, Wells SM et al (2014) Treatment with the C5a receptor/CD88 antagonist PMX205 reduces inflammation in a murine model of allergic asthma. Int Immunopharmacol 21:293–300
Donohue JF, Herje N, Crater G et al (2014) Characterization of airway inflammation in patients with COPD using fractional exhaled nitric oxide levels: a pilot study. Int J Chron Obstruct Pulmon Dis 9:745
Gibeon D, Menzies-Gow A (2013) Recent changes in the drug treatment of allergic asthma. Clin Med 13:477
Halwani R, Al-Muhsen S, Al-Jahdali H et al (2011) Role of transforming growth factor–β in airway remodeling in asthma. Am J Respir Cell Mol Biol 44:127–133
Postma DS, Timens W (2006) Remodeling in asthma and chronic obstructive pulmonary disease. Proc Am Thorac Soc 3:434–439
Elias JA, Zhu Z, Chupp G et al (1999) Airway remodeling in asthma. J Clin Investig 104:1001–1006
Homer RJ, Elias JA (2005) Airway remodeling in asthma: therapeutic implications of mechanisms. Physiology 20:28–35
Wei Y, Xu Y-D, Yin L-M, et al (2013) Recombinant rat CC10 protein inhibits PDGF-induced airway smooth muscle cells proliferation and migration. BioMed Res Int
Ozier A, Allard B, Bara I, et al (2011) The pivotal role of airway smooth muscle in asthma pathophysiology. J Allergy
Makinde T, Murphy RF, Agrawal DK (2007) The regulatory role of TGF-β in airway remodeling in asthma. Immunol Cell Biol 85:348–356
Redington AE, Madden J, Frew AJ et al (1997) Transforming growth factor-β 1 in asthma: measurement in bronchoalveolar lavage fluid. Am J Respir Crit Care Med 156:642–647
Holgate ST, Polosa R (2008) Treatment strategies for allergy and asthma. Nat Rev Immunol 8:218–230
Halwani R, Al-Muhsen S, Al-Jahdali H et al (2011) Role of transforming growth factor-β in airway remodeling in asthma. Am J Respir Cell Mol Biol 44:127–133. https://doi.org/10.1165/rcmb.2010-0027TR
Freyer AM, Johnson SR, Hall IP (2001) Effects of growth factors and extracellular matrix on survival of human airway smooth muscle cells. Am J Respir Cell Mol Biol 25:569–576. https://doi.org/10.1165/ajrcmb.25.5.4605
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116:281–297
Pua HH, Ansel KM (2015) MicroRNA regulation of allergic inflammation and asthma. Curr Opin Immunol 36:101–108
Tay HL, Plank M, Collison A et al (2014) MicroRNA: potential biomarkers and therapeutic targets for allergic asthma? Ann Med 46:633–639
Perry MM, Baker JE, Gibeon DS et al (2014) Airway smooth muscle hyperproliferation is regulated by microRNA-221 in severe asthma. Am J Respir Cell Mol Biol 50:7–17
Hu R, Pan W, Fedulov AV et al (2014) MicroRNA-10a controls airway smooth muscle cell proliferation via direct targeting of the PI3 kinase pathway. FASEB J 28:2347–2357
Zhang H, Sun Z, Yu L et al (2017) MiR-139-5p inhibits proliferation and promoted apoptosis of human airway smooth muscle cells by downregulating the Brg1 gene. Respir Physiol Neurobiol 246:9–16
Zhang X, Zhao X, Sun H et al (2018) The role of miR-29c/B7-H3 axis in children with allergic asthma. J Transl Med 16:218
Jude JA, Dileepan M, Subramanian S et al (2012) miR-140-3p regulation of TNF-α-induced CD38 expression in human airway smooth muscle cells. Am J Physiol-Lung Cell Mol Physiol 303:L460–L468
Zhang Y, Xue Y, Liu Y et al (2016) MicroRNA-146a expression inhibits the proliferation and promotes the apoptosis of bronchial smooth muscle cells in asthma by directly targeting the epidermal growth factor receptor. Exp Ther Med 12:854–858
Wen X, Yan J, Han X-R et al (2018) PTEN gene silencing contributes to airway remodeling and induces airway smooth muscle cell proliferation in mice with allergic asthma. J Thorac Dis 10:202
Ye L, Mou Y, Wang J et al (2017) Effects of microRNA-19b on airway remodeling, airway inflammation and degree of oxidative stress by targeting TSLP through the Stat3 signaling pathway in a mouse model of asthma. Oncotarget 8:47533
Cheng Z, Dai L-L, Wang X et al (2017) MicroRNA-145 down-regulates mucin 5AC to alleviate airway remodeling and targets EGFR to inhibit cytokine expression. Oncotarget 8:46312
Association GAotWM (2014) World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. J Am Coll Dentists 81:14
Agarwal V, Bell GW, Nam JW et al (2015) Predicting effective microRNA target sites in mammalian mRNAs. Elife 4:e05005. https://doi.org/10.7554/eLife.05005
Dharmage SC, Perret JL, Custovic A (2019) Epidemiology of asthma in children and adults. Front Pediatrics 7:246
Grainge CL, Lau LC, Ward JA et al (2011) Effect of bronchoconstriction on airway remodeling in asthma. N Engl J Med 364:2006–2015
Hong W, Peng G, Hao B et al (2017) Nicotine-induced airway smooth muscle cell proliferation involves TRPC6-dependent calcium influx via α7 nAChR. Cell Physiol Biochem 43:986–1002
Wenzel SE (2012) Asthma phenotypes: the evolution from clinical to molecular approaches. Nat Med 18:716–725. https://doi.org/10.1038/nm.2678
Tan H, Lei J, Xue L et al (2017) Relaxing effect of TSU-68, an antiangiogenic agent, on mouse airway smooth muscle. Cell Physiol Biochem 41:2350–2362
Samitas K, Delimpoura V, Zervas E et al (2015) Anti-IgE treatment, airway inflammation and remodelling in severe allergic asthma: current knowledge and future perspectives. Eur Respir Rev 24:594–601
Murakami A, Ohigashi H (2007) Targeting NOX, INOS and COX-2 in inflammatory cells: chemoprevention using food phytochemicals. Int J Cancer 121:2357–2363
Islam T, Breton C, Salam MT et al (2010) Role of inducible nitric oxide synthase in asthma risk and lung function growth during adolescence. Thorax 65:139–145
Singh VP, Patil CS, Jain NK et al (2003) Effect of nimesulide on acetic acid-and leukotriene-induced inflammatory bowel disease in rats. Prostaglandins Lipid Mediat 71:163–175
Yamauchi K (2006) Airway remodeling in asthma and its influence on clinical pathophysiology. Tohoku J Exp Med 209:75–87
Hirota S, Helli PB, Catalli A et al (2005) Airway smooth muscle excitation-contraction coupling and airway hyperresponsiveness. Can J Physiol Pharmacol 83:725–732
Hirst SJ (2003) Regulation of airway smooth muscle cell immunomodulatory function: role in asthma. Respir Physiol Neurobiol 137:309–326
Bara I, Ozier A, De Lara JT et al (2010) Pathophysiology of bronchial smooth muscle remodelling in asthma. Eur Respir J 36:1174–1184
Al-Muhsen S, Johnson JR, Hamid Q et al (2011) Remodeling in asthma. J Allergy Clin Immunol 128:451–462
Zhao Y, Wang P, Wu Q (2020) miR-1278 sensitizes nasopharyngeal carcinoma cells to cisplatin and suppresses autophagy via targeting ATG2B. Mol Cell Probes 53:101597
Zhou X, Lv L, Zhang Z et al (2020) LINC00294 negatively modulates cell proliferation in glioma through a neurofilament medium (NEFM)-mediated pathway via interacting with miR-1278. J Gene Med 22(10):e3235
Acknowledgements
Not applicable.
Funding
This work was supported by the NSFC (No. 81770028); the Key Laboratory of Shenzhen Respiratory Disease (No. ZDSYS201504301616234) and the Clinical Research of Shenzhen Municipal Health and Family Planning Commission (No. SZLY2017024). This work also was supported by special funding for high-level disciplines from the Shenzhen Institute of Respiratory Diseases.
Author information
Authors and Affiliations
Contributions
JL, RC, YL and YZ conceived and designed the research and interpreted the results of experiments. JL and RC performed experiments, analyzed data. All authors approved final version of manuscript. JL and RC edited and revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures were reviewed and approved by the Ethics Committee (EC) of Shenzhen People’s Hospital (The First Affiliated Hospital of Southern University of Science and Technology, The Second Clinical Medical College of Jinan University).
Informed consent
Written informed consent was obtained and signed by all patients or their legal guardian.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, J., Chen, R., Lu, Y. et al. The microRNA-1278/SHP-1/STAT3 pathway is involved in airway smooth muscle cell proliferation in a model of severe asthma both intracellularly and extracellularly. Mol Cell Biochem 477, 1439–1451 (2022). https://doi.org/10.1007/s11010-022-04358-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-022-04358-8