Abstract
Multiple circular RNAs (circRNAs) have been identified to act as essential mediators in diverse human cancers. However, the roles of circRNAs in neuroblastoma (NB) are largely unknown. In this study, we aimed to explore the function of circKIF2A in NB. Quantitative real-time polymerase chain reaction was executed to detect the levels of circKIF2A, KIF2A mRNA, miR-129-5p and polo-like kinase 4 (PLK4) mRNA. Actinomycin D assay and RNase R digestion assay were conducted to analyze the feature of circKIF2A. 3-(4, 5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide (MTT) assay, transwell assay and specific kits were utilized to evaluate cell proliferation, metastasis and glycolysis, respectively. Western blot assay was performed to examine the protein levels of matrix metalloproteinase 2 (MMP2), MMP9 and PLK4. Bioinformatics analysis, RNA pull-down assay and dual-luciferase reporter assay were conducted to analyze the relationship between miR-129-5p and circKIF2A or PLK4. Murine xenograft model assay was done to investigate the role of circKIF2A in NB in vivo. CircKIF2A level was increased in NB tissue samples and cell lines. Silencing of circKIF2A impeded NB cell proliferation, migration, invasion and glycolysis. For mechanism analysis, circKIF2A could positively modulate PLK4 expression via sponging miR-129-5p. Moreover, miR-129-5p inhibition reversed the inhibitory effects of circKIF2A silencing on the behaviors of NB cells. MiR-129-5p overexpression weakened the malignant biological behaviors of NB cells by targeting PLK4. Additionally, circKIF2A knockdown hampered tumorigenesis in vivo. CircKIF2A knockdown suppressed cell proliferation, migration, invasion and glycolysis via downregulating PLK4 expression through miR-129-5p.









Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article.
Abbreviations
- NB:
-
Neuroblastoma
- PLK4:
-
Polo-like kinase 4
- MMP2:
-
Matrix metalloproteinase 2
References
Park JR, Eggert A, Caron H (2008) Neuroblastoma: biology, prognosis, and treatment. Pediatr Clin North Am 55(1):97–120
Pugh TJ, Morozova O, Attiyeh EF, Asgharzadeh S, Wei JS, Auclair D, Carter SL, Cibulskis K, Hanna M, Kiezun A et al (2013) The genetic landscape of high-risk neuroblastoma. Nat Genet 45(3):279–284
Garaventa A, Parodi S, De Bernardi B, Dau D, Manzitti C, Conte M, Casale F, Viscardi E, Bianchi M, D’Angelo P et al (2009) Outcome of children with neuroblastoma after progression or relapse. A retrospective study of the Italian neuroblastoma registry. Eur J Cancer 45(16):2835–2842
Whittle SB, Smith V, Doherty E, Zhao S, McCarty S, Zage PE (2017) Overview and recent advances in the treatment of neuroblastoma. Expert Rev Anticancer Ther 17(4):369–386
Jeck WR, Sharpless NE (2014) Detecting and characterizing circular RNAs. Nat Biotechnol 32(5):453–461
Qu S, Zhong Y, Shang R, Zhang X, Song W, Kjems J, Li H (2017) The emerging landscape of circular RNA in life processes. RNA Biol 14(8):992–999
Liang HF, Zhang XZ, Liu BG, Jia GT, Li WL (2017) Circular RNA circ-ABCB10 promotes breast cancer proliferation and progression through sponging miR-1271. Am J Cancer Res 7(7):1566–1576
Zhang XL, Xu LL, Wang F (2017) Hsa_circ_0020397 regulates colorectal cancer cell viability, apoptosis and invasion by promoting the expression of the miR-138 targets TERT and PD-L1. Cell Biol Int 41(9):1056–1064
Li J, Zhen L, Zhang Y, Zhao L, Liu H, Cai D, Chen H, Yu J, Qi X, Li G (2017) Circ-104916 is downregulated in gastric cancer and suppresses migration and invasion of gastric cancer cells. Onco Targets Ther 10:3521–3529
Mahmoudi E, Kiltschewskij D, Fitzsimmons C, Cairns MJ (2019) Depolarization-associated CircRNA regulate neural gene expression and in some cases may function as templates for translation. Cells 9(1):25
He L, Hannon GJ (2004) MicroRNAs: small RNAs with a big role in gene regulation. Nat Rev Genet 5(7):522–531
Lee JJ, Drakaki A, Iliopoulos D, Struhl K (2012) MiR-27b targets PPARgamma to inhibit growth, tumor progression and the inflammatory response in neuroblastoma cells. Oncogene 31(33):3818–3825
Welch C, Chen Y, Stallings RL (2007) MicroRNA-34a functions as a potential tumor suppressor by inducing apoptosis in neuroblastoma cells. Oncogene 26(34):5017–5022
Wang X, Li J, Xu X, Zheng J, Li Q (2018) miR-129 inhibits tumor growth and potentiates chemosensitivity of neuroblastoma by targeting MYO10. Biomed Pharmacother 103:1312–1318
Wang J, Zuo J, Wang M, Ma X, Gao K, Bai X, Wang N, Xie W, Liu H (2019) Pololike kinase 4 promotes tumorigenesis and induces resistance to radiotherapy in glioblastoma. Oncol Rep 41(4):2159–2167
Liao Z, Zhang H, Fan P, Huang Q, Dong K, Qi Y, Song J, Chen L, Liang H, Chen X et al (2019) High PLK4 expression promotes tumor progression and induces epithelial–mesenchymal transition by regulating the Wnt/betacatenin signaling pathway in colorectal cancer. Int J Oncol 54(2):479–490
Marina M, Saavedra HI (2014) Nek2 and Plk4: prognostic markers, drivers of breast tumorigenesis and drug resistance. Front Biosci (Landmark Ed) 19:352–365
Zhang N, Liu FL, Ma TS, Zhang ZZJ (2019) LncRNA SNHG1 contributes to tumorigenesis and mechanism by targeting miR-338-3p to regulate PLK4 in human neuroblastoma. Eur Rev Med Pharmacol Sci 23(20):8971–8983
Glazar P, Papavasileiou P, Rajewsky N (2014) circBase: a database for circular RNAs. RNA 20(11):1666–1670
Li JH, Liu S, Zhou H, Qu LH, Yang JH (2014) starBase v2.0: decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA interaction networks from large-scale CLIP-Seq data. Nucleic Acids Res 42(Database issue):D92-97
Marchiq I, Pouyssegur J (2016) Hypoxia, cancer metabolism and the therapeutic benefit of targeting lactate/H(+) symporters. J Mol Med (Berl) 94(2):155–171
Li C, Zhang G, Zhao L, Ma Z, Chen H (2016) Metabolic reprogramming in cancer cells: glycolysis, glutaminolysis, and Bcl-2 proteins as novel therapeutic targets for cancer. World J Surg Oncol 14(1):15
Kulcheski FR, Christoff AP, Margis R (2016) Circular RNAs are miRNA sponges and can be used as a new class of biomarker. J Biotechnol 238:42–51
Sun X, Xue H, Xiong Y, Yu R, Gao X, Qian M, Wang S, Wang H, Xu J, Chen Z et al (2019) GALE promotes the proliferation and migration of glioblastoma cells and is regulated by miR-let-7i-5p. Cancer Manag Res 11:10539–10554
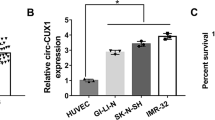
Li H, Yang F, Hu A, Wang X, Fang E, Chen Y, Li D, Song H, Wang J, Guo Y et al (2019) Therapeutic targeting of circ-CUX1/EWSR1/MAZ axis inhibits glycolysis and neuroblastoma progression. EMBO Mol Med 11(12):e10835
Pavlova NN, Thompson CB (2016) The emerging hallmarks of cancer metabolism. Cell Metab 23(1):27–47
Seyfried TN, Mukherjee P (2005) Targeting energy metabolism in brain cancer: review and hypothesis. Nutr Metab (Lond) 2:30
Bordone MP, Salman MM, Titus HE, Amini E, Andersen JV, Chakraborti B, Diuba AV, Dubouskaya TG, Ehrke E, Espindola de Freitas A et al (2019) The energetic brain—a review from students to students. J Neurochem 151(2):139–165
Li M, Tian L, Wang L, Yao H, Zhang J, Lu J, Sun Y, Gao X, Xiao H, Liu M (2013) Down-regulation of miR-129-5p inhibits growth and induces apoptosis in laryngeal squamous cell carcinoma by targeting APC. PLoS ONE 8(10):e77829
Wang Q, Yu J (2018) MiR-129-5p suppresses gastric cancer cell invasion and proliferation by inhibiting COL1A1. Biochem Cell Biol 96(1):19–25
Chen D, Wang H, Chen J, Li Z, Li S, Hu Z, Huang S, Zhao Y, He X (2018) MicroRNA-129-5p regulates glycolysis and cell proliferation by targeting the glucose transporter SLC2A3 in gastric cancer cells. Front Pharmacol 9:502
Tian X, Zhou D, Chen L, Tian Y, Zhong B, Cao Y, Dong Q, Zhou M, Yan J, Wang Y et al (2018) Polo-like kinase 4 mediates epithelial-mesenchymal transition in neuroblastoma via PI3K/Akt signaling pathway. Cell Death Dis 9(2):54
Abbott NJ, Patabendige AA, Dolman DE, Yusof SR, Begley DJ (2010) Structure and function of the blood-brain barrier. Neurobiol Dis 37(1):13–25
Franco C, Gerhardt H (2012) Tissue engineering: blood vessels on a chip. Nature 488(7412):465–466
Salman MM, Marsh G, Kusters I, Delince M, Di Caprio G, Upadhyayula S, de Nola G, Hunt R, Ohashi KG, Gray T et al (2020) Design and validation of a human brain endothelial microvessel-on-a-chip open microfluidic model enabling advanced optical imaging. Front Bioeng Biotechnol 8:573775
Cochrane A, Albers HJ, Passier R, Mummery CL, van den Berg A, Orlova VV, van der Meer AD (2019) Advanced in vitro models of vascular biology: human induced pluripotent stem cells and organ-on-chip technology. Adv Drug Deliv Rev 140:68–77
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no financial conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11010_2021_4096_MOESM1_ESM.tif
Figure S1. The selection of miR-129-5p and PLK4. (A) The expression levels of (miR-367-5p, miR-6134, miR-6839-3p, miR-6851-5p, miR-1250-3p, miR-1270, miR-129-5p, miR-1301-3p, miR-136-5p and miR-154-3p in NB tissues were detected by qRT-PCR assay. (B and C) The expression levels of miR-129-5p and miR-1301-3p in SK-N-SH and SK-N-AS cells transfected with si-NC or si-circKIF2A were detected by qRT-PCR assay. (D) The levels of SIAH1, PLK1, BCL2, HOXA7, CREB1 and BCL11A in NB tissues were measured by qRT-PCR assay. (E and F) The levels of SIAH1, PLK1, BCL2, HOXA7, CREB1 and BCL11A in SK-N-SH and SK-N-AS cells transfected with miR-NC or miR-129-5p were detected by qRT-PCR assay. *P<0.05. Supplementary file1 (TIF 759 KB)
Rights and permissions
About this article
Cite this article
Yang, Y., Pan, H., Chen, J. et al. CircKIF2A contributes to cell proliferation, migration, invasion and glycolysis in human neuroblastoma by regulating miR-129-5p/PLK4 axis. Mol Cell Biochem 476, 2513–2525 (2021). https://doi.org/10.1007/s11010-021-04096-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-021-04096-3