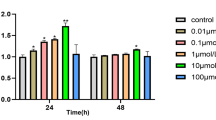
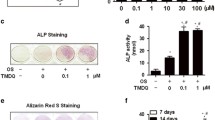
Abstract
Baicalin (BAI), a sort of flavonoid monomer, acquires from Scutellaria baicalensis Georgi, which was forcefully reported in diversified ailments due to the pleiotropic properties. But, the functions of BAI in osteoblast differentiation have not been addressed. The intentions of this study are to attest the influences of BAI in the differentiation of osteoblasts. MC3T3-E1 cells or rat primary osteoblasts were exposed to BAI, and then cell viability, ALP activity, mineralization process, and Runx2 and Ocn expression were appraised through implementing CCK-8, p-nitrophenyl phosphate (pNPP), Alizarin red staining, western blot, and RT-qPCR assays. The microRNA-217 (miR-217) expression was evaluated in MC3T3-E1 cells or rat primary osteoblasts after BAI disposition; meanwhile, the functions of miR-217 in BAI-administrated MC3T3-E1 cells were estimated after miR-217 inhibitor transfection. The impacts of BAI and miR-217 inhibition on Wnt/β-catenin and MEK/ERK pathways were probed to verify the involvements in BAI-regulated the differentiation of osteoblasts. BAI accelerated cell viability, osteoblast activity, and Runx2 and Ocn expression in MC3T3-E1 cells or rat primary osteoblasts, and the phenomena were mediated via activations of Wnt/β-catenin and MEK/ERK pathways. Elevation of miR-217 was observed in BAI-disposed MC3T3-E1 cells or rat primary osteoblasts, and miR-217 repression annulled the functions of BAI in MC3T3-E1 cell viability and differentiation. Additionally, the activations of Wnt/β-catenin and MEK/ERK pathways evoked by BAI were both restrained by repression of miR-217. These explorations uncovered that BAI augmented the differentiation of osteoblasts via activations of Wnt/β-catenin and MEK/ERK pathways by ascending miR-217 expression.







Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Change history
29 February 2024
This article has been retracted. Please see the Retraction Notice for more detail: https://doi.org/10.1007/s11010-024-04980-8
References
Meinberg EG, Agel J, Roberts CS, Karam MD, Kellam JF (2018) Fracture and dislocation classification compendium—2018. J Orthop Trauma 32(Suppl 1):S1–S170. https://doi.org/10.1097/bot.0000000000001063
Tsuda T (2017) Epidemiology of fragility fractures and fall prevention in the elderly: a systematic review of the literature. Curr Orthop Pract 28(6):580–585. https://doi.org/10.1097/BCO.0000000000000563
Bonafede M, Espindle D, Bower AG (2013) The direct and indirect costs of long bone fractures in a working age US population. J Med Econ 16(1):169–178. https://doi.org/10.3111/13696998.2012.737391
Hwang K, Ki SJ, Ko SH (2017) Etiology of nasal bone fractures. J Craniofac Surg 28(3):785–788
Kilicarslan K, Bektaser B, Ucguder A, Dogan M, Ugurlu M, Yildirim H (2011) Results of early surgical treatment of intra-articular complex fractures of the humerus in adults. Bratisl lek listy 112(9):501–505
Moon S-H, Kim H-S, Jung S-N, Kwon H (2016) The efficacy of transverse fixation and early exercise in the treatment of fourth metacarpal bone fractures. Arch Plast Surg 43(2):189–196. https://doi.org/10.5999/aps.2016.43.2.189
Jeong SJ, Jeong MJ (2016) Effect of thymosin beta4 on the differentiation and mineralization of MC3T3-E1 cell on a titanium surface. J Nanosci Nanotechnol 16(2):1979–1983
Zhang J, Zhang H, Deng X, Zhang Y, Xu K (2017) Baicalin protects AML-12 cells from lipotoxicity via the suppression of ER stress and TXNIP/NLRP3 inflammasome activation. Chem Biol Interact 278:189–196. https://doi.org/10.1016/j.cbi.2017.10.010
Lai W, Jia J, Yan B, Jiang Y, Shi Y, Chen L, Mao C, Liu X, Tang H, Gao M, Cao Y, Liu S, Tao Y (2017) Baicalin hydrate inhibits cancer progression in nasopharyngeal carcinoma by affecting genome instability and splicing. Oncotarget 9(1):901–914. https://doi.org/10.18632/oncotarget.22868
Lin D, Du Q, Wang H, Gao G, Zhou J, Ke L, Chen T, Shaw C, Rao P (2017) Antidiabetic micro-/nanoaggregates from ge-gen-qin-lian-tang decoction increase absorption of baicalin and cellular antioxidant activity in vitro. Biomed Res Int 2017:9217912. https://doi.org/10.1155/2017/9217912
Xu Z, Wang F, Tsang SY, Ho KH, Zheng H, Yuen CT, Chow CY, Xue H (2006) Anxiolytic-like effect of baicalin and its additivity with other anxiolytics. Planta Med 72(2):189–192. https://doi.org/10.1055/s-2005-873193
Dinda B, Dinda S, DasSharma S, Banik R, Chakraborty A, Dinda M (2017) Therapeutic potentials of baicalin and its aglycone, baicalein against inflammatory disorders. Eur J Med Chem 131:68–80. https://doi.org/10.1016/j.ejmech.2017.03.004
Moghaddam E, Teoh BT, Sam SS, Lani R, Hassandarvish P, Chik Z, Yueh A, Abubakar S, Zandi K (2014) Baicalin, a metabolite of baicalein with antiviral activity against dengue virus. Sci Rep 4:5452. https://doi.org/10.1038/srep05452
Kim JY, Lee JI, Song M, Lee D, Song J, Kim SY, Park J, Choi HY, Kim H (2015) Effects of Eucommia ulmoides extract on longitudinal bone growth rate in adolescent female rats. PTR 29(1):148–153. https://doi.org/10.1002/ptr.5195
Lu L, Rao L, Jia H, Chen J, Lu X, Yang G, Li Q, Lee KKH, Yang L (2017) Baicalin positively regulates osteoclast function by activating MAPK/Mitf signalling. J Cell Mol Med 21(7):1361–1372. https://doi.org/10.1111/jcmm.13066
Wu X, Zhi F, Lun W, Deng Q, Zhang W (2018) Baicalin inhibits PDGF-BB-induced hepatic stellate cell proliferation, apoptosis, invasion, migration and activation via the miR-3595/ACSL4 axis. Int J Mol Med 41(4):1992–2002. https://doi.org/10.3892/ijmm.2018.3427
Wang L, Zhang R, Chen J, Wu Q, Kuang Z (2017) Baicalin protects against TNF-alpha-induced injury by down-regulating miR-191a that targets the tight junction protein ZO-1 in IEC-6 cells. Biol Pharm Bull 40(4):435–443. https://doi.org/10.1248/bpb.b16-00789
Wei R, Deng Z, Su J (2015) miR-217 targeting Wnt5a in osteosarcoma functions as a potential tumor suppressor. Biomed Pharmacother 72:158–164. https://doi.org/10.1016/j.biopha.2015.04.012
Su J, Wang Q, Liu Y, Zhong M (2014) miR-217 inhibits invasion of hepatocellular carcinoma cells through direct suppression of E2F3. Mol Cell Biochem 392(1–2):289–296. https://doi.org/10.1007/s11010-014-2039-x
Zhu YL, Wang S, Ding DG, Xu L, Zhu HT (2017) miR217 inhibits osteogenic differentiation of rat bone marrowderived mesenchymal stem cells by binding to Runx2. Mol Med Rep 15(5):3271–3277. https://doi.org/10.3892/mmr.2017.6349
Peng Y, Fu Z-Z, Guo C-S, Zhang Y-X, Di Y, Jiang B, Li Q-W (2015) Effects and mechanism of baicalin on apoptosis of cervical cancer HeLa cells in vitro. IJPR 14(1):251–261
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408
Guo AJ, Choi RC, Cheung AW, Chen VP, Xu SL, Dong TT, Chen JJ, Tsim KW (2011) Baicalin, a flavone, induces the differentiation of cultured osteoblasts: an action via the Wnt/beta-catenin signaling pathway. J Biol Chem 286(32):27882–27893. https://doi.org/10.1074/jbc.M111.236281
Noguchi T, Ebina K, Hirao M, Otsuru S, Guess AJ, Kawase R, Ohama T, Yamashita S, Etani Y, Okamura G, Yoshikawa H (2018) Apolipoprotein E plays crucial roles in maintaining bone mass by promoting osteoblast differentiation via ERK1/2 pathway and by suppressing osteoclast differentiation via c-Fos, NFATc1, and NF-kappaB pathway. Biochem Biophys Res Commun 503(2):644–650. https://doi.org/10.1016/j.bbrc.2018.06.055
Feng G, Zhang J, Feng X, Wu S, Huang D, Hu J, Zhu S, Song D (2016) Runx2 modified dental pulp stem cells (DPSCs) enhance new bone formation during rapid distraction osteogenesis (DO). Differ Res Biol Divers 92(4):195–203. https://doi.org/10.1016/j.diff.2016.06.001
Bustos F, Sepúlveda H, Prieto CP, Carrasco M, Díaz L, Palma J, Lattus J, Montecino M, Palma V (2017) RUNX2 induction during differentiation of Wharton’s jelly mesenchymal stem cells to osteoblasts is regulated by JARID1B histone demethylase. Stem Cells (Dayton, Ohio) 35(12):2430–2441
Al Rifai O, Chow J, Lacombe J, Julien C, Faubert D, Susan-Resiga D, Essalmani R, Creemers JW, Seidah NG, Ferron M (2017) Proprotein convertase furin regulates osteocalcin and bone endocrine function. J Clin Investig 127(11):4104–4117. https://doi.org/10.1172/jci93437
Kang IH, Jeong BC, Hur SW, Choi H, Choi SH, Ryu JH, Hwang YC, Koh JT (2015) MicroRNA-302a stimulates osteoblastic differentiation by repressing COUP-TFII expression. J Cell Physiol 230(4):911–921. https://doi.org/10.1002/jcp.24822
Mai ZH, Peng ZL, Zhang JL, Chen L, Liang HY, Cai B, Ai H (2013) miRNA expression profile during fluid shear stress-induced osteogenic differentiation in MC3T3-E1 cells. Chin Med J 126(8):1544–1550
Shan Z, Cheng N, Huang R, Zhao B, Zhou Y (2018) Puerarin promotes the proliferation and differentiation of MC3T3-E1 cells via microRNA-106b by targeting receptor activator of nuclear factor-kappaB ligand. Exp Ther Med 15(1):55–60. https://doi.org/10.3892/etm.2017.5405
Huang Z, Cheng C, Wang J, Liu X, Wei H, Han Y, Yang S, Wang X (2018) Icariin regulates the osteoblast differentiation and cell proliferation of MC3T3-E1 cells through microRNA-153 by targeting Runt-related transcription factor 2. Exp Ther Med 15(6):5159–5166. https://doi.org/10.3892/etm.2018.6127
Zhu H, Hou L, Liu J, Li Z (2016) MiR-217 is down-regulated in psoriasis and promotes keratinocyte differentiation via targeting GRHL2. Biochem Biophys Res Commun 471(1):169–176. https://doi.org/10.1016/j.bbrc.2016.01.157
Zhang Y, Xie RL, Croce CM, Stein JL, Lian JB, van Wijnen AJ, Stein GS (2011) A program of microRNAs controls osteogenic lineage progression by targeting transcription factor Runx2. Proc Natl Acad Sci USA 108(24):9863–9868. https://doi.org/10.1073/pnas.1018493108
Fei Y, Xiao L, Doetschman T, Coffin DJ, Hurley MM (2011) Fibroblast growth factor 2 stimulation of osteoblast differentiation and bone formation is mediated by modulation of the Wnt signaling pathway. J Biol Chem 286(47):40575–40583. https://doi.org/10.1074/jbc.M111.274910
Kim MB, Song Y, Hwang JK (2014) Kirenol stimulates osteoblast differentiation through activation of the BMP and Wnt/beta-catenin signaling pathways in MC3T3-E1 cells. Fitoterapia 98:59–65. https://doi.org/10.1016/j.fitote.2014.07.013
Guo C, Yang RJ, Jang K, Zhou XL, Liu YZ (2017) Protective effects of pretreatment with quercetin against lipopolysaccharide-induced apoptosis and the inhibition of osteoblast differentiation via the MAPK and Wnt/beta-catenin pathways in MC3T3-E1 Cells. Cell Physiol Biochem 43(4):1547–1561. https://doi.org/10.1159/000481978
Zhai F, Song N, Ma J, Gong W, Tian H, Li X, Jiang C, Wang H (2017) FGF18 inhibits MC3T3E1 cell osteogenic differentiation via the ERK signaling pathway. Mol Med Rep 16(4):4127–4132. https://doi.org/10.3892/mmr.2017.7088
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article has been retracted. Please see the retraction notice for more detail:https://doi.org/10.1007/s11010-024-04980-8
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure 1
BAI expedited rat primary osteoblast viability and osteogenic differentiation. After BAI management (20, 50, and 100 μM), (A) CCK-8 assay for rat primary osteoblast viability evaluation; (B) p-nitrophenyl phosphate (pNPP) assay for ALP activity detection; (C) western blot assay for Runx2 and Ocn protein level estimation; (D and E) RT-qPCR assay for Runx2 and Ocn mRNA expression level determination. *P < 0.05, **P < 0.01, ***P < 0.001 (TIFF 641 kb)
Supplementary Figure 2
BAI promoted osteoblast activity. After BAI management (20, 50, and 100 μM, TRAP activity assay for mature osteoclasts evaluation in (A) MC3T3-E1 cells and (B) rat primary osteoblasts. **P < 0.01, ***P < 0.001 (TIFF 252 kb)
Supplementary Figure 3
BAI expedited the activations of Wnt/β-catenin and MEK/ERK pathways in rat primary osteoblasts. After BAI administration (20, 50, and 100 μM), (A and B) western blot assay for wnt3a and β-catenin protein level measurement; (C and D) western blot assay for p/t-MEK and p/t-ERK protein level determination. *P < 0.05, **P < 0.01 (TIFF 1437 kb)
Supplementary Figure 4
Enhancement of miR-217 was triggered by BAI in rat primary osteoblasts. After BAI management (20, 50, and 100 μM), RT-qPCR assay for the expression level of miR-217 evaluation in rat primary osteoblasts. *P < 0.05, **P < 0.01 (TIFF 171 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Q., Shi, D., Geng, Y. et al. RETRACTED ARTICLE: Baicalin augments the differentiation of osteoblasts via enhancement of microRNA-217. Mol Cell Biochem 463, 91–100 (2020). https://doi.org/10.1007/s11010-019-03632-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-019-03632-6