Abstract
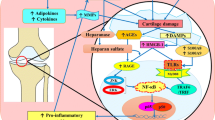
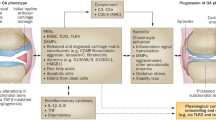
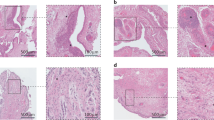
Osteoarthritis (OA) is a degenerative disease characterized by the destruction of cartilage. The greatest risk factors for the development of OA include age and obesity. Recent studies suggest the role of inflammation in the pathogenesis of OA. The two most common locations for OA to occur are in the knee and hip joints. The knee joint experiences more mechanical stress, cartilage degeneration, and inflammation than the hip joint. This could contribute to the increased incidence of OA in the knee joint. Damage-associated molecular patterns (DAMPs), including high-mobility group box-1, receptor for advanced glycation end products, and alarmins (S100A8 and S100A9), are released in the joint in response to stress-mediated chondrocyte and cartilage damage. This facilitates increased cartilage degradation and inflammation in the joint. Studies have documented the role of DAMPs in the pathogenesis of OA; however, the comparison of DAMPs and its influence on OA has not been discussed. In this study, we compared the DAMPs between OA knee and hip joints and found a significant difference in the levels of DAMPs expressed in the knee joint compared to the hip joint. The increased levels of DAMPs suggest a difference in the underlying pathogenesis of OA in the knee and the hip and highlights DAMPs as potential therapeutic targets for OA in the future.






Similar content being viewed by others
References
Yan CH, Chan WL, Yuen WH, Yung PS, Ip KY, Fan JC, Chiu KY (2015) Efficacy and safety of hylan G-F 20 injection in treatment of knee osteoarthritis in Chinese patients: results of a prospective, multicentre, longitudinal study. Hong Kong Med J 21(4):327–332
Turkiewicz A, Petersson IF, Bjork J, Hawker G, Dahlberg LE, Lohmander LS, Englund M (2014) Current and future impact of osteoarthritis on health care: a population-based study with projections to year 2032. Osteoarthr Cartil 22(11):1826–1832
Sokolove J, Lepus CM (2013) Role of inflammation in the pathogenesis of osteoarthritis: latest findings and interpretations. Ther Adv Musculoskelet Dis 5(2):77–94
Rai V, Dietz NE, Dilisio MF, Radwan MM, Agrawal DK (2016) Vitamin D attenuates inflammation, fatty infiltration, and cartilage loss in the knee of hyperlipidemic microswine. Arthritis Res Ther 18(1):203
Attur M, Belitskaya-Lévy I, Oh C, Krasnokutsky S, Greenberg J, Samuels J, Smiles S, Lee S, Patel J, Al-Mussawir H, McDaniel G, Kraus VB, Abramson SB (2011) Increased interleukin-1β gene expression in peripheral blood leukocytes is associated with increased pain and predicts risk for progression of symptomatic knee osteoarthritis. Arthritis Rheumatol 63(7):1908–1917
Chen YJ, Chan DC, Chiang CK, Wang CC, Yang TH, Lan KC, Chao SC, Tsai KS, Yang RS, Liu SH (2016) Advanced glycation end-products induced VEGF production and inflammatory responses in human synoviocytes via RAGE-NF-kappaB pathway activation. J Orthop Res 34(5):791–800
Dreier R, Grassel S, Fuchs S, Schaumburger J, Bruckner P (2004) Pro-MMP-9 is a specific macrophage product and is activated by osteoarthritic chondrocytes via MMP-3 or a MT1-MMP/MMP-13 cascade. Exp Cell Res 297(2):303–312
Dreier R, Wallace S, Fuchs S, Bruckner P, Grassel S (2001) Paracrine interactions of chondrocytes and macrophages in cartilage degradation: articular chondrocytes provide factors that activate macrophage-derived pro-gelatinase B (pro-MMP-9). J Cell Sci 114(Pt 21):3813–3822
Garcia-Arnandis I, Guillen MI, Gomar F, Pelletier JP, Martel-Pelletier J, Alcaraz MJ (2010) High mobility group box 1 potentiates the pro-inflammatory effects of interleukin-1beta in osteoarthritic synoviocytes. Arthritis Res Ther 12(4):R165
Goldring MB, Birkhead JR, Suen LF, Yamin R, Mizuno S, Glowacki J, Arbiser JL, Apperley JF (1994) Interleukin-1 beta-modulated gene expression in immortalized human chondrocytes. J Clin Invest 94(6):2307–2316
Liu-Bryan R (2013) Synovium and the innate inflammatory network in osteoarthritis progression. Curr Rheumatol Rep 15(5):323
Rasheed Z, Akhtar N, Haqqi TM (2011) Advanced glycation end products induce the expression of interleukin-6 and interleukin-8 by receptor for advanced glycation end product-mediated activation of mitogen-activated protein kinases and nuclear factor-kappaB in human osteoarthritis chondrocytes. Rheumatology 50(5):838–851
Sillat T, Barreto G, Clarijs P, Soininen A, Ainola M, Pajarinen J, Korhonen M, Konttinen YT, Sakalyte R, Hukkanen M, Ylinen P, Nordström DC (2013) Toll-like receptors in human chondrocytes and osteoarthritic cartilage. Acta Orthop 84(6):585–592
Zeng GQ, Chen AB, Li W, Song JH, Gao CY (2015) High MMP-1, MMP-2, and MMP-9 protein levels in osteoarthritis. Genet Mol Res 14(4):14811–14822
Sun XH, Liu Y, Han Y, Wang J (2016) Expression and significance of high-mobility group protein B1 (HMGB1) and the receptor for advanced glycation end-product (RAGE) in knee osteoarthritis. Med Sci Monit 22:2105–2112
Loeser RF, Yammani RR, Carlson CS, Chen H, Cole A, Im HJ, Bursch LS, Yan SD (2005) Articular chondrocytes express the receptor for advanced glycation end products: potential role in osteoarthritis. Arthritis Rheumatol 52(8):2376–2385
Loeser RF (2010) Age-related changes in the musculoskeletal system and the development of osteoarthritis. Clin Geriatr Med 26(3):371–386
Heinola T, Kouri VP, Clarijs P, Ciferska H, Sukura A, Salo J, Konttinen YT (2010) High mobility group box-1 (HMGB-1) in osteoarthritic cartilage. Clin Exp Rheumatol 28(4):511–518
Hamada T, Torikai M, Kuwazuru A, Tanaka M, Horai N, Fukuda T, Yamada S, Nagayama S, Hashiguchi K, Sunahara N, Fukuzaki K, Nagata R, Komiya S, Maruyama I, Fukuda T, Abeyama K (2008) Extracellular high mobility group box chromosomal protein 1 is a coupling factor for hypoxia and inflammation in arthritis. Arthritis Rheumatol 58(9):2675–2685
Ulloa L, Batliwalla FM, Andersson U, Gregersen PK, Tracey KJ (2003) High mobility group box chromosomal protein 1 as a nuclear protein, cytokine, and potential therapeutic target in arthritis. Arthritis Rheumatol 48(4):876–881
Terada C, Yoshida A, Nasu Y, Mori S, Tomono Y, Tanaka M, Takahashi HK, Nishibori M, Ozaki T, Nishida K (2011) Gene expression and localization of high-mobility group box chromosomal protein-1 (HMGB-1)in human osteoarthritic cartilage. Acta Med Okayama 65(6):369–377
Ingels C, Derese I, Wouters PJ, Van den Berghe G, Vanhorebeek I (2015) Soluble RAGE and the RAGE ligands HMGB1 and S100A12 in critical illness: impact of glycemic control with insulin and relation with clinical outcome. Shock 43(2):109–116
Gkogkolou P, Bohm M (2012) Advanced glycation end products: key players in skin aging? Dermatoendocrinology 4(3):259–270
Sunahori K, Yamamura M, Yamana J, Takasugi K, Kawashima M, Yamamoto H, Chazin WJ, Nakatani Y, Yui S, Makino H (2006) The S100A8/A9 heterodimer amplifies proinflammatory cytokine production by macrophages via activation of nuclear factor kappa B and p38 mitogen-activated protein kinase in rheumatoid arthritis. Arthritis Res Ther 8(3):R69
Zreiqat H, Belluoccio D, Smith MM, Wilson R, Rowley LA, Jones K, Ramaswamy Y, Vogl T, Roth J, Bateman JF, Little CB (2010) S100A8 and S100A9 in experimental osteoarthritis. Arthritis Res Ther 12(1):R16
Das SK, Farooqi A (2008) Osteoarthritis. Best Pract Res Clin Rheumatol 22(4):657–675
Goldring MB (2012) Articular cartilage degradation in osteoarthritis. HSS J 8(1):7–9
Harris EC, Coggon D (2015) HIP osteoarthritis and work. Best Pract Res Clin Rheumatol 29(3):462–482
van Lent PL, Blom AB, Schelbergen RF, Sloetjes A, Lafeber FP, Lems WF, Cats H, Vogl T, Roth J, van den Berg WB (2012) Active involvement of alarmins S100A8 and S100A9 in the regulation of synovial activation and joint destruction during mouse and human osteoarthritis. Arthritis Rheumatol 64(5):1466–1476
Liu-Bryan R, Terkeltaub R (2012) The growing array of innate inflammatory ignition switches in osteoarthritis. Arthritis Rheumatol 64(7):2055–2058
Loeser RF, Goldring SR, Scanzello CR, Goldring MB (2012) Osteoarthritis: a disease of the joint as an organ. Arthritis Rheumatol 64(6):1697–1707
Pearle AD, Scanzello CR, George S, Mandl LA, DiCarlo EF, Peterson M, Sculco TP, Crow MK (2007) Elevated high-sensitivity C-reactive protein levels are associated with local inflammatory findings in patients with osteoarthritis. Osteoarthritis Cartil 15(5):516–523
Rosenthal AK (2011) Crystals, inflammation, and osteoarthritis. Curr Opin Rheumatol 23(2):170–173
Schelbergen RF, Blom AB, van den Bosch MH, Slöetjes A, Abdollahi-Roodsaz S, Schreurs BW, Mort JS, Vogl T, Roth J, van den Berg WB, van Lent PL (2012) Alarmins S100A8 and S100A9 elicit a catabolic effect in human osteoarthritic chondrocytes that is dependent on Toll-like receptor 4. Arthritis Rheumatol 64(5):1477–1487
Berenbaum F (2013) Osteoarthritis as an inflammatory disease (osteoarthritis is not osteoarthrosis!). Osteoarthr Cartil 21(1):16–21
Thankam FG, Dilisio MF, Dietz NE, Agrawal DK (2016) TREM-1, HMGB1 and RAGE in the shoulder tendon: dual mechanisms for inflammation based on the coincidence of glenohumeral arthritis. PLoS ONE 11(10):e0165492
Chayanupatkul M, Honsawek S (2010) Soluble receptor for advanced glycation end products (sRAGE) in plasma and synovial fluid is inversely associated with disease severity of knee osteoarthritis. Clin Biochem 43(13–14):1133–1137
Li Y, Wei X, Zhou J, Wei L (2013) The age-related changes in cartilage and osteoarthritis. Biomed Res Int 2013:916530
Li Y, Zhang Y, Chen C, Zhang H, Ma C, Xia Y (2016) Establishment of a rabbit model to study the influence of advanced glycation end products accumulation on osteoarthritis and the protective effect of pioglitazone. Osteoarthr Cartil 24(2):307–314
Donato R, Cannon BR, Sorci G, Riuzzi F, Hsu K, Weber DJ, Geczy CL (2013) Functions of S100 proteins. Curr Mol Med 13(1):24–57
Leclerc E, Fritz G, Vetter SW, Heizmann CW (2009) Binding of S100 proteins to RAGE: an update. Biochim Biophys Acta 1793(6):993–1007
Goyette J, Geczy CL (2011) Inflammation-associated S100 proteins: new mechanisms that regulate function. Amino Acids 41(4):821–842
Perera C, McNeil HP, Geczy CL (2010) S100 calgranulins in inflammatory arthritis. Immunol Cell Biol 88(1):41–49
Subramanian S, Pallati PK, Rai V, Sharma P, Agrawal DK, Nandipati KC (2017) Increased expression of triggering receptor expressed on myeloid cells-1 in the population with obesity and insulin resistance. Obesity 25(3):527–538
Srikrishna G, Freeze HH (2009) Endogenous damage-associated molecular pattern molecules at the crossroads of inflammation and cancer. Neoplasia 11(7):615–628
Sellam J, Berenbaum F (2010) The role of synovitis in pathophysiology and clinical symptoms of osteoarthritis. Nat Rev Rheumatol 6(11):625–635
Scanzello CR, Goldring SR (2012) The role of synovitis in osteoarthritis pathogenesis. Bone 51(2):249–257
Favre J, Erhart-Hledik JC, Chehab EF, Andriacchi TP (2016) Baseline ambulatory knee kinematics are associated with changes in cartilage thickness in osteoarthritic patients over 5 years. J Biomech 49(9):1859–1864
Varady NH, Grodzinsky AJ (2016) Osteoarthritis year in review 2015: mechanics. Osteoarthr Cartil 24(1):27–35
Hubertsson J, Turkiewicz A, Petersson IF, Englund M (2017) Understanding occupation, sick leave and disability pension due to knee and hip osteoarthritis from a gender perspective. Arthritis Care Res 69(2):226–233
Goldring MB, Berenbaum F (2015) Emerging targets in osteoarthritis therapy. Curr Opin Pharmacol 22:51–63
Sun MM, Beier F, Pest MA (2017) Recent developments in emerging therapeutic targets of osteoarthritis. Curr Opin Rheumatol 29(1):96–102
Yang H, Tracey KJ (2010) Targeting HMGB1 in inflammation. Biochim Biophys Acta 1799(1):149–156
Funding
This work was supported by research Grants R01 HL112597, R01 HL116042, and R01 HL120659 to DK Agrawal from the National Heart, Lung and Blood Institute, National Institutes of Health, USA. The content of this review article is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
Conception and design: VR, MFD, and DKA; contributed reagents/materials/analysis tool: MFD, TDS, and DKA; analysis and interpretation of the data: JHR and VR; drafting of the manuscript: JHR, VR, MFD, TDS, and DKA; critical revision of the article for important intellectual content: VR, MFD, and DKA; final approval of the article: JHR, VR, MFD, TDS, and DKA.
Corresponding author
Ethics declarations
Conflict of interest
As the corresponding author, I declare that this manuscript is original, that the article does not infringe upon any copyright or other proprietary rights of any third party, and that neither the text nor the data have been reported or published previously. All the authors have no conflict of interest and have read the journal’s authorship statement.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rosenberg, J.H., Rai, V., Dilisio, M.F. et al. Increased expression of damage-associated molecular patterns (DAMPs) in osteoarthritis of human knee joint compared to hip joint. Mol Cell Biochem 436, 59–69 (2017). https://doi.org/10.1007/s11010-017-3078-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-017-3078-x