Abstract
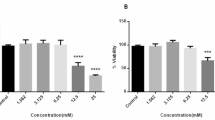
MMP9 is a member of the family of zinc-containing endopeptidases which degrade various components of the extracellular matrix, thereby regulating matrix remodeling. Since matrix remodeling plays an important role during growth and progression of cancer and considering the fact that, tumor cells switch to aerobic glycolysis as its major energy source, this study was designed to analyze if partial inhibition of glycolysis (the major energy pathway during hypoxia) can be used as a means to control matrix remodeling in terms of MMP9 activity and expression. For this, human epithelial carcinoma cells were treated with glycolytic inhibitor, 2-deoxy glucose (2DG) at sub-lethal concentrations followed by analysis of the expression and activity of MMP2 and MMP9. The experimental findings demonstrate that exposure of cancer cells to glycolytic inhibitor at concentration that does not induce ER stress, downregulates the activity and expression of MMP9 without affecting the expression levels and activity of MMP2. Further mechanistic analysis revealed that the regulation of MMP9 was mediated in a SIRT-1 dependent mechanism and did not alter the NFkB signaling pathway. The overall results presented here, therefore suggest that the use of glycolytic inhibitor, 2DG at concentration that do not affect cell viability or induce ER stress can be an effective strategy to control matrix remodeling.





Similar content being viewed by others
Abbreviations
- 2DG:
-
2-Deoxy glucose
- MMP9:
-
Matrix metalloprotease 9
- SIRT-1:
-
Sirtuin-1
- NFkB:
-
Nuclear factor kB
- ER:
-
Endoplasmic reticulum
- ELISA:
-
Enzyme-linked immunosorbent assay
- SDS-PAGE:
-
Sodium dodecyl sulphate Poly acrylamide gel electrophoresis
- NAD+ :
-
Nicotinamide adenine dinucleotide
- qPCR:
-
Quantitative real-time PCR
References
Kiefer JA, Farach Carson MC (2001) Type I collagen-mediated proliferation of PC3 prostate carcinoma cell line: implications for enhanced growth in the bone microenvironment. Matrix Biol 20:429–437
Bossman FT, Stamenkovic I (2003) Functional structure and composition of extracellular matrix. J Pathol 200:423–428
Paszek MJ, Weaver VM (2004) The tension mounts: mechanics meets morphogenesis and malignancy. J Mammary Gland Biol Neoplasia 9:325–342
Kass L, Erler JT, Dembo M, Weaver VM (2007) Mammary epithelial cell: influence of extracellular matrix composition and organization during development and tumorigenesis. Int J Biochem Cell Biol 39:1987–1994
Wynn TA (2007) Common and unique mechanisms regulate fibrosis in various fibro proliferative diseases. J Clin Invest 117:524–529
Butcher DT, Alliston T, Weaver VMA (2009) Tense situation: forcing tumour progression. Nat Rev Cancer 9:108–122
Strongin AY (2006) Mislocalization and unconventional functions of cellular MMPs in cancer. Cancer Metastasis Rev 25:87–98
Rajeshwar P Verma, Hansch Corwin (2007) Matrix metalloproteinases (MMPs): chemical–biologicalfunctions and (Q) SARs. Bioorg Med Chem 15:2223–2268
Nelson AR, Fingleton B, Rothenberg ML, Matrisian LM (2000) Matrix metalloproteinases: biologic activity and clinical implications. J Clin Oncol 18:1135–1149
Bode W, Maskos K (2003) Structural basis of the matrix metalloproteinases and their physiological inhibitors, the tissue inhibitors of metalloproteinases. J Biol Chem 384:863–872
Lopez Otin, Pendas AM, Knauper V, Puente XS, Llano E, Mattei MG, Apte S, Murphy G (1997) Identification and characterization of a novel human matrix metalloproteinase with unique structural characteristics, chromosomal location, and tissue distribution. J Biol Chem 272:4281–4286
Parks WC, Shapir SD (2001) Matrix metalloproteinases in lung biology. Respir Res 2:10–19
Ambili M, Sudhakaran PR (1998) Assay of matrix metalloproteinases in substrate impregnated gels in multiwells. Ind J Biochem Biophys 35(5):317–320
Engvall E, Perlmann P (1971) Enzyme linked immunosorbent assay (ELISA). Quantitative assay of immunoglobulin G. Immunochemistry 8:871–874
Chomczynski P, Mackey K (1995) Short technical reports. Modification of the TRI reagent procedure for isolation of RNA from polysaccharide- and proteoglycan-rich sources. Biotechniques 19(6):942–945
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Gao Z, Ye J (2008) Inhibion of transcriptional activity of c-JUN by SIRT-1. Biochem Biophys Res Commun 376:793–796
Ozbek S, Balasubramanian PG, Chiquet Ehrismann R, Tucker RP, Adams JC (2010) The evolution of extracellular matrix. Mol Biol Cell 21:4300–4305
Zhang XD, Deslandes E, Villedieu M, Poulain L, Duval M, Gauduchon P, Schwartz L, Icard P (2006) Effect of 2-deoxy-d-glucose on various malignant cell lines in vitro. Anticancer Res 26(5A):3561–3566
Aft RL, Zhang TW, Gius D (2002) Evaluation of 2DG as a chemotherapeutic agent:mechanism of celldeath. Br J Cancer 87:805–812
Haibin Xi, Kurtoglu Metin, Theodore J (2011) 2-deoxy-D- glucose activates autophagy via endoplasmic reticulam stress rather than ATP depletion. Cancer Chemother Pharmacol 67:899–910
Bonnans Caroline, Chou Jonathan, Werb Zena (2014) Remodelling the extracellular matrix in development and disease. Nat Rev Mol Cell Biol 15:786–801
Kessenbrock Kai, Plaks Vicki, Werb Zena (2010) Matrix metalloproteinases: regulators of the tumor microenvironment. Cell 141:52–67
Kim KA, Ching SB, Hawng EY, Noh SH, Song KH, Kim HH, Kim CH, Park YG (2013) J Periodontal implant Sci 43:123–127
Chou YC, Sheu JR, Chung CL, Chen CY, Lin FL, Hsu MJ, Kuo YH, Hasiao G (2010) Nuclear targeter inhibition of NFkB on MMP9 production by N-2(4-bromophenyl) ethyl caffaemide in human monocytic cells. Chem Biol Interact 184:403–412
Pascucci Barbara, Lemma Tiziana, Iorio Egidio, Giovannini Sara, Iavarone Ivano, Calcagnile Angelo, Narciso Laura (2012) An altered redox balance mediates the hypersensitivity of Cockayne syndrome primary fibroblasts to oxidative stress. Aging Cell 11:520–529
Kunhiraman Haritha, Edatt Lincy, Thekkeveedu Sruthi, Poyyakkara Aswini, Raveendran Viji, Kiran Manikantan Syamala, Sudhakaran Perumana, Kumar Sameer VB (2016) 2-Deoxy Glucose modulates expression and biological activity of VEGF in a SIRT-1 dependent mechanism. J Cell Biochem 9999:1–11
Yang T, Sauve AA (2006) NAD metabolism and sirtuins: metabolic regulation of protein deacetylation in stress and toxicity. AAPS J8:E632–E643
Hwang JW, Yao H, Caito S, Sundar IK, Rahman I (2013) Redox regulation of SIRT1 in inflammation and cellular senescence. Free Radic Biol Med 61C:95–110
Nakamaru Yuji, Vuppusetty Chaitanya, Wada Hiroo, Milne Jill C, Ito Misako, Rossios Christos, Elliot Mark, Hogg James, Kharitonov Sergei, Goto Hajime, Bemis Jean E, Elliott Peter, Barnes Peter J, Ito Kazuhiro (2009) A protein deacetylase SIRT1 is a negative regulator metalloproteinase-9. FASEB J 23:2810–2819
Acknowledgments
Financial assistance received from DST-SERB and KSCSTE is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Edatt, L., Haritha, K., Sruthi, T.V. et al. 2-Deoxy glucose regulate MMP-9 in a SIRT-1 dependent and NFkB independent mechanism. Mol Cell Biochem 423, 197–206 (2016). https://doi.org/10.1007/s11010-016-2837-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-016-2837-4