Abstract
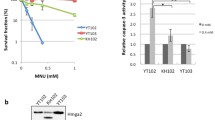
The mammalian DNA mismatch repair (MMR) system consists of a number of proteins that play important roles in repair of base pair mismatch mutations and in maintenance of genomic integrity. A defect in this system can cause genetic instability, which can lead to carcinogenesis. For instance, a germline mutation in one of the mismatch repair proteins, especially MLH1 or MSH2, is responsible for hereditary non-polyposis colorectal cancer. These MMR proteins also play an important role in the induction of apoptosis. Accordingly, altered expression of or a defect in MLH1 or MSH2 may confer resistance to anti-cancer drugs used in chemotherapy. We hypothesized that the ability of these two MMR proteins to regulate apoptosis are interdependent. Moreover, a defect in either one may confer resistance to chemotherapy by an inability to trigger apoptosis. To this end, we studied three cell lines—SW480, LoVo, and HTC116. These cell lines were selected based on their differential expression of MLH1 and MSH2 proteins. SW480 expresses both MLH1 and MSH2; LoVo expresses only MLH1 but not MSH2; HCT116 expresses only MSH2 but not MLH1 protein. MTT assays, a measure of cytotoxicity, showed that there were different cytotoxic effects of an anti-cancer drug, etoposide, on these cell lines, effects that were correlated with the MMR status of the cells. Cells that are deficient in MLH1 protein (HCT116 cells) were resistant to the drug. Cells that express both MLH1 and MSH2 proteins (SW480 cells) showed caspase-3 cleavage, an indicator of apoptosis. Cells that lack MLH1 (HCT116 cells) did not show any caspase-3 cleavage. Expression of full-length MLH1 protein was decreased in MMR proficient (SW480) cells during apoptosis; it remained unchanged in cells that lack MSH2 (LoVo cells). The expression of MSH2 protein remained unchanged during apoptosis both in MMR proficient (SW480) and deficient (HCT116) cells. Studies on translocation of MLH1 protein from nucleus to cytosolic fraction, an indicator of apoptosis, showed that MLH1 translocation only occurred in MMR proficient (SW480) cells upon induction of apoptosis further suggested a MSH2 dependent role of MLH1 in apoptosis. These data suggest a role of MLH1 in mediation of apoptosis in a MSH2-dependent manner. Taken together, our data supported an interdependence of mismatch repair proteins, particularly MLH1 and MSH2, in the mediation of apoptosis in human colorectal carcinoma cell lines.









Similar content being viewed by others
Abbreviations
- HNPCC:
-
Hereditary non-polyposis colorectal cancer
- MMR:
-
Mismatch repair
- Caspases:
-
Cysteine-aspartic proteases
- MLH1:
-
MutL
- MSH2:
-
MutS
References
Meyers M, Wagner MW, Hwang H-S, Kinsella TJ, Boothman DA (2001) Role of the hMLH1 DNA mismatch repair protein in fluoropyrimidine-mediated cell death and cell cycle responses. Cancer Res 61:5193–5201
Modrich P (1997) Strand-specific mismatch repair in mammalian cells. J Biol Chem 272:24727–24730
Peterson-Roth E, Reynolds M, Quievryn G, Zhitkovich A (2005) Mismatch repair proteins are activators of toxic responses to chromium-DNA damage. Mol Cell Biol 25:3596–3607
Shimodaira H, Yoshioka-Yamashita A, Kolodner RD, Wang JY (2002) Interaction of mismatch repair protein PMS2 and the p53-related transcription factor p73 in apoptosis response to cisplatin. PNAS 100:2420–2425
Narayan S, Roy D (2003) Role of APC and DNA mismatch repair genes in the development of colorectal cancers. Mol Cancer 2:1–15
Hickman MJ, Samson LD (1999) Role of DNA mismatch repair and p53 in signaling induction of apoptosis by alkylating agents. Proc Natl Acad Sci 96:10764–10769
Reed JC (1999) Dysregulation of apoptosis in cancer. J Clin Oncol 17:2941–2953
Haupt S, Berger M, Goldberg Z, Haupt Y (2003) Apoptosis: the p53 network. J Cell Sci 116:4077–4085
Bernstein C, Bernstein H, Payne CM, Garewal H (2002) DNA repair/pro-apoptotic dual-role proteins in five major DNArepair pathways: fail-safe protection against carcinogenesis. Mutat Res 511:145–178
Zhang H, Richards B, Wilson T, Lloyd M, Cranston A, Thorburn A, Fishel R, Meuth M (1999) Apoptosis Induced by Overexpression of hMSH2 or hMLH1. Cancer Res 59:3021–3027
Yanamadala S, Ljungman M (2003) Potential Role of MLH1 in the induction of p53 and apoptosis by blocking transcription on damaged DNA templates. Mol Cancer Res 1:747–754
Mihaylova VT, Bindra RS, Yuan J, Campisi D, Narayanan L, Jensen R, Giordano F, Johnson RS, Rockwell S, Glazer PM (2003) Decreased expression of the DNA mismatch repair gene Mlh1 under hypoxic stress in mammalian cells. Mol Cell Biol 23:3265–3273
Qi W, Vasquez KM (2008) Human MLH1 protein participates in genomic damage checkpoint signaling in response to DNA interstrand crosslinks, while MSH2 functions in DNA repair. PLoS Genet 4:1–10
Yan T, Seo Y, Kinsella TJ (2009) Differential cellular responses to prolonged low dose rate ionizing radiation in MLH1-proficient and -deficient colorectal cancer HCT116 cells. Clin Cancer Res 15:6912–6920
Jacob S, Aguado M, Fallik D, Praz F (2001) The role of the DNA mismatch repair system in the cytotoxicity of thetopoisomerase inhibitors camptothecin and etoposide to human colorectal cancer cells. Cancer Res 61:6555–6562
Hassen S, Boman BM, Ali N, Parker M, Somerman C, Ali-Khan ZJ, Catts AA, Ali AA, Fields JZ (2011) Detection of DNA mismatch repair proteins in fresh human blood lymphocytes: towards a novel method for hereditary non-polyposis colorectal cancer (Lynch syndrome) screening. J Exp Clin Cancer Res 30:1–7
Agarwal R, Mumtaz H, Ali N (2009) Role of inositol polyphosphates in programmed cell death. Mol Cell Biochem 328:155–165
Agarwal R, Hassen S, Ali N (2010) Changes in cellular levels of inositol polyphosphates during apoptosis. Mol Cell Biochem 345:61–68
Jiang S, Storrie B (2005) Cisternal rab proteins regulate Golgi apparatus redistribution in response to hypotonic stress. Mol Biol Cell 16:2586–2596
Ramirez CD, Sleiman RJ, Catchpoole DR, Stewart BW (2000) Morphological and molecular evidence of differentiation during etoposide-induced apoptosis in human lymphoblastoid cells. Cell Death Differ 7:548–555
Chen F, Arseven OK, Cryns VL (2004) Proteolysis of the mismatch repair protein MLH1 by caspase-3 promotes DNA damage-induced apoptosis. J Biol Chem 279:27542–27548
Papouli E, Cejka P, Jiricny J (2004) Dependence of the cytotoxicity of DNA-damaging agents on the mismatch repair status of human cells. Cancer Res 64:3391–3394
Aebi S, Fink D, Gordon R et al (1997) Resistance to cytotoxic drugs in DNA mismatch repair-deficient cells. Clin Cancer Res 3:763–1767
Acknowledgments
This study was supported in part by Kathleen Thomsen-Hall Charitable Foundation, NSF-EPSCoR P3 center and NASA-EPSCoR Research Infrastructure Development (RID) funds to Dr. Nawab Ali. Samar Hassen is grateful to the Department of Applied Science, College of Science, University of Arkansas at Little Rock, for a research assistantship. Support was also provided by an NIH Phase II Grant (R44 CA 090122) awarded to Dr Fields and a Phase IIB SBIR/STTR Grant from the Delaware Strategic Fund awarded by the Delaware Economic Development Office to Dr Boman.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hassen, S., Ali, A.A., Kilaparty, S.P. et al. Interdependence of DNA mismatch repair proteins MLH1 and MSH2 in apoptosis in human colorectal carcinoma cell lines. Mol Cell Biochem 412, 297–305 (2016). https://doi.org/10.1007/s11010-015-2636-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-015-2636-3