Abstract
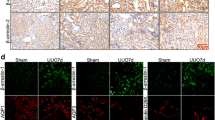
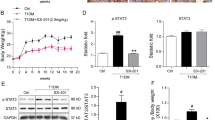
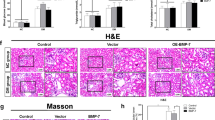
The present study aimed to investigate the relationship between wnt/β-catenin signaling pathway and kidney impairment in diabetic nephropathy (DN) mice as well as the renoprotective effect of rhein (RH). Mice were randomly divided into four groups (n = 6): db/db mice treated with RH (DN + RH), db/db mice (DN), db/m mice treated with RH (NC + RH) and db/m mice (NC). RH-treated groups were administered orally at a daily dose 120 mg/kg. Mice were sacrificed after 12 weeks of treatments. In our study, increased albuminuria, together with weight gain and hyperglycemia was observed in the beginning of the study and continued to increase throughout the length of the study (12 weeks). Histopathologic changes were observed in the DN group. Expectedly, mice receiving the treatment with RH were protected from this injury. Meanwhile, the expression of nephrin, a podocyte-specific marker, was significantly reduced while wnt1, p-GSK-3β/tGSK-3β, p-β-catenin/tβ-catenin were higher in the DN group mice when analyzed by immunofluorescence and Western blotting. RH reversed these above changes. wnt/β-catenin signaling pathway participates in RH ameliorating kidney injury in DN mice. The manipulation of RH might act as a promising therapeutic intervention for DN.







Similar content being viewed by others
Abbreviations
- DN:
-
Diabetic nephropathy
- NC:
-
Normal control
- ESRD:
-
End-stage renal disease
References
Badal SS, Danesh FR (2014) New insights into molecular mechanisms of diabetic kidney disease. Am J Kidney Dis 63:S63–S83
Reidy K, Kang HM, Hostetter T, Susztak K (2014) Molecular mechanisms of diabetic kidney disease. J Clin Invest 124:2333–2340
Dai C, Stolz DB, Kiss LP, Monga SP, Holzman LB, Liu Y (2009) wnt/beta-catenin signaling promotes podocyte dysfunction and albuminuria. J Am Soc Nephrol 20:1997–2008
Heikkila E, Juhila J, Lassila M, Messing M, Perala N, Lehtonen E, Lehtonen S, Sjef VJ, Holthofer H (2010) Beta-catenin mediates adriamycin-induced albuminuria and podocyte injury in adult mouse kidneys. Nephrol Dial Transplant 25:2437–2446
Iglesias DM, Hueber PA, Chu L, Campbell R, Patenaude AM, Dziarmaga AJ, Quinlan J, Mohamed O, Dufort D, Goodyer PR (2007) Canonical wnt signaling during kidney development. Am J Physiol Renal Physiol 293:F494–F500
Saleem MA, O’Hare MJ, Reiser J, Coward RJ, Inward CD, Farren T, Xing CY, Ni L, Mathieson PW, Mundel P (2002) A conditionally immortalized human podocyte cell line demonstrating nephrin and podocin expression. J Am Soc Nephrol 13:630–638
Peng L, Yang J, Ning C, Zhang J, Xiao X, He D, Wang X, Li Z, Fu S, Ning J (2012) Rhein inhibits integrin-linked kinase expression and regulates matrix metalloproteinase-9/tissue inhibitor of metalloproteinase-1 ratio in high glucose-induced epithelial-mesenchymal transition of renal tubular cell. Biol Pharm Bull 35:1676–1685
Hummel KP, Dickie MM, Coleman DL (1966) Diabetes, a new mutation in the mouse. Science 153:1127–1128
Zeng CC, Liu X, Chen GR, Wu QJ, Liu WW, Luo HY, Cheng JG (2014) The molecular mechanism of rhein in diabetic nephropathy. Evid Based Complement Altern Med 2014:487
Mifsud SA, Allen TJ, Bertram JF, Hulthen UL, Kelly DJ, Cooper ME, Wilkinson-Berka JL, Gilbert RE (2001) Podocyte foot process broadening in experimental diabetic nephropathy: amelioration with renin-angiotensin blockade. Diabetologia 44:878–882
Li CX, Xia M, Han WQ, Li XX, Zhang C, Boini KM, Liu XC, Li PL (2011) Reversal by growth hormone of homocysteine-induced epithelial-to-mesenchymal transition through membrane raft-redox signaling in podocytes. Cell Physiol Biochem 27:691–702
Riediger F, Quack I, Qadri F, Hartleben B, Park JK, Potthoff SA, Sohn D, Sihn G, Rousselle A, Fokuhl V, Maschke U, Purfürst B, Schneider W, Rump LC, Luft FC, Dechend R, Bader M, Huber TB, Nguyen G, Muller DN (2011) Prorenin receptor is essential for podocyte autophagy and survival. J Am Soc Nephrol 22:2193–2202
Benigni A, Gagliardini E, Tomasoni S, Abbate M, Ruggenenti P, Kalluri R, Remuzzi G (2004) Selective impairment of gene expression and assembly of nephrin in human diabetic nephropathy. Kidney Int 65:2193–2200
MacDonald BT, Tamai K, He X (2009) wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell 17:9–26
Naves MA, Requiao-Moura LR, Soares MF, Silva-Junior JA, Mastroianni-Kirsztajn G, Teixeira VP (2012) Podocyte wnt/β-catenin pathway is activated by integrin-linked kinase in clinical and experimental focal segmental glomerulosclerosis. J Nephrol 25:401–409
Jope RS, Johnson GV (2004) The glamour and gloom of glycogen synthase kinase-3. Trends Biochem Sci 29:95–102
Patel S, Doble B, Woodgett JR (2004) Glycogen synthase kinase-3 in insulin and wnt signalling: a double-edged sword? Biochem Soc Trans 32:803–808
Kim K, Lu Z, Hay ED (2002) Direct evidence for a role of beta-catenin/LEF-1 signaling pathway in induction of EMT. Cell Biol Int 26:463–476
Sarafidis PA, Bakris GL (2006) Protection of the kidney by thiazolidinediones: an assessment from bench to bedside. Kidney Int 70:1223–1233
Fornoni A (2010) Proteinuria, the podocyte, and insulin resistance. N Engl J Med 363:2068–2069
Mao J, Wang J, Liu B, Pan W, Farr GH 3rd, Flynn CY, Yuan H, Takada S, Kimelman D, Li L, Wu D (2001) Low-density lipoprotein receptor-related protein-5 binds to Axin and regulates the canonical wnt signaling pathway. Mol Cell 7:801–809
Fukumoto S, Hsieh CM, Maemura K, Layne MD, Yet SF, Lee KH, Matsui T, Rosenzweig A, Taylor WG, Rubin JS, Perrella MA, Lee ME (2001) Akt participation in the wnt signaling pathway through dishevelled. J Biol Chem 276:17479–17483
Zhou BP, Deng J, Xia W, Xu J, Li YM, Gunduz M, Hung MC (2004) Dual regulation of snail by GSK-3beta-mediated phosphorylation in control of epithelial-mesenchymal transition. Nat Cell Biol 6:931–940
Zhao X, Xu K, Shi H, Cheng J, Ma J, Gao Y, Li Q, Ye X, Lu Y, Yu X, Du J, Du W, Ye Q, Zhou L (2014) Application of the back-error propagation artificial neural network (BPANN) on genetic variants in the PPAR-γ and RXR-α gene and risk of metabolic syndrome in a Chinese Han population. J Biomed Res 28(2):114–122
Jia Z, Sun Y, Yang G, Zhang A, Huang S, Heiney KM, Zhang Y (2014) New insights into the PPAR γ agonists for the treatment of diabetic nephropathy. PPAR Res 2014:818530
Acknowledgments
We thank Dr. Zhihong Liu in Jinling Hospital for providing rhein in this study, and the Priority Academic Program Development (PAPD) of Jiangsu Higher Education Institutions. This work was supported by grants from the National Natural Science Foundation of China (81170660/H0509,81370815/H0509, to Changying Xing; 81100512/H0510, to Bo Zhang; 81300573, to Yuanyang Gang), the Ph.D. Programs Foundation of the Ministry of Education of China (2011BAI10B06, 2011BAI10B08, to Changying Xing), the Natural Science Foundation of Jiangsu (LJ201125, BK20151588, BL2012032, BL2014080, to Changying Xing; BK20131030, to Yuanyang Gang).
Authors Contribution
Suyan Duan designed the study and drafted the paper. Bo Zhang was involved in revising the paper. Suyan Duan, Yingyi Wu, Chuanyan Zhao and Yuming Chen carried out the full experiment. Yanggang Yuan and Changying Xing were responsible for study oversight, data collection, analysis. All authors have read the final paper and approved the submission.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
No conflicts of interest.
Rights and permissions
About this article
Cite this article
Duan, S., Wu, Y., Zhao, C. et al. The wnt/β-catenin signaling pathway participates in rhein ameliorating kidney injury in DN mice. Mol Cell Biochem 411, 73–82 (2016). https://doi.org/10.1007/s11010-015-2569-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-015-2569-x