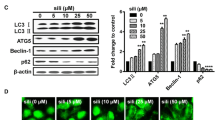
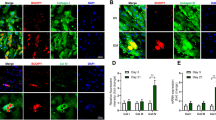
Abstract
The process of preadipocytes differentiation plays a vital role in adipose tissue expansion and many factors are involved in this event. Cathepsin B (CTSB), secreted from lysosome, has been reported in regulating a variety of physiological processes. In this study, we demonstrated CTSB promotes lipid accumulation and adipogenic genes expression in porcine primary preadipocytes by degrading fibronectin (Fn), a key component of extracellular matrix. Lithium chloride (LiCl) is an activator of Wnt/β-catenin signaling through stabilizing β-catenin. We found that CTSB can relieve the anti-adipogenic effects of LiCl, indicating that CTSB could impact Wnt/β-catenin signaling pathway. Interestingly, Fn is an important target gene of Wnt/β-catenin. So we considered that CTSB promote preadipocytes differentiation by suppressing these two pathways.






Similar content being viewed by others
References
Bulcao C, Ferreira SR, Giuffrida F, Ribeiro-Filho FF (2006) The new adipose tissue and adipocytokines. Curr Diabetes Rev 2:19–28
Gandhi H, Upaganlawar A, Balaraman R (2010) Adipocytokines: the pied pipers. J Pharmacol Pharmacother 1:9–17
Krysiak R, Okopień B, Herman ZS (2005) Adipose tissue: a new endocrine organ. Przegl Lek 62:919–923
Chun T-H (2012) Peri-adipocyte ECM remodeling in obesity and adipose tissue fibrosis. Adipocyte 1:89–95
Hirsch J, Batchelor B (1976) Adipose tissue cellularity in human obesity. Clin Endocrinol Metab 5:299–311
Gregor MF, Hotamisligil GS (2011) Inflammatory mechanisms in obesity. Annu Rev Immunol 29:415–445
Lumeng CN, Saltiel AR (2011) Inflammatory links between obesity and metabolic disease. J Clin Invest 121:2111
Schenk S, Saberi M, Olefsky JM (2008) Insulin sensitivity: modulation by nutrients and inflammation. J Clin Invest 118:2992–3002
Nakajima I, Muroya S, Tanabe R-I, Chikuni K (2002) Extracellular matrix development during differentiation into adipocytes with a unique increase in type V and VI collagen. Biol Cell 94:197–203
Antras J, Hilliou F, Redziniak G, Pairault J (1989) Decreased biosynthesis of actin and cellular fibronectin during adipose conversion of 3T3-F442A cells. Reorganization of the cytoarchitecture and extracellular matrix fibronectin. Biol Cell 66:247–254
Rodríguez Fernández JL, Ben-Ze’ev A (1989) Regulation of fibronectin, integrin and cytoskeleton expression in differentiating adipocytes: inhibition by extracellular matrix and polylysine. Differentiation 42:65–74
Spiegelman BM, Ginty CA (1983) Fibronectin modulation of cell shape and lipogenic gene expression in 3T3-adipocytes. Cell 35:657–666
Brix K, Dunkhorst A, Mayer K, Jordans S (2008) Cysteine cathepsins: cellular roadmap to different functions. Biochimie 90:194–207
Conus S, Simon H-U (2008) Cathepsins: key modulators of cell death and inflammatory responses. Biochem Pharmacol 76:1374–1382
Tardy C, Codogno P, Autefage H et al (2006) Lysosomes and lysosomal proteins in cancer cell death (new players of an old struggle). Biochim Biophys Acta 1765:101–125
Zava\vsnik-Bergant T, Turk B (2007) Cysteine proteases: destruction ability versus immunomodulation capacity in immune cells. Biol Chem 388:1141–1149
Chavey C, Mari B, Monthouel M-N et al (2003) Matrix metalloproteinases are differentially expressed in adipose tissue during obesity and modulate adipocyte differentiation. J Biol Chem 278:11888–11896
Taleb S, Lacasa D, Bastard J-P et al (2005) Cathepsin S, a novel biomarker of adiposity: relevance to atherogenesis. FASEB J 19:1540–1542
Taleb S, Cancello R, Clément K, Lacasa D (2006) Cathepsin s promotes human preadipocyte differentiation: possible involvement of fibronectin degradation. Endocrinology 147:4950–4959
Masson O, Prébois C, Derocq D et al (2011) Cathepsin-D, a key protease in breast cancer, is up-regulated in obese mouse and human adipose tissue, and controls adipogenesis. PLoS One 6:e16452
Chiellini C, Costa M, Novelli SE et al (2003) Identification of cathepsin K as a novel marker of adiposity in white adipose tissue. J Cell Physiol 195:309–321
Funicello M, Novelli M, Ragni M et al (2007) Cathepsin K null mice show reduced adiposity during the rapid accumulation of fat stores. PLoS One 2:e683
Withana NP, Blum G, Sameni M et al (2012) Cathepsin B inhibition limits bone metastasis in breast cancer. Cancer Res 72:1199–1209
Cavallo-Medved D, Rudy D, Blum G et al (2009) Live-cell imaging demonstrates extracellular matrix degradation in association with active cathepsin B in caveolae of endothelial cells during tube formation. Exp Cell Res 315:1234–1246
Wong WP, Altemus JB, Hester JF et al (2013) Cathepsin B is a novel gender-dependent determinant of cholesterol absorption from the intestine. J Lipid Res 54:816–822
Morchang A, Panaampon J, Suttitheptumrong A et al (2013) Role of cathepsin B in dengue virus-mediated apoptosis. Biochem Biophys Res Commun 438:20–25
Tatti M, Motta M, Di Bartolomeo S et al (2013) Cathepsin-mediated regulation of autophagy in saposin C deficiency. Autophagy 9:241–243
Freese JL, Pino D, Pleasure SJ (2010) Wnt signaling in development and disease. Neurobiol Dis 38:148–153
Lefterova MI, Lazar MA (2009) New developments in adipogenesis. Trends Endocrinol Metab 20:107–114
Rosen ED, MacDougald OA (2006) Adipocyte differentiation from the inside out. Nat Rev Mol Cell Biol 7:885–896
He XC, Zhang J, Tong W-G et al (2004) BMP signaling inhibits intestinal stem cell self-renewal through suppression of Wnt–β-catenin signaling. Nat Genet 36:1117–1121
Logan CY, Nusse R (2004) The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol 20:781–810
Luo J, Deng Z-L, Luo X et al (2007) A protocol for rapid generation of recombinant adenoviruses using the AdEasy system. Nat Protoc 2:1236–1247
De Langhe SP, Sala FG, Del Moral P-M et al (2005) Dickkopf-1 (DKK1) reveals that fibronectin is a major target of Wnt signaling in branching morphogenesis of the mouse embryonic lung. Dev Biol 277:316–331
Ten Berge D, Koole W, Fuerer C et al (2008) Wnt signaling mediates self-organization and axis formation in embryoid bodies. Cell Stem Cell 3:508–518
Eguchi A, Feldstein AE (2013) Lysosomal Cathepsin D contributes to cell death during adipocyte hypertrophy. Adipocyte 2:0–1
Naour N, Rouault C, Fellahi S et al (2010) Cathepsins in human obesity: changes in energy balance predominantly affect cathepsin s in adipose tissue and in circulation. J Clin Endocrinol Metab 95:1861–1868
Xiao Y, Junfeng H, Tianhong L et al (2006) Cathepsin K in adipocyte differentiation and its potential role in the pathogenesis of obesity. J Clin Endocrinol Metab 91:4520–4527
Yang M, Zhang Y, Pan J et al (2007) Cathepsin L activity controls adipogenesis and glucose tolerance. Nat Cell Biol 9:970–977
Li S, Du L, Zhang L et al (2013) Cathepsin B contributes to autophagy-related 7 (Atg7)-induced nod-like receptor 3 (NLRP3)-dependent proinflammatory response and aggravates lipotoxicity in rat insulinoma cell line. J Biol Chem 288:30094–30104
Santoni M, Amantini C, Morelli MB, et al (2013) Effect of sunitinib and pazopanib on necrosis and autophagic cell death in cancer cells: role of cathepsin B. J Clin Oncol. Abstr. p e15513
Li XJ, Yang H, Li GX et al (2012) Transcriptome profile analysis of porcine adipose tissue by high-throughput sequencing. Anim Genet 43:144–152
Gregoire FM, Smas CM, Sul HS (1998) Understanding adipocyte differentiation. Physiol Rev 78:783–809
Antras-Ferry J, Hilliou F, Lasnier F, Pairault J (1994) Forskolin induces the reorganization of extracellular matrix fibronectin and cytoarchitecture in 3T3-F442A adipocytes: its effect on fibronectin gene expression. Biochim Biophys Acta 1222:390–394
Bortell R, Owen TA, Ignotz R et al (1994) TGFβ1 prevents the down-regulation of type I procollagen, fibronectin, and TGFβ1 gene expression associated with 3T3-L1 pre-adipocyte differentiation. J Cell Biochem 54:256–263
Weiner FR, Shah A, Smith PJ et al (1989) Regulation of collagen gene expression in 3T3-L1 cells. Effects of adipocyte differentiation and tumor necrosis factor alpha. Biochemistry 28:4094–4099
Ross SE, Hemati N, Longo KA et al (2000) Inhibition of adipogenesis by Wnt signaling. Science 289:950–953
Vertino AM, Taylor-Jones JM, Longo KA et al (2005) Wnt10b deficiency promotes coexpression of myogenic and adipogenic programs in myoblasts. Mol Biol Cell 16:2039–2048
Vangipuram SD, Sheele J, Atkinson RL et al (2004) A human adenovirus enhances preadipocyte differentiation. Obes Res 12:770–777
Wang Y, Zhao L, Smas C, Sul HS (2010) Pref-1 interacts with fibronectin to inhibit adipocyte differentiation. Mol Cell Biol 30:3480–3492
Acknowledgments
This work was funded by grants from the National Natural Science Foundation of China (No. 31272410), the National Basic Research Program of China (No. 2012CB124705), the Program for Innovative Research Team in the Northwest A&F University, and the National Swine Industry Technology System (No. CARS-36-04B).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, ZY., Mai, Y., Yang, H. et al. CTSB promotes porcine preadipocytes differentiation by degrading fibronectin and attenuating the Wnt/β-catenin signaling pathway. Mol Cell Biochem 395, 53–64 (2014). https://doi.org/10.1007/s11010-014-2111-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-014-2111-6