Abstract
Cytochrome P4502E1 (CYP2E1), glutathione-S-transferase A4-4 (GSTA4-4), and inducible nitric oxide synthase (iNOS) are implicated in maneb- and paraquat-induced toxicity leading to various pathological conditions. The study aimed to investigate the role of CYP2E1 in maneb- and paraquat-induced oxidative stress in rat polymorphonuclear leukocytes (PMNs) and its crosstalk with iNOS-mediated nitrosative stress and GSTA4-4-linked protective effect, if any and their consequent links with the nuclear factor erythoid 2-related factor 2 (Nrf2) activation and heme oxygenase-1 (HO-1) expression. Rats were treated with/without maneb and/or paraquat for 1, 2, and 3 weeks along with vehicle controls. Subsets of rats were also treated with diallyl sulfide (DAS) or aminoguanidine (AG) along with the respective controls. Maneb and paraquat augmented the reactive oxygen species (ROS), lipid peroxidation (LPO) and 4-hydroxy nonenal (4-HNE) contents, and superoxide dismutase (SOD) activity in the PMNs. However, maneb and paraquat attenuated the reduced glutathione (GSH) level and the expression/activity of total GST and GST-pi. Maneb and paraquat increased the expression/activity of CYP2E1, GSTA4-4, iNOS, Nrf2 and HO-1, and nitrite content. CYP2E1 inhibitor, DAS noticeably alleviated maneb- and paraquat-induced ROS, LPO, 4-HNE, SOD, Nrf2 and HO-1, GST, GSH, and GST-pi while iNOS, nitrite content and GSTA4-4 levels were unchanged. Conversely, AG, an iNOS inhibitor, attenuated maneb- and paraquat-directed changes in nitrite, LPO, iNOS but it did not alter ROS, GSH, SOD, GST, GST-pi, Nrf2, HO-1, CYP2E1, and GSTA4-4. The results demonstrate that CYP2E1 induces iNOS-independent free radical generation and subsequently modulates the Nrf2-dependent HO-1 and 4-HNE-mediated GST expression in maneb- and paraquat-treated PMNs.







Similar content being viewed by others
Change history
19 July 2019
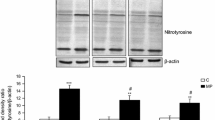
In the original publication of the article, the wrong ��-actin blots were pasted in Figs. 1b and 2c. The correct versions of Figs. 1b and 2c are given in this correction.
19 July 2019
In the original publication of the article, the wrong ��-actin blots were pasted in Figs. 1b and 2c. The correct versions of Figs. 1b and 2c are given in this correction.
19 July 2019
In the original publication of the article, the wrong ��-actin blots were pasted in Figs. 1b and 2c. The correct versions of Figs. 1b and 2c are given in this correction.
References
Dalvie MA, White N, Raine R, Myers JE, London L, Thompson M, Christiani DC (1999) Long-term respiratory health effects of the herbicide, paraquat, among workers in the Western Cape. Occup Environ Med 56:391–396
Costello PR, Drechsel DA, Patel M (2007) Mitochondria are a major source of paraquat-induced reactive oxygen species production in the brain. J Biol Chem 282:14186–14193
Patel S, Singh V, Kumar A, Gupta YK, Singh MP (2006) Status of antioxidant defense system and expression of toxicant responsive genes in striatum of maneb- and paraquat-induced Parkinson’s disease phenotype in mouse: mechanism of neurodegeneration. Brain Res 1081:9–18
Ahmad I, Shukla S, Kumar A, Singh BK, Kumar V, Chauhan AK, Singh D, Pandey HP, Singh C (2013) Biochemical and molecular mechanisms of N-acetyl cysteine and silymarin-mediated protection against maneb- and paraquat-induced hepatotoxicity in rats. Chem Biol Interact 201:9–18
Dixit A, Srivastava G, Verma D, Mishra M, Singh PK, Prakash O, Singh MP (2013) Minocycline, levodopa and MnTMPyP induced changes in the mitochondrial proteome profile of MPTP and menab and paraquat mice models of Parkinson’s disease. Biochim Biophys Acta 1832:1227–1240
McCormack AL, Atienza JG, Johnston LC, Anderson JK, Vu S, Di Monte DA (2005) Role of oxidative stress in paraquat-induced dopaminergic cell degeneration. J Neurochem 93:1030–1037
Zhang J, Fitsanakis V, Gu G, Jing D, Ao M, Amarnath V, Montine T (2003) Manganese ethylene-bis-dithiocarbamate and selective dopaminergic neurodegeneration in rat: a link through mitochondrial disruption. J Neurochem 84:1–11
Kumar A, Singh BK, Ahmad I, Shukla S, Patel DK, Srivastava G, Kumar V, Pandey HP, Singh C (2012) Involvement of NADPH oxidase and glutathione in zinc-induced dopaminergic neurodegeneration in rats: similarity with paraquat neurotoxicity. Brain Res 1438:48–64
Gupta SP, Patel S, Yadav S, Singh AK, Singh S, Singh MP (2010) Involvement of nitric oxide in maneb- and paraquat-induced Parkinson’s disease phenotype in mouse: is there any link with lipid peroxidation? Neurochem Res 35:1206–1213
Nussler AK, Billiar TR (1993) Inflammation, immunoregulation and inducible nitric oxide synthase. J Leukoc Biol 54:171–178
Kumar A, Patel S, Gupta YK, Singh MP (2006) Involvement of endogenous nitric oxide in myeloperoxidase mediated benzo(a)pyrene induced polymorphonuclear leukocytes injury. Mol Cell Biochem 286:43–51
Vitte J, Michel BF, Bongrand P, Gastaut JE (2004) Oxidative stress level in circulating neutrophils is linked to neurodegenerative diseases. J Clin Immunol 24:683–692
Ahmad I, Kumar A, Shukla S, Pandey HP, Singh C (2008) The involvement of nitric oxide in maneb- and paraquat-induced oxidative stress in rat polymorphonuclear leukocytes. Free Radic Res 42:849–862
Riedl AG, Watts PM, Edwards RJ, Boobis AR, Jenner P, Marsden CD (1996) Selective localisation of P450 enzymes and NADPH-P450 oxidoreductase in rat basal ganglia using anti-peptide antisera. Brain Res 743:324–328
Tiwari MN, Singh AK, Ahmad I, Upadhyay G, Singh D, Patel DK, Singh C, Prakash O, Singh MP (2010) Effects of cypermethrin on monoamine transporters, xenobiotic metabolizing enzymes and lipid peroxidation in the rat nigrostriatal system. Free Radic Res 44:1416–1424
Singhal NK, Srivastava G, Patel DK, Jain SK, Singh MP (2011) Melatonin or silymarin reduces maneb- and paraquat-induced Parkinson’s disease phenotype in the mouse. J Pineal Res 50:97–109
Engle MR, Singh SP, Czernik PJ, Gaddy D, Montague DC, Ceci JD, Yang Y, Awasthi S, Awasthi YC, Zimniak P (2004) Physiological role of mGSTA4-4, a glutathione S-transferase metabolizing 4-hydroxynonenal: generation and analysis of mGsta4 null mouse. Toxicol Appl Pharmacol 194:296–308
Cederbaum AI, Yang L, Wang X, Wu D (2012) CYP2E1 sensitizes the liver to LPS- and TNF α-induced toxicity via elevated oxidative and nitrosative stress and activation of ASK-1 and JNK mitogen-activated kinases. Int J Hepatol 2012:582790
Zamora R, Vodovotz Y, Alarcon L, Betten B, Loughran PA, Aulak KS, Stuehr DJ, Gibson KF, Billiar TR (2001) Nitric oxide from the inducible nitric oxide synthase (iNOS) increases the expression of cytochrome P450 2E1 in iNOS-null hepatocytes in the absence of inflammatory stimuli. Arch Biochem Biophys 390:287–294
Gergel D, Misik V, Riesz P, Cederbaum AI (1997) Inhibition of rat and human cytochrome P4502E1 catalytic activity and reactive oxygen radical formation by nitric oxide. Arch Biochem Biophys 337:239–250
Nicholson TE, Dibb S, Renton KW (2004) Nitric oxide mediates an LPS-induced depression of cytochrome P450 (CYP1A) activity in astrocytes. Brain Res 1029:148–154
Westerholt A, Himpel S, Hager-Gensch B, Maier S, Werner M, Stadler J, Doehmer J, Heidecke CD (2004) Intragraft iNOS induction during human liver allograft rejection depresses cytochrome p450 activity. Transpl Int 17:370–378
Tarsitano CA, Paffaro VA Jr, Pauli JR, da Silva GH, Saad MJ, Salgado I, da Cruz-Hofling MA, Hyslop S (2007) Hepatic morphological alterations, glycogen content and cytochrome P450 activities in rats treated chronically with N-(omega)-nitro-l-arginine methyl ester (l-NAME). Cell Tissue Res 329:45–58
Gong P, Cederbaum AI (2006) Nrf2 is increased by CYP2E1 in rodent liver and HepG2 cells and protects against oxidative stress caused by CYP2E1. Hepatology 43:144–153
Ahmad I, Shukla S, Kumar A, Singh BK, Patel DK, Pandey HP, Singh C (2010) Maneb and paraquat-induced modulation of toxicant responsive genes in the rat liver: comparison with polymorphonuclear leukocytes. Chem Biol Interact 188:566–579
Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Tepel M, Echelmeyer M, Orie NN, Zidek W (2000) Increased intracellular reactive oxygen species in patients with end-stage renal failure: effect of hemodialysis. Kidney Int 58:867–872
Desmots F, Rissel M, Pigeon C, Loyer P, Loréal O, Guillouzo A (2002) Differential effects of iron overload on GST isoform expression in mouse liver and kidney and correlation between GSTA4 induction and overproduction of free radicals. Free Radic Biol Med 32:93–101
Lii CK, Liu KL, Cheng YP, Lin AH, Chen HW, Tsai CW (2010) Sulforaphane and a-lipoic acid upregulate the expression of the pi class of glutathione S-transferase through c-Jun and Nrf2 activation. J Nutr 140:885–892
Luceri C, Caderni G, Sanna A, Dolara P (2002) Red wine and black tea polyphenols modulate the expression of cycloxygenase-2, inducible nitric oxide synthase and glutathione-related enzymes in azoxymethane-induced F344 rat colon tumors. J Nutr 32:1376–1379
Singh C, Ahmad I, Kumar A (2007) Pesticides and metals induced Parkinson’s disease: involvement of free radicals and oxidative stress. Cell Mol Biol (Noisy-le-grand) 53:19–28
Kumar A, Ahmad I, Shukla S, Singh BK, Patel DK, Pandey HP, Singh C (2010) Effect of zinc and paraquat co-exposure on neurodegeneration: modulation of oxidative stress and expression of metallothioneins, toxicant responsive and transporter genes in rats. Free Radic Res 44:950–965
Ruiz-Laguna J, Abril N, Garcia-Barrera T, Gomez-Ariza JL, Lopez-Barea J, Pueyo C (2006) Absolute transcript expression signatures of Cyp and Gst genes in Mus spretus to detect environmental contamination. Environ Sci Technol 40:3646–3652
Awasthi YC, Yang Y, Tiwari NK, Patrick B, Sharma A, Li J, Awasthi S (2004) Regulation of 4-hydroxynonenal-mediated signaling by glutathione S-transferases. Free Radic Biol Med 37:607–619
Shaik IH, George JM, Thekkumkara TJ, Mehvar R (2008) Protective effects of diallyl sulfide, a garlic constituent, on the warm hepatic ischemia-reperfusion injury in a rat model. Pharm Res 25:2231–2242
Raza H, Prabu SK, Robin MA, Avadhani NG (2004) Elevated mitochondrial cytochrome P450 2E1 and glutathione S-transferase A4-4 in streptozotocin-induced diabetic rats: tissue-specific variations and roles in oxidative stress. Diabetes 53:185–194
Zimniak L, Awasthi S, Srivastava SK, Zimniak P (1997) Increased resistance to oxidative stress in transfected cultured cells overexpressing glutathione-S-transferase mGSTA4-4. Toxicol Appl Pharmacol 143:221–229
van Iersel ML, Ploemen JP, Lo Bello M, Federici G, van Bladeren PJ (1997) Interactions of alpha, beta-unsaturated aldehydes and ketones with human glutathione S-transferase P1-1. Chem Biol Interact 108:67–78
Shireman LM, Kripps KA, Balogh LM, Conner KP, Whittington D, Atkins WM (2010) Glutathione S-transferase A4-4 resists adduction by 4-hydroxynonenal. Arch Biochem Biophys 504:182–189
Ray S, Sengupta A, Ray A (2007) Effect of paraquat anti-oxidant system in rats. Ind J Exp Biol 45:432–438
Thiruchelvam M, Prokopenko O, Cory-Slechta DA, Buckley B, Mirochnitchenko O (2005) Overexpression of superoxide dismutase or glutathione peroxidase protects against the paraquat + maneb-induced Parkinson disease phenotype. J Biol Chem 280:22530–22539
Mollace V, Iannone M, Muscoli C, Palma E, Granato T, Rispoli V, Nistico R, Rotiroti D, Salvemini D (2003) The role of oxidative stress in paraquat-induced neurotoxicity in rats: protection by non-peptidyl superoxide dismutase. Neurosci Lett 335:163–166
Marí M, Cederbaum AI (2001) Induction of catalase, alpha, and microsomal glutathione S-transferase in CYP2E1 overexpressing HepG2 cells and protection against short-term oxidative stress. Hepatology 33:652–661
Minelli A, Conte C, Grottelli S, Bellezza I, Emiliani C, Bolanos JP (2009) Cyclo (His-Pro) up-regulates heme oxygenase-1 via activation of Nrf2-ARE signaling. J Neurochem 111:956–966
Acknowledgments
Authors acknowledge the Indian Council of Medical Research (ICMR), New Delhi, India for the financial support of a part of the study. Authors sincerely thank the CSIR, New Delhi, India for providing research fellowships to Israr Ahmad and Amit Kumar Chauhan; ICMR, New Delhi, India to Smriti Shukla; University Grants Commission (UGC), New Delhi, India to Brajesh Kumar Singh and Deepali Singh and Department of Biotechnology, New Delhi, India to Vinod Kumar. The IITR communication number of this article is 3131.
Conflicts of interest
The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ahmad, I., Shukla, S., Singh, D. et al. CYP2E1-mediated oxidative stress regulates HO-1 and GST expression in maneb- and paraquat-treated rat polymorphonuclear leukocytes. Mol Cell Biochem 393, 209–222 (2014). https://doi.org/10.1007/s11010-014-2062-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-014-2062-y