Abstract
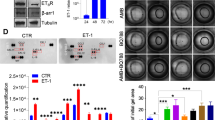
Ovarian carcinoma is a common gynecological malignancy and a great threat to health as a result of metastasis. The chemokine stromal-derived factor (SDF-1) plays multiple roles in tumor pathogenesis. However, the precise molecular mechanism underlying SDF-1-induced ovarian cancer cell invasion is still undefined. αvβ6 integrin is an important factor in tumor progression. Therefore, we speculate that SDF-1-enhanced ovarian cancer cell invasion is related to αvβ6 integrin-mediated signaling. After culturing with SDF-1, an obvious time- and dose-dependent increase in αvβ6 integrin was demonstrated. Furthermore, CXC receptor 4 (CXCR4) was responsible for SDF-1-induced αvβ6 integrin expression. Simultaneously, SDF-1 was found to dramatically enhance extracellular matrix degradation via urokinase-type plasminogen activator (uPA) expression and cell invasion by αvβ6 integrin expression; these reinforce failed to be increased when pretreatment was performed with the CXCR4 inhibitor AMD3100 or anti-αvβ6 integrin antibody, respectively. In addition, αvβ6 integrin induced the phosphorylation of p38 MAPK and PI3 K/Akt, contributing to the up-regulation of uPA, as treatment with the specific inhibitor for p38 mitogen-activated protein kinases (MAPK) (SB203580) or phosphatidylinositol 3-kinase (PI3 K)/Akt (LY294002) strikingly abrogated uPA expression. Taken together, these results demonstrated that SDF-1 enhanced ovarian cancer cell invasion through αvβ6 integrin-mediated uPA expression via the p38 MAPK and PI3 K/Akt pathway. Consequently, our findings will provide a new explanation about how SDF-1 aggravates the pathogenesis of ovarian cancer.





Similar content being viewed by others
References
Slamon DJ, Godolphin W, Jones LA, Holt JA, Wong SG, Keith DE, Levin WJ, Stuart SG, Udove J, Ullrich A (1989) Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science (New York, NY) 244(4905):707
Naora H, Montell DJ (2005) Ovarian cancer metastasis: integrating insights from disparate model organisms. Mol Cell Biochem 5(5):355–366
Kessenbrock K, Plaks V, Werb Z (2010) Matrix metalloproteinases: regulators of the tumor microenvironment. Cell 141(1):52–67
Guo W, Giancotti FG (2004) Integrin signalling during tumour progression. Nat Rev Mol Cell Biol 5(10):816–826
Arihiro K, Kaneko M, Fujii S, Inai K, Yokosaki Y (2000) Significance of α9β1 and αvβ6 integrin expression in breast carcinoma. Breast Cancer 7(1):19–26
Yue J, Zhang K, Chen JF (2012) Role of integrins in regulating proteases to mediate extracellular matrix remodeling. Cancer Microenviron 5(3):1–9
Hezel AF, Deshpande V, Zimmerman SM, Contino G, Alagesan B, O’Dell MR, Rivera LB, Harper J, Lonning S, Brekken RA (2012) TGF-β and αvβ6 integrin act in a common pathway to suppress pancreatic cancer progression. Cancer Res 72(18):4840–4845
Yang SB, Du Y, Wu BY, Xu SP, Wen JB, Zhu M, Cai CH, Yang PC (2012) Integrin alphavbeta6 promotes tumor tolerance in colorectal cancer. Cancer Immunol Immunother 61(3):335–342
Eberlein C, Kendrew J, McDaid K, Alfred A, Kang J, Jacobs V, Ross S, Rooney C, Smith N, Rinkenberger J (2012) A human monoclonal antibody 264RAD targeting αvβ6 integrin reduces tumour growth and metastasis, and modulates key biomarkers in vivo. Oncogene
Thomas GJ, Poomsawat S, Lewis MP, Hart IR, Speight PM, Marshall JF (2001) αvβ6 integrin upregulates matrix metalloproteinase 9 and promotes migration of normal oral keratinocytes. J Invest Dermatol 116(6):898–904
Hazelbag S, Kenter G, Gorter A, Dreef E, Koopman L, Violette S, Weinreb P, Fleuren G (2007) Overexpression of the αvβ6 integrin in cervical squamous cell carcinoma is a prognostic factor for decreased survival. J Pathol 212(3):316–324
Bandyopadhyay A, Raghavan S (2009) Defining the role of integrin αvβ6 in cancer. Curr Drug Targets 10(7):645
Teicher BA, Fricker SP (2010) CXCL12 (SDF-1)/CXCR4 pathway in cancer. Clin Cancer Res 16(11):2927–2931
Righi E, Kashiwagi S, Yuan J, Santosuosso M, Leblanc P, Ingraham R, Forbes B, Edelblute B, Collette B, Xing D (2011) CXCL12/CXCR4 blockade induces multimodal antitumor effects that prolong survival in an immunocompetent mouse model of ovarian cancer. Cancer Res 71(16):5522–5534
Gillette JM, Larochelle A, Dunbar CE, Lippincott-Schwartz J (2009) Intercellular transfer to signalling endosomes regulates an ex vivo bone marrow niche. Nat Cell Biol 11(3):303–311
Phillips RJ, Burdick MD, Lutz M, Belperio JA, Keane MP, Strieter RM (2003) The stromal derived factor-1/CXCL12-CXC chemokine receptor 4 biological axis in non-small cell lung cancer metastases. Am J Respir Crit Care Med 167(12):1676–1686
Liang Z, Wu T, Lou H, Yu X, Taichman RS, Lau SK, Nie S, Umbreit J, Shim H (2004) Inhibition of breast cancer metastasis by selective synthetic polypeptide against CXCR4. Cancer Res 64(12):4302–4308
Müller A, Homey B, Soto H, Ge N, Catron D, Buchanan ME, McClanahan T, Murphy E, Yuan W, Wagner SN (2001) Involvement of chemokine receptors in breast cancer metastasis. Nature 410(6824):50–56
Sun YX, Fang M, Wang J, Cooper CR, Pienta KJ, Taichman RS (2006) Expression and activation of αvβ3 integrins by SDF-1/CXC12 increases the aggressiveness of prostate cancer cells. Prostate 67(1):61–73
Kryczek I, Wei S, Keller E, Liu R, Zou W (2007) Stroma-derived factor (SDF-1/CXCL12) and human tumor pathogenesis. Am J Physiol Cell Physiol 292(3):C987–C995
Scotton CJ, Wilson JL, Scott K, Stamp G, Wilbanks GD, Fricker S, Bridger G, Balkwill FR (2002) Multiple actions of the chemokine CXCL12 on epithelial tumor cells in human ovarian cancer. Cancer Res 62(20):5930–5938
Yuecheng Y, Xiaoyan X (2007) Stromal-cell derived factor-1 regulates epithelial ovarian cancer cell invasion by activating matrix metalloproteinase-9 and matrix metalloproteinase-2. Eur J Cancer Prev 16(5):430–435
Benasciutti E, Pagès G, Kenzior O, Folk W, Blasi F, Crippa MP (2004) MAPK and JNK transduction pathways can phosphorylate Sp1 to activate the uPA minimal promoter element and endogenous gene transcription. Blood 104(1):256–262
Xu MY, Porte J, Knox AJ, Weinreb PH, Maher TM, Violette SM, McAnulty RJ, Sheppard D, Jenkins G (2009) Lysophosphatidic acid induces alphavbeta6 integrin-mediated TGF-beta activation via the LPA2 receptor and the small G protein G alpha(q). Am J Pathol 174(4):1264–1279
Lakka SS, Jasti SL, Gondi C, Boyd D, Chandrasekar N, Dinh DH, Olivero WC, Gujrati M, Rao JS (2002) Downregulation of MMP-9 in ERK-mutated stable transfectants inhibits glioma invasion in vitro. Oncogene 21(36):5601–5608
Thomas G, Nyström M, Marshall J (2006) αvβ6 integrin in wound healing and cancer of the oral cavity. J Oral Pathol Med 35(1):1–10
Stewart DA, Cooper CR, Sikes RA (2004) Changes in extracellular matrix (ECM) and ECM-associated proteins in the metastatic progression of prostate cancer. Reprod Biol Endocrinol 2(2):1–13
Burns JM, Summers BC, Wang Y, Melikian A, Berahovich R, Miao Z, Penfold MET, Sunshine MJ, Littman DR, Kuo CJ (2006) A novel chemokine receptor for SDF-1 and I-TAC involved in cell survival, cell adhesion, and tumor development. J Exp Med 203(9):2201–2213
Giancotti FG, Ruoslahti E (1999) Integrin signaling. Science 285(5430):1028–1033
Wiseman BS, Werb Z (2002) Stromal effects on mammary gland development and breast cancer. Science 296(5570):1046–1049
Vivanco I, Sawyers CL (2002) The phosphatidylinositol 3-kinase-AKT pathway in human cancer. Nat Rev Cancer 2(7):489–501
Acknowledgments
Financial support was provided by National Natural Science Foundation of China (NSFC) (No. 30872740).
Disclosures
The authors have no financial conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Baoyao Xue, Weiguang Wu, and Kan Huang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Xue, B., Wu, W., Huang, K. et al. Stromal cell-derived factor-1 (SDF-1) enhances cells invasion by αvβ6 integrin-mediated signaling in ovarian cancer. Mol Cell Biochem 380, 177–184 (2013). https://doi.org/10.1007/s11010-013-1671-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-013-1671-1