Abstract
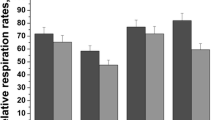
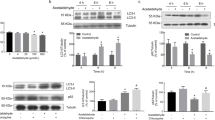
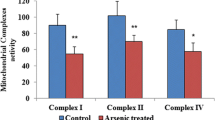
Chronic alcohol consumption causes numerous biochemical and biophysical changes in the central nervous system, in which mitochondria is the primary organelle affected. In the present study, we hypothesized that alcohol alters the mitochondrial membrane properties and leads to mitochondrial dysfunction via mitochondrial reactive oxygen species (mROS) and reactive nitrogen species (RNS). Alcohol-induced hypoxia further enhances these effects. Administration of alcohol to rats significantly increased the mitochondrial lipid peroxidation and protein oxidation with decreased SOD2 mRNA and protein expression was decreased, while nitric oxide (NO) levels and expression of iNOS and nNOS in brain cortex were increased. In addition, alcohol augmented HIF-1α mRNA and protein expression in the brain cortex. Results from this study showed that alcohol administration to rats decreased mitochondrial complex I, III, IV activities, Na+/K+-ATPase activity and cardiolipin content with increased anisotropic value. Cardiolipin regulates numerous enzyme activities, especially those related to oxidative phosphorylation and coupled respiration. In the present study, decreased cardiolipin could be ascribed to ROS/RNS-induced damage. In conclusion, alcohol-induced ROS/RNS is responsible for the altered mitochondrial membrane properties, and alcohol-induced hypoxia further enhance these alterations, which ultimately leads to mitochondrial dysfunction.





Similar content being viewed by others
References
Harper C, Matsumoto I (2005) Ethanol and brain damage. Curr Opin Pharmacol 5:73–78
Lin MT, Beal MF (2006) Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 443:787–795
Almansa I, Fernandez A, García-Ruiz C, Muriach M, Barcia JM, Miranda M, Fernández-Checa JC, Romero FJ (2009) Brain mitochondrial alterations after chronic alcohol consumption. J Physiol Biochem 65:305–312
Reddy VD, Padmavathi P, Kavitha G, Gopi S, Varadacharyulu N (2011) Emblica officinalis ameliorates alcohol-induced brain mitochondrial dysfunction in rats. J Med Food 14:62–68
Bailey SM (2003) A review of the role of reactive oxygen and nitrogen species in alcohol-induced mitochondrial dysfunction. Free Radic Res. 37:585–596
Venkatraman A, Shiva S, Wigley A, Ulasova E, Shhieng D, Bailey SM, Darley-Usmar VM (2004) The role of iNOS in alcohol-dependent hepatotoxicity and mitochondrial dysfunction in mice. Hepatol 40:565–573
Zelickson BR, Benavides GA, Johnson MS, Chacko BK, Venkatraman A, Landar A, Betancourt AM, Bailey SM, Darley-Usmar VM (2011) Nitric oxide and hypoxia exacerbate alcohol-induced mitochondrial dysfunction in hepatocytes. Biochim Biophys Acta 1807:1573–1582
Deng X, Deitrich RA (2007) Ethanol metabolism and effects: nitric oxide and its interaction. Curr Clin Pharmacol 2:145–153
Haorah J, Ramirez SH, Floreani N, Gorantla S, Morsey B, Persidsky Y (2008) Mechanism of alcohol-induced oxidative stress and neuronal injury. Free Radic Biol Med 45:1542–1550
Young TA, Bailey SM, Van Horn CG, Cunningham CC (2006) Chronic ethanol consumption decreases mitochondrial and glycolytic production of ATP in liver. Alcohol 41:254–260
Wang SM, Wu R (2009) The double danger of ethanol and hypoxia: their effects on a hepatoma cell line. Int J Clin Exp Pathol 2:182–189
Semenza GL (2012) Hypoxia-inducible factors in physiology and medicine. Cell 148:399–408
Taylor CT, Moncada S (2010) Nitric oxide, cytochrome c oxidase, and the cellular response to hypoxia. Arterioscler Thromb Vasc Biol 30:643–647
Ball KA, Nelson AW, Foster DG, Poyton RO (2012) Nitric oxide produced by cytochrome c oxidase helps stabilize HIF-1α in hypoxic mammalian cells. Biochem Biophys Res Commun 420:727–732
Poyton RO, Ball KA, Castello PR (2009) Mitochondrial generation of free radicals and hypoxic signaling. Trends Endocrinol Metab 20:332–340
Reddy VD, Padmavathi P, Paramahamsa M, Varadacharyulu N (2009) Modulatory role of Emblica officinalis against alcohol induced biochemical and biophysical changes in rat erythrocyte membranes. Food Chem Toxicol 47:1958–1963
Paradies G, Petrosillo G, Paradies V, Ruggiero FM (2009) Role of cardiolipin peroxidation and Ca2+ in mitochondrial dysfunction and disease. Cell Calcium 45:643–650
Pfeiffer K, Gohil V, Stuart RA, Hunte C, Brandt U, Greenberg ML, Schägger H (2003) Cardiolipin stabilizes respiratory chain supercomplexes. J Biol Chem 278:52873–52880
Chicco AJ, Sparagna GC (2007) Role of cardiolipin alterations in mitochondrial dysfunction and disease. Am J Physiol Cell Physiol 292:C33–C44
Muriel P, Sandoval G (2000) Nitric oxide and peroxynitrite anion modulate liver plasma membrane fluidity and Na+/K+-ATPase activity. Nitric Oxide 4:333–342
Comellas AP, Dada LA, Lecuona E, Pesce LM, Chandel NS, Quesada N, Budinger GR, Strous GJ, Ciechanover A, Sznajder JI (2006) Hypoxia-mediated degradation of Na, K-ATPase via mitochondrial reactive oxygen species and the ubiquitin-conjugating system. Circ Res 98:1314–1322
Brustovetsky W, Dubinsky JM (2000) Dual responses of CNS mitochondria to elevated calcium. J Neurosci 20:103–113
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Reznick AZ, Packer L (1994) Oxidative damage to proteins: spectroscopic method for carbonyl assay. Methods Enzymol 233:357–363
Sastry KHV, Moudgal RP, Mohan J, Tyagi JS, Rao GS (2002) Spectrophotometric determination of serum nitrite and nitrate by copper–cadmium alloy. Anal Biochem 306:79–82
King TE, Robert HL (1967) Preparations and properties of soluble NADH dehydrogenases from cardiac muscle. In: Estabrook RW, Pullman ME (eds) Methods in enzymology, vol 10. Academic Press Inc., New York, pp 275–294
King TE (1967) Preparation of succinate dehydrogenase and reconstitution of succinate oxidase. In: Estabrook RW, Pullman ME (eds) Methods enzymol, vol 10. Academic Press Inc., New York, pp 322–331
Krahenbuhl S, Talos C, Wiesmann U, Hoppel CL (1994) Development and evaluation of a spectrophotometric assay for complex III in isolated mitochondria, tissues and fibroblasts from rats and humans. Clin Chim Acta 230:177–187
Cooperstein SJ, Lazarow A (1951) A microspectrophotometric method for the determination of cytochrome oxidase. J Biol Chem 189:665–670
Ismail B, Edelman IS (1985) Assay of Na+, K+-ATPase. Biochem Pharmacol 34:2685–2689
Fiske CH, Subbarow Y (1925) The colorimetric determination of inorganic phosphorus. J Biol Chem 66:375–404
Folch J, Ascoli I, Lees M, Meath JA, Le Baron FN (1951) Preparation of lipid extracts from brain tissue. J Biol Chem 191:833–841
Padmavathi P, Reddy VD, Kavitha G, Paramahamsa M, Varadacharyulu N (2010) Chronic cigarette smoking alters erythrocyte membrane lipid composition and properties in male human volunteers. Nitric Oxide 23:181–186
Lowry OH, Rosebrough NJ, Farr AL, Randall R (1951) Protein measurement with the Folin-phenol reagent. J Biol Chem 193:263–275
Yuan G, Khan SA, Luo W, Nanduri J, Semenza GL, Prabhakar NR (2011) Hypoxia-inducible factor 1 mediates increased expression of NADPH oxidase-2 in response to intermittent hypoxia. J Cell Physiol 226:2925–2933
Omodeo-Sale F, Gramigna D, Campaniello R (1997) Lipid peroxidation and antioxidant systems in rat brain: effect of chronic alcohol consumption. Neurochem Res 22:577–582
Das SK, Hiran KR, Mukherjee S, Vasudevan DM (2007) Oxidative stress is the primary event: effects of ethanol consumption in brain. Indian J Clin Biochem 22:99–104
Crews FT, Nixon K (2009) Mechanisms of neurodegeneration and regeneration in alcoholism. Alcohol 44:115–127
Rump TJ, Abdul Muneer PM, Szlachetka AM, Lamb A, Haorei C, Alikunju S, Xiong H, Keblesh J, Liu J, Zimmerman MC, Jones J, Donohue TM Jr, Persidsky Y, Haorah J (2010) Acetyl-l-carnitine protects neuronal function from alcohol-induced oxidative damage in the brain. Free Radic Biol Med 49:1494–1504
Catala A (2009) Lipid peroxidation of membrane phospholipids generates hydroxy-alkenals and oxidized phospholipids active in physiological and/or pathological conditions. Chem Phys Lipids 157:1–11
Davis RL, Syapin PJ (2005) Interactions of alcohol and nitric-oxide synthase in the brain. Brain Res Rev 49:494–504
Olson N, van der Vliet A (2011) Interactions between nitric oxide and hypoxia-inducible factor signaling pathways in inflammatory disease. Nitric Oxide 25:125–137
Mantena SK, King AL, Andringa KK, Landar A, Darley-Usmar V, Bailey SM (2007) Novel interactions of mitochondria and reactive oxygen/nitrogen species in alcohol mediated liver disease. World J Gastroenterol 13:4967–4973
Nath B, Szabo G (2012) Hypoxia and hypoxia inducible factors: diverse roles in liver diseases. Hepatology 55:622–633
Mansfield KD, Guzy RD, Pan Y, Young RM, Cash TP, Schumacker PT, Simon MC (2005) Mitochondrial dysfunction resulting from loss of cytochrome c impairs cellular oxygen sensing and hypoxic HIF-alpha activation. Cell Metab 1:393–399
Padmavathi P, Reddy VD, Maturu P, Varadacharyulu N (2010) Smoking-induced alterations in platelet membrane fluidity and Na(+)/K(+)-ATPase activity in chronic cigarette smokers. J Atheroscler Thromb 17:619–627
Wiswedel I, Gardemann A, Storch A, Peter D, Schild L (2010) Degradation of phospholipids by oxidative stress-exceptional significance of cardiolipin. Free Radic Res 44:135–145
Petrosillo G, Portincasa P, Grattagliano I, Casanova G, Matera M, Ruggiero FM, Ferri D, Paradies G (2007) Mitochondrial dysfunction in rat with nonalcoholic fatty liver Involvement of complex I, reactive oxygen species and cardiolipin. Biochim Biophys Acta 1767:1260–1267
Chandel NS, McClintock DS, Feliciano CE, Wood TM, Melendez JA, Rodriguez AM, Schumacker PT (2000) Reactive oxygen species generated at mitochondrial complex III stabilize hypoxia-inducible factor-1alpha during hypoxia: a mechanism of O2 sensing. J Biol Chem 275:25130–25138
Castello PR, David PS, McClure T, Crook Z, Poyton RO (2006) Mitochondrial cytochrome oxidase produces nitric oxide under hypoxic conditions: implications for oxygen sensing and hypoxic signaling in eukaryotes. Cell Metab 3:277–287
Chong PL, Fortes PA, Jameson DM (1985) Mechanisms of inhibition of (Na, K)-ATPase by hydrostatic pressure studied with fluorescent probes. J Biol Chem 260:14484–14490
Sutherland E, Dixon BS, Leffert HL, Skally H, Zaccaro L, Simon FR (1998) Biochemical localization of hepatic surface membrane Na+, K+-ATPase activity depends on membrane lipid fluidity. Proc Nat Acad Sci 85:8673–8677
Acknowledgments
The authors thank the Director and staff of the Center for Cellular and Molecular Biology, Hyderabad, India, for providing facilities to carry out fluidity studies. This study was supported in part by the University Grants Commission (Grant No. F-3-11/97), New Delhi, India.
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reddy, V.D., Padmavathi, P., Kavitha, G. et al. Alcohol-induced oxidative/nitrosative stress alters brain mitochondrial membrane properties. Mol Cell Biochem 375, 39–47 (2013). https://doi.org/10.1007/s11010-012-1526-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-012-1526-1