Abstract
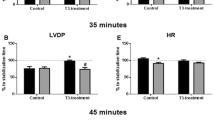
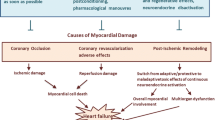
The present study explored the effects of thyroid hormone (TH) treatment on post-ischemic cardiac function and potential implicated mechanisms. Acute myocardial infarction (AMI) was induced in mice by coronary artery ligation while sham-operated animals served as controls. This procedure resulted in a marked depression of cardiac function and significant reduction in TH levels in plasma. TH was given at a dose aiming to normalize T3 levels in plasma [AMI-TH (A)] and also at higher doses. The group of animals treated with the highest dose of TH, which displayed significantly increased mortality rate was included in the study [AMI-TH (B)]. In AMI-TH (A) mice, TH significantly improved left ventricular (LV) ejection fraction (EF%), [27.9% (1.4) in AMI versus 38.0 (3.1) in AMI-TH (A), P < 0.05], and favorably remodeled LV chamber while α-MHC was the dominant isoform expressed. In AMI-TH (B) mice, TH treatment resulted in increased mortality as compared to untreated mice (73% vs 47%, P < 0.05), while the favorable effect of TH was not evident in the survived animals. At the molecular level, TH, at the replacement dose, modestly increased p-Akt levels in the myocardium without any change in p-ERK levels. On the contrary, TH at the higher dose resulted in further increase in p-Akt along with an increase in p-ERK levels. In conclusion, TH appears to have a dose-dependent bimodal effect on post-ischemic cardiac performance and this effect may, at least in part, be mediated by a distinct pattern of activation of Akt and ERK signaling.



Similar content being viewed by others
References
Bello G, Ceaichisciuc I, Silva S, Antonelli M (2010) The role of thyroid dysfunction in the critically ill: a review of the literature. Minerva Anestesiol 76(11):919–928
De Groot LJ (1999) Dangerous dogmas in medicine: the nonthyroidal illness syndrome. J Clin Endocrinol Metab 84(1):151–164
Friberg L, Werner S, Eggertsen G, Ahnve S (2002) Rapid down-regulation of thyroid hormones in acute myocardial infarction: is it cardioprotective in patients with angina? Arch Intern Med 162(12):1388–1394
Lymvaios I, Mourouzis I, Cokkinos DV, Dimopoulos MA, Toumanidis ST, Pantos C (2011) Thyroid hormone and recovery of cardiac function in patients with acute myocardial infarction: a strong association? Eur J Endocrinol 165(1):107–114. doi:10.1530/EJE-11-0062
Cerillo AG, Storti S, Clerico A, Iervasi G (2010) Thyroid function and cardiac surgery: what should we measure, and when? Ann Thorac Surg 89 (3):1010–1011; author reply 1011–1012. doi:10.1016/j.athoracsur.2009.08.046
Pingitore A, Landi P, Taddei MC, Ripoli A, L’Abbate A, Iervasi G (2005) Triiodothyronine levels for risk stratification of patients with chronic heart failure. Am J Med 118(2):132–136
Iervasi G, Pingitore A, Landi P, Raciti M, Ripoli A, Scarlattini M, L’Abbate A, Donato L (2003) Low-T3 syndrome: a strong prognostic predictor of death in patients with heart disease. Circulation 107(5):708–713
Fontana M, Passino C, Poletti R, Zyw L, Prontera C, Scarlattini M, Clerico A, Emdin M, Iervasi G (2010) Low triiodothyronine and exercise capacity in heart failure. Int J Cardiol 160(4):721–728
Pantos C, Dritsas A, Mourouzis I, Dimopoulos A, Karatasakis G, Athanassopoulos G, Mavrogeni S, Manginas A, Cokkinos DV (2007) Thyroid hormone is a critical determinant of myocardial performance in patients with heart failure: potential therapeutic implications. Eur J Endocrinol 157(4):515–520
Belke DD, Gloss B, Swanson EA, Dillmann WH (2007) Adeno-associated virus-mediated expression of thyroid hormone receptor isoforms-alpha1 and -beta1 improves contractile function in pressure overload-induced cardiac hypertrophy. Endocrinology 148(6):2870–2877
Chen YF, Kobayashi S, Chen J, Redetzke RA, Said S, Liang Q, Gerdes AM (2008) Short term triiodo-l-thyronine treatment inhibits cardiac myocyte apoptosis in border area after myocardial infarction in rats. J Mol Cell Cardiol 44(1):180–187
Forini F, Lionetti V, Ardehali H, Pucci A, Cecchetti F, Ghanefar M, Nicolini G, Ichikawa Y, Nannipieri M, Recchia FA, Iervasi G (2011) Early long-term L-T3 replacement rescues mitochondria and prevents ischemic cardiac remodelling in rats. J Cell Mol Med 15(3):514–524. doi:10.1111/j.1582-4934.2010.01014.x
Henderson KK, Danzi S, Paul JT, Leya G, Klein I, Samarel AM (2009) Physiological replacement of T3 improves left ventricular function in an animal model of myocardial infarction-induced congestive heart failure. Circ Heart Fail 2(3):243–252. doi:10.1161/CIRCHEARTFAILURE.108.810747
Kalofoutis C, Mourouzis I, Galanopoulos G, Dimopoulos A, Perimenis P, Spanou D, Cokkinos DV, Singh J, Pantos C (2010) Thyroid hormone can favorably remodel the diabetic myocardium after acute myocardial infarction. Mol Cell Biochem 345(1–2):161–169. doi:10.1007/s11010-010-0569-4
Kinugawa K, Jeong MY, Bristow MR, Long CS (2005) Thyroid hormone induces cardiac myocyte hypertrophy in a thyroid hormone receptor alpha1-specific manner that requires TAK1 and p38 mitogen-activated protein kinase. Mol Endocrinol 19(6):1618–1628
Kinugawa K, Yonekura K, Ribeiro RC, Eto Y, Aoyagi T, Baxter JD, Camacho SA, Bristow MR, Long CS, Simpson PC (2001) Regulation of thyroid hormone receptor isoforms in physiological and pathological cardiac hypertrophy. Circ Res 89(7):591–598
Ojamaa K, Kenessey A, Shenoy R, Klein I (2000) Thyroid hormone metabolism and cardiac gene expression after acute myocardial infarction in the rat. Am J Physiol Endocrinol Metab 279(6):E1319–E1324
Pantos C, Mourouzis I, Galanopoulos G, Gavra M, Perimenis P, Spanou D, Cokkinos DV (2010) Thyroid hormone receptor alpha1 downregulation in postischemic heart failure progression: the potential role of tissue hypothyroidism. Horm Metab Res 42(10):718–724
Pantos C, Mourouzis I, Markakis K, Dimopoulos A, Xinaris C, Kokkinos AD, Panagiotou M, Cokkinos DV (2007) Thyroid hormone attenuates cardiac remodeling and improves hemodynamics early after acute myocardial infarction in rats. Eur J Cardiothorac Surg 32(2):333–339
Pantos C, Mourouzis I, Markakis K, Tsagoulis N, Panagiotou M, Cokkinos DV (2008) Long-term thyroid hormone administration reshapes left ventricular chamber and improves cardiac function after myocardial infarction in rats. Basic Res Cardiol 103(4):308–318
Pantos C, Mourouzis I, Tsagoulis N, Markakis K, Galanopoulos G, Roukounakis N, Perimenis P, Liappas A, Cokkinos DV (2009) Thyroid hormone at supra-physiological dose optimizes cardiac geometry and improves cardiac function in rats with old myocardial infarction. J Physiol Pharmacol 60(3):49–56
Pantos C, Xinaris C, Mourouzis I, Perimenis P, Politi E, Spanou D, Cokkinos DV (2008) Thyroid hormone receptor alpha 1: a switch to cardiac cell “metamorphosis”? J Physiol Pharmacol 59(2):253–269
Suarez J, Scott BT, Suarez-Ramirez JA, Chavira CV, Dillmann WH (2010) Thyroid hormone inhibits ERK phosphorylation in pressure overload-induced hypertrophied mouse hearts through a receptor-mediated mechanism. Am J Physiol Cell Physiol 299(6):C1524–C1529. doi:10.1152/ajpcell.00168.2010
Pol CJ, Muller A, Zuidwijk MJ, van Deel ED, Kaptein E, Saba A, Marchini M, Zucchi R, Visser TJ, Paulus WJ, Duncker DJ, Simonides WS (2011) Left-ventricular remodeling after myocardial infarction is associated with a cardiomyocyte-specific hypothyroid condition. Endocrinology 152(2):669–679. doi:10.1210/en.2010-0431
The coronary drug project (1972). JAMA 221 (8):918
The coronary drug project research group (1972) The coronary drug project. Findings leading to further modifications of its protocol with respect to dextrothyroxine. JAMA 220(7):996–1008
Goldman S, McCarren M, Morkin E, Ladenson PW, Edson R, Warren S, Ohm J, Thai H, Churby L, Barnhill J, O’Brien T, Anand I, Warner A, Hattler B, Dunlap M, Erikson J, Shih MC, Lavori P (2009) DITPA (3, 5-Diiodothyropropionic Acid), a thyroid hormone analog to treat heart failure: phase II trial veteran’s affairs cooperative study. Circulation 119(24):3093–3100. doi:10.1161/CIRCULATIONAHA.108.834424
Pingitore A, Iervasi G, Gerdes MA (2010) Letter by Pingitore et al. regarding article, “DITPA (3,5-diiodothyropropionic acid), a thyroid hormone analog to treat heart failure: phase II trial Veterans Affairs cooperative study”. Circulation 121 (10):e240; author reply e241. doi:10.1161/CIR.0b013e3181d77f5e
Lee SU, Kim DY, Park SH, Choi DH, Park HW, Han TR (2009) Mild to moderate early exercise promotes recovery from cerebral ischemia in rats. Can J Neurol Sci 36(4):443–449
Schultz RL, Swallow JG, Waters RP, Kuzman JA, Redetzke RA, Said S, de Escobar GM, Gerdes AM (2007) Effects of excessive long-term exercise on cardiac function and myocyte remodeling in hypertensive heart failure rats. Hypertension 50(2):410–416. doi:10.1161/HYPERTENSIONAHA.106.086371
Pantos C, Mourouzis I, Saranteas T, Brozou V, Galanopoulos G, Kostopanagiotou G, Cokkinos DV (2011) Acute T3 treatment protects the heart against ischemia-reperfusion injury via TRalpha1 receptor. Mol Cell Biochem 353(1–2):235–241. doi:10.1007/s11010-011-0791-8
Pantos C, Mourouzis I, Saranteas T, Clave G, Ligeret H, Noack-Fraissignes P, Renard PY, Massonneau M, Perimenis P, Spanou D, Kostopanagiotou G, Cokkinos DV (2009) Thyroid hormone improves postischemic recovery of function while limiting apoptosis: a new therapeutic approach to support hemodynamics in the setting of ischaemia-reperfusion? Basic Res Cardiol 104(1):69–77
Minicucci MF, Azevedo PS, Martinez PF, Lima AR, Bonomo C, Guizoni DM, Polegato BF, Okoshi MP, Okoshi K, Matsubara BB, Paiva SA, Zornoff LA (2011) Critical infarct size to induce ventricular remodeling, cardiac dysfunction and heart failure in rats. Int J Cardiol 151(2):242–243. doi:10.1016/j.ijcard.2011.06.068
Hers I, Vincent EE, Tavare JM (2011) Akt signalling in health and disease. Cell Signal 23(10):1515–1527. doi:10.1016/j.cellsig.2011.05.004
Kim SP, Ha JM, Yun SJ, Kim EK, Chung SW, Hong KW, Kim CD, Bae SS (2011) Transcriptional activation of peroxisome proliferator-activated receptor-gamma requires activation of both protein kinase A and Akt during adipocyte differentiation. Biochem Biophys Res Commun 399(1):55–59. doi:10.1016/j.bbrc.2010.07.038
Sussman MA, Volkers M, Fischer K, Bailey B, Cottage CT, Din S, Gude N, Avitabile D, Alvarez R, Sundararaman B, Quijada P, Mason M, Konstandin MH, Malhowski A, Cheng Z, Khan M, McGregor M (2011) Myocardial AKT: the omnipresent nexus. Physiol Rev 91(3):1023–1070. doi:10.1152/physrev.00024.2010
Kehat I, Davis J, Tiburcy M, Accornero F, Saba-El-Leil MK, Maillet M, York AJ, Lorenz JN, Zimmermann WH, Meloche S, Molkentin JD (2010) Extracellular signal-regulated kinases 1 and 2 regulate the balance between eccentric and concentric cardiac growth. Circ Res 108(2):176–183. doi:10.1161/CIRCRESAHA.110.231514
Matsui T, Li L, Wu JC, Cook SA, Nagoshi T, Picard MH, Liao R, Rosenzweig A (2002) Phenotypic spectrum caused by transgenic overexpression of activated Akt in the heart. J Biol Chem 277(25):22896–22901. doi:10.1074/jbc.M200347200
Bueno OF, De Windt LJ, Tymitz KM, Witt SA, Kimball TR, Klevitsky R, Hewett TE, Jones SP, Lefer DJ, Peng CF, Kitsis RN, Molkentin JD (2000) The MEK1-ERK1/2 signaling pathway promotes compensated cardiac hypertrophy in transgenic mice. EMBO J 19(23):6341–6350. doi:10.1093/emboj/19.23.6341
Gerdes AM, Iervasi G (2010) Thyroid replacement therapy and heart failure. Circulation 122(4):385–393. doi:10.1161/CIRCULATIONAHA.109.917922
Pingitore A, Chen Y, Gerdes AM, Iervasi G (2011) Acute myocardial infarction and thyroid function: new pathophysiological and therapeutic perspectives. Ann Med:(in press). doi:10.3109/07853890.2011.573501
Acknowledgment
The study was partly funded by ongoing European Community ITC-STREP FP7 PONTE Research Project (Project No. 247945).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mourouzis, I., Mantzouratou, P., Galanopoulos, G. et al. Dose-dependent effects of thyroid hormone on post-ischemic cardiac performance: potential involvement of Akt and ERK signalings. Mol Cell Biochem 363, 235–243 (2012). https://doi.org/10.1007/s11010-011-1175-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-011-1175-9