Abstract
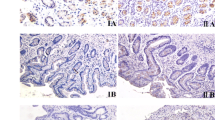
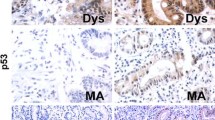
Helicobacter pylori (H. pylori) infection plays a significant role in causing gastric cancer; the exact molecular mechanisms of gastric carcinogenesis have not yet been fully determined. Therefore, this study was planned to examine the role of c-H-ras p21 expression in H. pylori infection at different stages of disease progression from precursor lesions to gastric carcinoma. This study was carried out in 200 patients, consisting of normal gastric mucosa (n = 20), mucosa with chronic gastritis (n = 63), intestinal metaplasia (n = 20), dysplasia (n = 11), and gastric adenocarcinoma (n = 86), in which the H. pylori status have been analysed. The expression of c-H-ras p21 was studied at mRNA as well as protein level using RT-PCR and western blotting, respectively. The localization of c-H-ras p21 was also studied semiquantitatively by immunohistochemistry. The RT-PCR and western blotting results of c-H-ras p21 mRNA and protein expressions were significantly increased in chronic gastritis, intestinal metaplasia, dysplasia, and gastric adenocarcinoma patients, respectively. Immunohistochemical study also showed the increased expression of c-H-ras p21 in the similar way. Overexpression of c-H-ras p21 might be due to H-ras mutation at codon 12 of ras gene family in H. pylori infection. The rate of expression of ras p21 was higher in the H. pylori-infected precursor lesions, chronic gastritis 49/56 (87.5%), intestinal metaplasia 16/17 (94%), and dysplasia 9/11(82%) whereas in the case of H. pylori negative cases these groups, show 12.5, 5.9, and 18.2%, respectively. The data suggested that H. pylori infection may increase the expression of c-H-ras p21 early in the process of gastric carcinogenesis.







Similar content being viewed by others
References
Kato M, Asaka M (2010) Recent knowledge of the relationship between Helicobacter pylori and gastric cancer and recent progress of gastroendoscopic diagnosis and treatment for gastric cancer. Jpn J Clin Oncol 40:828–837
Wang H, Sun Y, Liu S, Yu H, Li W, Zeng J, Chen C, Jia J (2011) Upregulation of progranulin by Helicobacter pylori in human gastric epithelial cells via p38MAPK and MEK1/2 signaling pathway: role in epithelial cell proliferation and migration. FEMS Immunol Med Microbiol 63:82–92
Uemura N, Okamoto S, Yamamoto S, Matsumura N, Yamaguchi S, Yamakido M, Taniyama K, Sasaki N, Schlemper RJ (2001) Helicobacter pylori infection and the development of gastric cancer. N Engl J Med 345:784–789
Huang JQ, Zheng GF, Sumanac K, Irvine EJ, Hunt RH (2003) Meta-analysis of the relationship between cagA seropositivity and gastric cancer. Gastroenterology 125:1636–1644
Houghton J, Wang TC (2005) Helicobacter pylori and gastric cancer: a new paradigm for inflammation-associated epithelial cancers. Gastroenterology 128:1567–1578
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM (2010) Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 127:2893–2917
Wang J, Chi DS, Kalin GB, Sosinski C, Miller LE, Burja I, Thomas E (2002) Helicobacter pylori infection and oncogene expressions in gastric carcinoma and its precursor lesions. Diges Dis Sci 47:107–113
Tahara E (1995) Molecular biology of gastric cancer. World J Surg 19:484–488
Fukuda S, Pelus LM (2004) Activated H-Ras regulates hematopoietic cell survival by modulating survivin. Biochem Biophys Res Commun 323:636–644
Sommer KW, Schamberger CJ, Schmidt GE, Sasgary S, Cerni C (2003) Inhibitor of apoptosis protein (IAP) survivin inhibitor of apoptosis protein (IAP) survivin is upregulated by oncogenic c-H-Ras. Oncogene 22:4266–4280
Spandidos D, Zoumpourlis V, Zachos G, Toas S, Halazonetis T (1995) Specific recognition of a transcriptional element within the human H-ras protooncogene by the p53 tumor-suppressor. Int J Oncol 7:1029–1034
Lee CS (1997) Ras p21 protein immunoreactivity and its relationship to p53 expression and prognosis in gallbladder and extrahepatic biliary carcinoma. Eur J Surg Oncol 23:233–237
Miturski R, Semczuk A, Jakowicki JA (1998) Expression of ras p21 in the stromal cells of human neoplastic endometrium. Eur J Gyanecol Oncol 19:308–311
Bos JL (1989) ras oncogenes in human cancer: a review. Cancer Res 49:4682–4689
Barbacid M (1987) ras genes. Ann Rev Biochem 56:779–827
Schmeiser HH, Janssen JWG, Lyons J, Scherf HR, Pfau W, Buchmann A, Bartram CR, Wiessler M (1990) Aristolochic acid activates ras genes in rat tumors at deoxyadenosine residues. Cancer Res 50:5464–5469
Schmeiser HH, Scherf HR, Wiessler M (1991) Activating mutations at codon 61 of the c-Ha-ras gene in thin-tissue sections of tumors induced by aristolochic acid in rats and mice. Cancer Lett 59:139–143
Wang Y, Meng F, Arlt VM, Mei N, Chen T, Parsons BL (2011) Aristolochic acid-induced carcinogenesis examined by ACB-PCR quantification of H-Ras and K-Ras mutant fraction. Mutagenesis. doi:10.1093/mutage/ger023
Chang EH, Furth ME, Scolnick EM, Lowy DR (1982) Tumorigenic transformation of mammalian cells induced by a normal human gene homologous to the oncogene of Harvey murine sarcoma virus. Nature 297:479–483
Feramisco JR, Gross M, Kamata T, Rosenberg M, Sweet RW (1984) Microinjection of the oncogene form of the human H-ras (T-24) protein results in rapid proliferation of quiescent cells. Cell 38:109–117
Theodorescu D, Cornil I, Fernandez BJ, Kerbel RS (1990) Over expression of normal and mutated forms of H-ras induces orthotopic bladder invasion in a human transitional cell carcinoma. Proc Natl Acad Sci USA 87:9047–9051
Tahara E, Yasui W, Taniyama K, Ochiai A, Yamamoto T, Nakajo S, Yamamoto M (1986) Ha-ras oncogene product in human gastric carcinoma: correlation with invasiveness, metastasis or prognosis. Jpn J Cancer Res 77:517–522
Ohuchi N, Hand PH, Merlo G, Fujita J, Mariani-Costantini R, Thor A, Nose M, Callahan R, Schlom J (1987) Enhanced expression of c-Ha-rasp21 in human stomach adenocarcinomas defined by immunoassays using monoclonal antibodies and in situ hybridization. Cancer Res 47:1413–1420
Dixon MF, Genta RM, Yardley JH, Correa P (1996) Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Pathol 20:1161–1181
Czerniak B, Herz F, Gorczyca W, Koss L (1989) Expression of ras oncogene p21 protein in early gastric carcinoma and adjacent gastric epithelia. Cancer 64:1467–1473
Koh EH, Chung HC, Lee KB, Han EK, Oh SH, Min JS, Choi EM, Youn JK, Kim BS (1992) Point mutation at codon 12 of the c-Ha-ras gene in human gastric cancers. J Korean Med Sci 7:110–115
Kim YJ, Ghu HD, Kim DY, Kim HJ, Kim SK, Park CS (1993) Expression of cellular oncogenes in human gastric carcinoma: c-myc, c-erbB2, and c-Ha-ras. J Surg Oncol 54:167–170
Lee KH, Lee JS, Suh C, Kim SW, Kim SB, Lee JH, Lee MS, Park MY, Sun HS, Kim SH (1995) Clinicopathologic significance of the K-ras gene codon 12 point mutation in stomach cancer: an analysis of 140 cases. Cancer 75:2794–2801
Adjei AA (2001) Blocking oncogenic Ras signaling for cancer therapy. J Natl Cancer Inst 93:1062–1074
Satoh T, Nakafuku M, Kaziro Y (1992) Function of Ras as a molecular switch in signal transduction. J Biol Chem 267:24149–24152
Furth ME, Aldrich TH, Cordon-Cardo C (1987) Expression of ras proto-oncogene proteins in normal human tissues. Oncogene 1:47–58
Suriano G, Oliveira C, Ferreira P et al (2003) Identification of CDH1 germline missense mutations associated with functional inactivation of the E-cadherin protein in young gastric cancer probands. Hum Mol Genet 12:575–582
Kountouras J, Boura P, Lygidakis NJ (2000) New concepts of molecular biology for colon carcinogenesis. Hepatogastroenterology 47:1291–1297
Ierardi E, Francavilla A, Balzano T et al (1997) Effect of Helicobacter pylori eradication on gastric epithelial proliferation. Relationship with ras oncogene p21 expression. Ital J Gastroenterol Hepatol 29:214–219
Watari J, Tanaka A, Tanabe H et al (2007) K-ras mutations and cell kinetics in Helicobacter pylori associated gastric intestinal metaplasia: a comparison before and after eradication in patients with chronic gastritis and gastric cancer. J Clin Pathol 60:921–926
Kim YB, Han JY, Kim TS, Kim PS, Chu YC (2000) Overexpression of c-H-ras p21 is correlated with vascular endothelial growth factor expression and neovascularization in advanced gastric carcinoma. J Gastroenterol Hepatol 15:1393–1399
Costa I, Moral R, Solanas M, Andreu FJ, Ruiz de Villa MC, Escrich E (2011) High corn oil and extra virgin olive oil diets and experimental mammary carcinogenesis: clinicopathological and immunohistochemical p21Ha-Ras expression study. Virchows Arch 458:141–151
Ying H, Jinkun Z, Youyong L, Cuiqiong Y, Wei Q, Jiantao C (1998) The role of Ras gene mutation in gastric cancer and precancerous lesions. J Tongji Med Univ 18:141–144
Farinati F, Cardin R, Cassaro M, Bortolami M, Nitti D, Tieppo C, Zaninotto G, Rugge M (2008) Helicobacter pylori, inflammation, oxidative damage and gastric cancer: a morphological, biological and molecular pathway. Eur J Cancer Prev 17:195–200
Farinati F, Cardin R, Bortolami M, Nitti D, Basso D, de Bernard M, Cassaro M, Sergio A, Rugge M (2008) Oxidative DNA damage in gastric cancer: CagA status and OGG1 gene polymorphism. Int J Cancer 123:51–55
Yu J, Zhang JK (1994) Study on the association between Helicobacter pylori infection and the pathogenesis of gastric cancer by using molecular biological techniques. J Tongji Med Univ 14:65–70
Noguchi M, Hirohashi S, Shimosato Y, Thor A, Schlom J, Tsunokawa Y, Terada M, Sugimura T (1986) Histological demonstration of antigens reactive with anti-p21 ras monoclonal antibody (RAP-5) in human stomach cancers. J Natl Cancer Inst 77:379–385
Suganuma M, Kuruesu M, Okabe S, Sueoka N, Yoshida M, Wakatsuki Y, Fujiki H (2001) Helicobacter pylori membrane protein 1: a new carcinogenic factor of Helicobacter pylori. Cancer Res 61:6356–6359
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sureka, C., Ramesh, T. Molecular assessment of c-H-ras p21 expression in Helicobacter pylori-mediated gastric carcinogenesis. Mol Cell Biochem 362, 169–176 (2012). https://doi.org/10.1007/s11010-011-1139-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-011-1139-0