Abstract
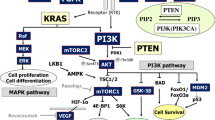
Loss or mutation of the PTEN (phosphatase and tensin homologue deleted on chromosome 10) gene is associated with resistance to epidermal growth factor receptor (EGFR) inhibitors. However, the mechanism underlying remains elusive. In this study, we aimed to explore whether sensitivity to the EGFR tyrosine kinase inhibitor (TKI) is affected by PTEN status in endometrial cancer cells. PTEN siRNA and the PTEN gene were transfected into HEC-1A and Ishikawa endometrial cancer cells using lentiviral vectors. Cells were treated under various concentrations of RG14620 and rapamycin, which are EGFR and mammalian target of rapamycin (mTOR) inhibitors, respectively. The IC50 of RG16420 was determined by using the MTT method. Cell apoptosis and the cell cycle were studied, and activation of EGFR, AKT, and p70S6 were detected by Western blot analysis. Loss of PTEN promoted cell proliferation and led to significant increases in the levels of EGFR, phospho-EGFR, AKT, phospho-AKT, and phospho-mTOR proteins. Ishikawa and HEC-1APTENkd cells that displayed loss and inactivation of PTEN function were resistant to RG14620. HEC-1A and IshikawaPTEN cells with intact PTEN were sensitive to RG14620. The combination of two inhibitors was more effective than both monotherapies, particularly in carcinoma cells with PTEN dysfunction. Decreased phospho-EGFR protein expression was observed in all cell lines that were sensitive to RG14620. Decreased phospho-AKT and phospho-p70S6 protein expression was observed in PTEN-intact cells that were sensitive to RG14620. PTEN loss results in resistance to EGFR TKI, which was reversed by PTEN reintroduction or mTOR inhibitor treatment. The combined treatment of EGFR TKI and the mTOR inhibitor provided a synergistic effect by promoting cell death in PTEN-deficient and PTEN-intact endometrial cancer cells, particularly in PTEN-deficient carcinoma cells with up-regulated EGFR activation.







Similar content being viewed by others
References
Okuda T, Sekizawa A, Purwosunu Y, Nagatsuka M, Morioka M, Hayashi M, Okai T (2010) Genetics of endometrial cancers. Obstet Gynecol Int 2010:984013
Doll A, Abal M, Rigau M, Monge M, Gonzalez M, Demajo S, Colas E, Llaurado M, Alazzouzi H, Planaguma J, Lohmann MA, Garcia J, Castellvi S, Ramon YCJ, Gil-Moreno A, Xercavins J, Alameda F, Reventos J (2008) Novel molecular profiles of endometrial cancer-new light through old windows. J Steroid Biochem Mol Biol 108:221–229
Herbst RS (2004) Review of epidermal growth factor receptor biology. Int J Radiat Oncol Biol Phys 59:21–26
Laurent-Puig P, Lievre A, Blons H (2009) Mutations and response to epidermal growth factor receptor inhibitors. Clin Cancer Res 15:1133–1139
Chon HS, Hu W, Kavanagh JJ (2006) Targeted therapies in gynecologic cancers. Curr Cancer Drug Targets 6:333–363
Bansal N, Yendluri V, Wenham RM (2009) The molecular biology of endometrial cancers and the implications for pathogenesis, classification, and targeted therapies. Cancer Control 16:8–13
Cheung AN (2007) Molecular targets in gynaecological cancers. Pathology 39:26–45
Gadducci A, Tana R, Cosio S, Fanucchi A, Genazzani AR (2008) Molecular target therapies in endometrial cancer: from the basic research to the clinic. Gynecol Endocrinol 24:239–249
She QB, Solit D, Basso A, Moasser MM (2003) Resistance to gefitinib in PTEN-null HER-overexpressing tumor cells can be overcome through restoration of PTEN function or pharmacologic modulation of constitutive phosphatidylinositol 3′-kinase/AKT pathway signaling. Clin Cancer Res 9:4340–4346
Festuccia C, Muzi P, Millimaggi D, Biordi L, Gravina GL, Speca S, Angelucci A, Dolo V, Vicentini C, Bologna M (2005) Molecular aspects of gefitinib antiproliferative and pro-apoptotic effects in PTEN-positive and PTEN-negative prostate cancer cell lines. Endocr Relat Cancer 12:983–998
Mutter GL (2001) PTEN, a protean tumor suppressor. Am J Pathol 158:1895–1898
Mutter GL, Lin MC, Fitzgerald JT, Kum JB, Baak JP, Lees JA, Weng LP, Eng C (2000) Altered PTEN expression as a diagnostic marker for the earliest endometrial precancers. J Natl Cancer Inst 92:924–930
Uegaki K, Kanamori Y, Kigawa J, Kawaguchi W, Kaneko R, Naniwa J, Takahashi M, Shimada M, Oishi T, Itamochi H, Terakawa N (2006) PTEN is involved in the signal transduction pathway of contact inhibition in endometrial cells. Cell Tissue Res 323:523–528
DeGraffenried LA, Fulcher L, Friedrichs WE, Grunwald V, Ray RB, Hidalgo M (2004) Reduced PTEN expression in breast cancer cells confers susceptibility to inhibitors of the PI3 kinase/AKT pathway. Ann Oncol 15:1510–1516
Neshat MS, Mellinghoff IK, Tran C, Stiles B, Thomas G, Petersen R, Frost P, Gibbons JJ, Wu H, Sawyers CL (2001) Enhanced sensitivity of PTEN-deficient tumors to inhibition of FRAP/mTOR. Proc Natl Acad Sci USA 98:10314–10319
Albitar L, Carter MB, Davies S, Leslie KK (2007) Consequences of the loss of p53, RB1, and PTEN: relationship to gefitinib resistance in endometrial cancer. Gynecol Oncol 106:94–104
Chon HS, Hu W, Kavanagh JJ (2006) Targeted therapies in gynecologic cancers. Curr Cancer Drug Targets 6:333–363
Wang MY, Lu KV, Zhu S, Dia EQ, Vivanco I, Shackleford GM, Cavenee WK, Mellinghoff IK, Cloughesy TF, Sawyers CL, Mischel PS (2006) Mammalian target of rapamycin inhibition promotes response to epidermal growth factor receptor kinase inhibitors in PTEN-deficient and PTEN-intact glioblastoma cells. Cancer Res 66:7864–7869
Oza AM, Eisenhauer EA, Elit L, Cutz JC, Sakurada A, Tsao MS, Hoskins PJ, Biagi J, Ghatage P, Mazurka J, Provencher D, Dore N, Dancey J, Fyles A (2008) Phase II study of erlotinib in recurrent or metastatic endometrial cancer: NCIC IND-148. J Clin Oncol 26:4319–4325
Tang LL, Yokoyama Y, Wan X, Iwagaki S, Niwa K, Tamaya T (2006) PTEN sensitizes epidermal growth factor-mediated proliferation in endometrial carcinoma cells. Oncol Rep 15:855–859
Xiao L, Yang YB, Li XM, Xu CF, Li T, Wang XY (2010) Differential sensitivity of human endometrial carcinoma cells with different PTEN expression to mitogen-activated protein kinase signaling inhibits and implications for therapy. J Cancer Res Clin Oncol 136:1089–1099
Xiao L, Yang YB, Shen HM, Xu CF, Wang XY, Li XM (2009) Effect of signal transduction inhibitors on human endometrial carcinoma cells with differential PTEN gene expression. Zhonghua Fu Chan Ke Za Zhi 44:681–685
Wan X, Yokoyama Y, Shinohara A, Takahashi Y, Tamaya T (2002) PTEN augments staurosporine-induced apoptosis in PTEN-null Ishikawa cells by downregulating PI3K/AKT signaling pathway. Cell Death Differ 9:414–420
Wan X, Li J, Xie X, Lu W (2007) PTEN augments doxorubicin-induced apoptosis in PTEN-null Ishikawa cells. Int J Gynecol Cancer 17:808–812
Zhao LJ, Liu N, Li XP, Wang JL, Wei LH (2010) Phosphatase and tensin homolog gene inhibits the effect induced by gonadotropin-releasing hormone subtypes in human endometrial carcinoma cells. Chin Med J (Engl) 123:1170–1175
Sakurada A, Hamada H, Fukushige S, Yokoyama T, Yoshinaga K, Furukawa T, Sato S, Yajima A, Sato M, Fujimura S, Horii A (1999) Adenovirus-mediated delivery of the PTEN gene inhibits cell growth by induction of apoptosis in endometrial cancer. Int J Oncol 15:1069–1074
Wendel HG, Malina A, Zhao Z, Zender L, Kogan SC, Cordon-Cardo C, Pelletier J, Lowe SW (2006) Determinants of sensitivity and resistance to rapamycin-chemotherapy drug combinations in vivo. Cancer Res 66:7639–7646
Neshat MS, Mellinghoff IK, Tran C, Stiles B, Thomas G, Petersen R, Frost P, Gibbons JJ, Wu H, Sawyers CL (2001) Enhanced sensitivity of PTEN-deficient tumors to inhibition of FRAP/mTOR. Proc Natl Acad Sci USA 98:10314–10319
Darb-Esfahani S, Faggad A, Noske A, Weichert W, Buckendahl AC, Muller B, Budczies J, Roske A, Dietel M, Denkert C (2009) Phospho-mTOR and phospho-4EBP1 in endometrial adenocarcinoma: association with stage and grade in vivo and link with response to rapamycin treatment in vitro. J Cancer Res Clin Oncol 135:933–941
Acknowledgments
This study was supported by a grant from the Science and Technology Development Fund of the Macao Special Administrative Region (002/2009/A) and the National Natural Science Foundation of China (30772332). We thank Professor Zehua Wang (Department of Obstetrics & Gynecology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei, P.R. China) for supplying the cell lines.
Author information
Authors and Affiliations
Corresponding author
Additional information
Tian Li and Yuebo Yang contributed equally to this study.
Rights and permissions
About this article
Cite this article
Li, T., Yang, Y., Li, X. et al. EGFR- and AKT-mediated reduction in PTEN expression contributes to tyrphostin resistance and is reversed by mTOR inhibition in endometrial cancer cells. Mol Cell Biochem 361, 19–29 (2012). https://doi.org/10.1007/s11010-011-1082-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-011-1082-0