Abstract
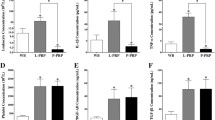
Platelets are involved in hemostasis, wound healing, and tumor growth. Autologous blood products are commonly used to facilitate healing in a variety of clinical surgery applications. Recently, it was shown that platelet-rich plasma (PRP) has more specific growth factors that participate in the healing process. This study investigated the expression of PRP growth factors and evaluated their potential role in the cartilage regeneration using primary isolated chondrocytes. PRP obtained from New Zealand White rabbit by low speed centrifugation. Extracted PRPs contained 6–10 × 106 platelet/μl and concentration of platelets was slightly variable. Primary isolated chondrocytes from the same rabbits were cultured and treated with 0.1–20% PRP. The cells were collected and examined by reverse transcription-polymerase chain reaction and cytochemical staining. The expression of sex determining region Y-box 9, transforming growth factor-beta, vascular endothelial growth factor, and chondromdulin-I was increased in chondrocyte cultures with 10% PRP by time-dependent manner. To maintain the integrity of the cartilage, the proteoglycan contents were also up-regulated from the mRNA of aggrecan and positive Safranin-O staining in PRP concentration- and time-dependent manner. PRP provides crucial growth factors related to chondrocyte proliferation and differentiation through time-sequential modulation. Controlled in vivo trials for cartilage regeneration are needed.






Similar content being viewed by others
References
Woolf AD, Pfleger B (2003) Burden of major musculoskeletal conditions. Bull World Health Organ 81:646–656
Anitua M, Sanchez E, Nurden A, Nurden P, Orive G, Andia I (2006) New insights into and novel applications for platelet-rich fibrin therapies. Trends Biotechnol 24:227–234
Folkman J, Browder T, Palmblad J (2001) Angiogenesis research: guidelines for translation to clinical application. Thromb Haemost 86:23–33
Pietramaggiori G, Scherer SS, Mathews JC, Gennaoui T, Lancerotto L, Ragno G, Valeri CR, Orgill DP (2010) Quiescent platelets stimulate angiogenesis and diabetic wound repair. J Surg Res 160:169–177
Eppley BL, Pietrzak WS, Blanton M (2006) Platelet-rich plasma: a review of biology and applications in plastic surgery. Plast Reconstr Surg 118:147e–159e
Knighton DR, Ciresi KF, Fiegel VD, Austin LL, Butler EL (1986) Classification and treatment of chronic nonhealing wounds, successful treatment with autologous platelet-derived wound healing factors (PDWHF). Ann Surg 204:322–330
Ma L, Perini R, McKnight W, Dicay M, Klein A, Hollenberg MD, Wallace JL (2005) Proteinase-activated receptors 1 and 4 counter-regulate endostatin and VEGF release from human platelets. Proc Natl Acad Sci USA 102:216–220
Andreas K, Lübke C, Häupl T, Dehne T, Morawietz L, Ringe J, Kaps C, Sittinger M (2008) Key regulatory molecules of cartilage destruction in rheumatoid arthritis: an in vitro study. Arthritis Res Ther 10:R9
Ringe J, Sittinger M (2009) Tissue engineering in the rheumatic diseases. Arthritis Res Ther 11:211
Marx RE (2004) Patelet-rich plasma: evidence to support its use. J Oral Maxillofac Surg 62:489–496
Kim DJ, Moon SH, Kim H, Kwon UH, Park MS, Han KJ, Hahn SB, Lee HM (2003) Bone morphogenetic protein-2 facilitates expression of chondrogenic, not osteogenic, phenotype of human intervertebral disc cells. Spine 28:2679–2684
Thompson JP, Oegema TR Jr, Bradford DS (1991) Stimulation of mature canine intervertebral disc by growth factors. Spine 16:253–260
Versijil N, DeGroot J, Thorpe SR, Bank RA, Shaw JN, Lyons TJ, Bijlsma JW, Lafeber FP, Baynes JW, TeKoppele JM (2000) Effect of collagen turnover on the accumulation of advanced glycation end products. J Biol Chem 275:39027–39031
Paul R, Haydon RC, Cheng H, Ishikawa A, Nenadovich N, Jiang W, Zhou L, Breyer B, Feng T, Gupta P, He TC, Phillips FM (2003) Potential use of Sox9 gene therapy for intervertebral degenerative disc disease. Spine 28:755–763
Kuhne M, John T, El-Sayed K, Marzahn U, Aue A, Kohl B, Stoelzel K, Ertel W, Blottner D, Haisch A, Schulze-Tanzil G (2010) Characterization of auricular chondrocytes and auricular/articular chondrocyte co-cultures in terms of an application in articular cartilage repair. Int J Mol Med 25:701–708
Lefebvre V, Behringer RR, de Crombrugghe B (2001) L-Sox5, Sox6 and Sox9 control essential steps of the chondrocyte differentiation pathway. Osteoarthritis Cartil 9(Suppl A):S69–S75
Aigner T, Gebhard PM, Schmid E, Bau B, Harley V, Pöschl E (2003) SOX9 expression does not correlate with type II collagen expression in adult articular chondrocytes. Matrix Biol 22:363–372
Zhao Q, Eberspaecher H, Lefebvre V, De Crombrugghe B (1997) Parallel expression of Sox9 and Col2a1 in cells undergoing chondrogenesis. Dev Dyn 209:377–386
Rabie AB, Leung FY, Chayanupatkul A, Hagg U (2002) The correlation between neovascularization and bone vormation in the condyle during forward mandibular positioning. Angle Orthod 72:431–438
Rabie AB, Hagg U (2002) Factors regulating mandibular condylar growth. Am J Orthod Dentrofacial Orthop 122:401–409
Hashimoto S, Creighton-Achermann L, Takahashi K, Amiel D, Coutts RD, Lotz M (2002) Development and regulation of osteophyte formation during experimental osteoarthritis. Osteoarthritis Cartil 10:180–187
Bluteau G, Julien M, Magne D, Mallein-Gerin F, Weiss P, Daculsi G, Guicheux J (2007) VEGF and VEGF receptors are differentially expressed in chondrocytes. Bone 40:568–576
Brew CJ, Clegg PD, Boot-Handford RP, Andrew JG, Hardingham T (2010) Gene expression in human chondrocytes in late osteoarthritis is changed in both fibrillated and intact cartilage without evidence of generalised chondrocyte hypertrophy. Ann Rheum Dis 69:234–240
Yee G, Yu U, Walsh WR, Linderman R, Poole MD (2003) The immunolocalisation of VEGF in the articular cartilage of sheep mandibular condyles. J Craniomaxillofac Surg 31:244–251
Emons J, Chagin AS, Malmlöf T, Lekman M, Tivesten A, Ohlsson C, Wit JM, Karperien M, Sävendahl L (2010) Expression of vascular endothelial growth factor in the growth plate is stimulated by estradiol and increases during pubertal development. J Endocrinol 205:61–68
Ashraf S, Walsh DA (2008) Angiogenesis in osteoarthritis. Curr Opin Rheumatol 20:573–580
Kitahara H, Hayami T, Tounga K, Endo N, Funaki H, Yoshida Y, Yaoita E, Yamamoto T (2003) Chondromodulin-1 expression in rat articular cartilage. Arch Histol Cytol 66:221–228
Hiraki Y, Tanaka H, Inoue H, Kondo J, Kamizono A, Suzuki F (1991) Molecular cloning of a new class of cartilage-specific matrix, chondromodulin-1, which stimulates growth of cultured chondrocytes. Biochem Biophys Res Commun 175:971–977
Wtahiki J, Yamaguchi T, Enomoto A, Irie T, Yoshie K, Tachikawa T, Maki K (2008) Identification of differentially expressed genes in mandibular condylar and tibial growth cartilages using laser microdissection and fluorescent differential display: chondromodulin-I (ChM-1) and tenomodulin (TeM) are differentially expressed in mandibular condylar and other growth cartilages. Bone 42:1053–1060
Hiraki Y, Inoue H, Iyama K, Kamizono A, Ochiai M, Shukunami C, Iijima S, Suzuki F, Kondo J (1997) Identification of chondromodulin I as a novel endothelial cell growth inhibitor, purification and its localization in the avascular zone of epiphyseal cartilage. J Biol Chem 272:32419–32426
Shukunami C, Hiraki Y (1998) Expression of cartilage-specific functional matrix chondromodulin-I mRNA in rabbit growth plate chondrocytes and its responsiveness to growth stimuli in vitro. Biochem Biophys Res Commun 249:885–890
Acknowledgment
This study was supported by grants of the Nano-Biotechnology Project (Regenomics), Ministry of Science and Technology, Republic of Korea (B020214) and the National Research Foundation of Korea Grant, Ministry of Education, Science and Technology, Korean Government (NRF-2009-351-E00060).
Author information
Authors and Affiliations
Corresponding author
Additional information
Se-Il Park and Hye-Rim Lee contributed equally to this study.
Rights and permissions
About this article
Cite this article
Park, SI., Lee, HR., Kim, S. et al. Time-sequential modulation in expression of growth factors from platelet-rich plasma (PRP) on the chondrocyte cultures. Mol Cell Biochem 361, 9–17 (2012). https://doi.org/10.1007/s11010-011-1081-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-011-1081-1