Abstract
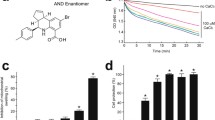
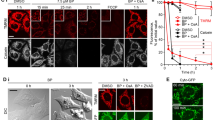
A recent report has described that S-15176 (N-[(3,5-di-tert-butyl-4-hydroxy-1-thiophenyl)]-3-propyl-N′-(2,3,4-trimethoxybenzyl) piperazine), an anti-ischemic agent, inhibits the mitochondrial permeability transition (PT) induced by not only Ca2+ and inorganic phosphate, but also by tert-butylhydroperoxide or phenylarsine oxide [Morin et al. (Biochem Pharmacol 72:911–918, 2006)]. In the present study, we tested the effects of S-15176 on the PT induced by Ag+, PT of which is not suppressed by cyclosporin A or oligomycin. S-15176 was effective in suppressing the PT and the subsequent cytochrome c release induced by Ag+, and hence, it was concluded to be a more universal PT inhibitor than cyclosporin A or oligomycin. In addition to the PT-suppression activity, S-15176 also showed weak protonophoric activity. Thus, we further tested to investigate whether the hydroxyl group of S-15176 was involved in its PT-suppression or weak protonophoric activities. The methylated derivative of S-15176 also showed both PT suppression and weak protonophoric activities; hence, the hydroxyl group of S-15176 was concluded not to be involved in these activities.






Similar content being viewed by others
Abbreviations
- CsA:
-
Cyclosporin A
- PT:
-
Permeability transition
- VDAC:
-
Voltage-dependent anion channel
References
Gunter TE, Pfeiffer DR (1990) Mechanisms by which mitochondria transport calcium. Am J Physiol 258:C755–C786
Zoratti M, Szabo I (1995) The mitochondrial permeability transition. Biochim Biophys Acta 1241:139–176
Bernardi P (1999) Mitochondrial transport of cations: channels, exchangers, and permeability transition. Physiol Rev 79:1127–1155
Fournier N, Ducet G, Crevat A (1987) Action of cyclosporine on mitochondrial calcium fluxes. J Bioenerg Biomembr 19:297–303
Broekemeier KM, Dempsey ME, Pfeiffer DR (1989) Cyclosporin A is a potent inhibitor of the inner membrane permeability transition in liver mitochondria. J Biol Chem 264:7826–7830
Brenner C, Grimm S (2006) The permeability transition pore complex in cancer cell death. Oncogene 25:4744–4756
Kroemer G, Galluzzi L, Brenner C (2007) Mitochondrial membrane permeabilization in cell death. Physiol Rev 87:99–163
Orrenius S, Gogvadze V, Zhivotovsky B (2007) Mitochondrial oxidative stress: implications for cell death. Annu Rev Pharmacol Toxicol 47:143–183
Pfeiffer DR, Gudz TI, Novgorodov SA, Erdahl WL (1995) The peptide mastoparan is a potent facilitator of the mitochondrial permeability transition. J Biol Chem 270:4923–4932
Morin D, Barthélémy S, Zini R, Labidalle S, Tillement JP (2001) Curcumin induces the mitochondrial permeability transition pore mediated by membrane protein thiol oxidation. FEBS Lett 495:131–136
Zhu Y, Xu H, Huang K (2002) Mitochondrial permeability transition and cytochrome c release induced by selenite. J Inorg Biochem 90:43–50
Shinohara Y, Bandou S, Kora S, Kitamura S, Inazumi S, Terada H (1998) Cationic uncouplers of oxidative phosphorylation are inducers of mitochondrial permeability transition. FEBS Lett 428:89–92
Almofti MR, Ichikawa T, Yamashita K, Terada H, Shinohara Y (2003) Silver ion induces a cyclosporine a-insensitive permeability transition in rat liver mitochondria and release of apoptogenic cytochrome C. J Biochem 134:43–49
Yamamoto T, Tachikawa A, Terauchi S, Yamashita K, Kataoka M, Terada H, Shinohara Y (2004) Multiple effects of DiS-C3(5) on mitochondrial structure and function. Eur J Biochem 271:3573–3579
Elimadi A, Jullien V, Tillement JP, Morin D (2003) S-15176 inhibits mitochondrial permeability transition via a mechanism independent of its antioxidant properties. Eur J Pharmacol 468:93–101
Morin D, Zini R, Berdeaux A, Tillement JP (2006) Effect of the mitochondrial transition pore inhibitor, S-15176, on rat liver mitochondria: ATP synthase modulation and mitochondrial uncoupling induction. Biochem Pharmacol 72:911–918
Yamada A, Yamamoto T, Yamazaki N, Yamashita K, Kataoka M, Nagata T, Terada H, Shinohara Y (2009) Differential permeabilization effects of Ca2+ and valinomycin on the inner and outer mitochondrial membranes as revealed by proteomics analysis of proteins released from mitochondria. Mol Cell Proteomics 8:1265–1277
Yamamoto T, Terauchi S, Tachikawa A, Yamashita K, Kataoka M, Terada H, Shinohara Y (2005) Two critical factors affecting the release of mitochondrial cytochrome C as revealed by studies using N, N′-dicyclohexylcarbodiimide as an atypical inducer of permeability transition. J Bioenerg Biomembr 37:299–306
Yamamoto T, Yoshimura Y, Yamada A, Gouda S, Yamashita K, Yamazaki N, Kataoka M, Nagata T, Terada H, Shinohara Y (2008) Distinct behaviors of adenylate kinase and cytochrome c observed following induction of mitochondrial permeability transition by Ca2+ in the absence of respiratory substrate. J Bioenerg Biomembr 40:619–623
Shinohara Y, Almofti MR, Yamamoto T, Ishida T, Kita F, Kanzaki H, Ohnishi M, Yamashita K, Shimizu S, Terada H (2002) Permeability transition-independent release of mitochondrial cytochrome c induced by valinomycin. Eur J Biochem 269:5224–5230
Carbonera D, Azzone GF (1988) Permeability of inner mitochondrial membrane and oxidative stress. Biochim Biophys Acta 943:245–255
Novgorodov SA, Gudz TI, Mohr YE, Goncharenko EN, Yaguzhinsky LS (1989) ATP-synthase complex: the mechanism of control of ion fluxes induced by cumene hydroperoxide in mitochondria. FEBS Lett 247:255–258
Muraoka S, Terada H (1972) 3,5-di-tert-butyl-4-hydroxybenzylidene malononitrile; a new powerful uncoupler of respiratory-chain phosphorylation. Biochim Biophys Acta 275:271–275
Terada H (1981) The interaction of highly active uncouplers with mitochondria. Biochim Biophys Acta 639:225–242
Terada H (1990) Uncouplers of oxidative phosphorylation. Environ Health Perspect 87:213–218
Terada H, Fukui Y, Shinohara Y, Ju-ichi M (1988) Unique action of a modified weakly acidic uncoupler without an acidic group, methylated SF 6847, as an inhibitor of oxidative phosphorylation with no uncoupling activity: possible identity of uncoupler binding protein. Biochim Biophys Acta 933:193–199
Author information
Authors and Affiliations
Corresponding author
Additional information
Satoshi Kawashima, and Takenori Yamamoto contributed equally to this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kawashima, S., Yamamoto, T., Horiuchi, Y. et al. S-15176 and its methylated derivative suppress the CsA-insensitive mitochondrial permeability transition and subsequent cytochrome c release induced by silver ion, and show weak protonophoric activity. Mol Cell Biochem 358, 45–51 (2011). https://doi.org/10.1007/s11010-011-0919-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-011-0919-x