Abstract
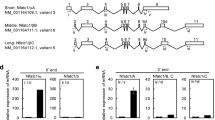
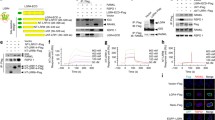
Receptor activator of NF-κB (RANK) is a member of the tumor necrosis factor receptor (TNFR) family expressed in osteoclast precursors, and RANK–RANK ligand (RANKL) signaling is a key system for differentiation, activation and survival of osteoclasts. Here, we report the identification of a novel alternative splicing variant of mouse RANK gene (vRANK) that contains a new intervening exon between exon 1 and exon 2 of mouse full-length RANK (fRANK) mRNA. Since this novel exon contains the stop codon, vRANK encodes truncated amino acids that have a portion of the signal peptide of fRANK and an additional 19 amino acids that show no homology to previously reported domains. By transient transfection studies with vRANK–GFP and –Flag expressing constructs, vRANK was found localized mostly in the cytoplasm and partly in the cell membrane, but was not secreted into the culture supernatant. Under the stimulation of various factors, the expression of vRANK mRNA was almost parallel to that of fRANK in RAW264.7 cells not treated with M-CSF. Overexpression of vRANK, on the other hand, decreased TRACP (a marker of osteoclasts) mRNA expression as well as the number of TRACP-positive multinucleated giant cells. While the mRNA expression levels of NFATc1 (a master transcriptional factor of the osteoclast differentiation program) were not affected, apoptotic cells increased significantly in vRAN K-transfected cells treated with sRANKL. Taken together, these results suggest that vRANK is a novel osteoclast suppressor that reduces the number of RANKL-induced mature osteoclasts mainly by negating the anti-apoptotic effect of RANKL.







Similar content being viewed by others
Abbreviations
- RANK:
-
Receptor activator of NF-κB
- RANKL:
-
Receptor activator of NF-κB (RANK) ligand
- GFP:
-
Green fluorescent protein
- M-CSF:
-
Macrophage colony-stimulating factor
- TRACP:
-
Tartrate-resistant acid phosphatase
- NFAT:
-
Nuclear factor of activated T cells
- OPG:
-
Osteoprotegerin
- TNF:
-
Tumor necrosis factor
- TRAF:
-
Tumor necrosis factor receptor-associated factor
- JNK:
-
c-Jun N-terminal kinase
- ERK:
-
Extracellular signal-regulated kinase
- Akt:
-
AKR mouse T-cell lymphoma
- LPS:
-
Lipopolysaccharide
- MITF:
-
Microphthalmia-associated transcription factor
References
Ross FP, Teitelbaum SL (2005) Alphavbeta3 and macrophage colony-stimulating factor: partners in osteoclast biology. Immunol Rev 208:88–105
Tanaka S, Takahashi N, Udagawa N, Tamura T, Akatsu T, Stanley ER et al (1993) Macrophage colony-stimulating factor is indispensable for both proliferation and differentiation of osteoclast progenitors. J Clin Invest 91:257–263
Teitelbaum SL, Ross FP (2003) Genetic regulation of osteoclast development and function. Nat Rev Genet 4:638–649
Kaji H, Sugimoto T, Kanatani M, Nishiyama K, Chihara K (1997) Dexamethasone stimulates osteoclast-like cell formation by directly acting on hemopoietic blast cells and enhances osteoclast-like cell formation stimulated by parathyroid hormone and prostaglandin E2. J Bone Miner Res 12:734–741
Mizuno A, Kanno T, Hoshi M, Shibata O, Yano K, Fujise N et al (2002) Transgenic mice overexpressing soluble osteoclast differentiation factor (sODF) exhibit severe osteoporosis. J Bone Miner Metab 20:337–344
Tolar J, Teitelbaum SL, Orchard PJ (2004) Osteopetrosis. N Engl J Med 351:2839–2849
Yasuda H, Shima N, Nakagawa N, Yamaguchi K, Kinosaki M, Mochizuki S et al (1998) Osteolast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc Natl Acad Sci USA 95:3597–3602
Lacey DL, Timms E, Tan HL, Kelley MJ, Dunstan CR, Burgess T et al (1998) Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 93:165–176
Anderson DM, Maraskovsky E, Billingsley WL, Dougall WC, Tometsko ME, Roux ER et al (1997) A homologue of the TNF receptor and its ligand enhance T-cell growth and dendritic-cell function. Nature 390:175–179
Liu W, Xu D, Yang H, Xu H, Shi Z, Cao X et al (2004) Functional identification of three receptor Activator of NF-κB cytoplasmic motifs mediating osteoclast differentiation and function. J Biol Chem 279:54759–54769
Boyle WJ, Simonet WS, Lacey DL (2003) Osteoclast differentiation and activation. Nature 423:337–342
Takayanagi H, Kim S, Matsuo K, Suzuki H, Suzuki T, Sato K et al (2002) RANKL maintains bone homeostasis through c-Fos-dependent induction of interferon-beta. Nature 416:744–749
Feng X (2005) Regulatory roles and molecular signaling of TNF fanily members in osteoclasts. Gene 350:1–13
Ikeda F, Nishimura R, Matsubara T, Tanaka S, Inoue J, Reddy SV et al (2004) Critical roles of c-Jun signaling in regulation of NFAT family and RANKL-regulated osteoclast differentiation. J Clin Investig 114:475–484
Day CJ, Kim MS, Stephens SR, Simcock WE, Aitken CJ, Nicholson GC et al (2004) Gene array identification of osteoclast genes: differential inhibition of osteoclastogenesis by cyclosporin A and granulocyte macrophage colony stimulating factor. J Cell Biochem 91:303–315
Matsumoto M, Kogawa M, Wada S, Takayanagi H, Tsujimoto M, Katayama S et al (2004) Essential role of p38 mitogen-activated protein kinase in cathepsin K gene expression during osteoclastogenesis through association of NFATc1 and PU.1. J Biol Chem 279:45969–45979
Karsdal MA, Hjorth P, Henriksen K, Kirkegaard T, Nielsen KL, Lou H et al (2003) Transforming growth factor-β controls human osteoclastogenesis through the p38 MAPK and regulation of RANK expression. J Biol Chem 278:44975–44987
Ishii J, Kitazawa R, Mori K, McHugh KP, Morii E, Kondo T, Kitazawa S (2008) Lipopolysaccharide suppresses RANK gene expression in macrophages by down-regulating PU.1 and MITF. J Cell Biochem 105:896–904
Hakozaki A, Yoda M, Tohmonda T, Furukawa M, Hikata T, Uchikawa S, Takaishi H, Matsumoto M, Chiba K, Horiuchi K, Toyama Y (2010) RANKL induces ectodomain shedding of RANK in murine RAW264.7 macrophages. J Immunol 184:2442–2448
Mironov AA, Fickett JW, Gelafnd MS (1999) Frequent alternative splicing of human genes. Genome Res 9:1288–1293
Croft L, Schandroff S, Clark F, Burrage K, Arctander P, Mattick JS (2000) ISIS, the intron information system, reveals the high frequency of alternative splicing in the human genom. Nat Genet 24:340–341
Brett D, Hanke J, Lehmann G, Haase S, Delbruck S, Krueger S et al (2000) EST comparison indicates 38% of human mRNAs contain possible alternative splice forms. FEBS Lett 474:83–86
Kan Z, Rouchka EC, Gish WR, States DJ (2001) Gene structure prediction and alternative splicing analysis using genomically aligned ESTs. Genome Res 11:889–900
Modrek B, Resch A, Grasso C, Lee C (2001) Genome-wide detection of alternative splicing in expressed sequences of human genes. Nucl Acid Res 29:2850–2859
Johnson JM, Castle J, Garrett-Engele P, Kan Z, Loerch PM, Armour CD et al (2003) Genome-wide survey of human alternative pre-mRNA splicing with exon junction microarrays. Science 302:2141–2144
Kitazawa S, Ross FP, McHugh K, Teitelbaum SL (1995) Interleukin-4 induces expression of the integrin alphavbeta3 via transactivation of the beta3 gene. J Biol Chem 270:4115–4120
Mulder NJ, Apweiler R, Attwood TK, Bairoch A, Barrell D, Bateman A et al (2003) The InterPro Database, 2003 brings increased coverage and new features. Nucl Acids Res 31:315–318
Hirokawa T, Boon-Chieng S, Mitaku S (1998) SOSUI: classification and secondary structure prediction system for membrane proteins. Bioinformatics 14:378–379
Wang C, Steer JH, Joyce DA, Yip KH, Zheng MH, Xu J (2003) 12-O-tetradecanoylphorbol-13-acetate (TPA) inhibits osteoclastogenesis by suppressing RANKL-induced NF-κB activation. J Bone Miner Res 18:2159–2168
Acknowledgments
The authors thank Mr. Shuichi Matsuda and Ms. Noriko Sakamoto for excellent technical assistance. This work was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology, Japan (16590278, 19390100 and 19659086 to S.K., 20790283 to T.K., 18590372 and 21590419 to R.K., and 21790351 to K.M.).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mukai, S., Kitazawa, R., Ishii, J. et al. Identification and analysis of function of a novel splicing variant of mouse receptor activator of NF-κB. Mol Cell Biochem 350, 29–38 (2011). https://doi.org/10.1007/s11010-010-0679-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-010-0679-z