Abstract
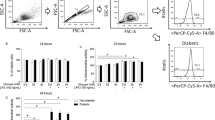
Altered cellular mitochondrial membrane potential (MMP) has been implicated in the increased insulin resistance and the risk for diabetes. Hyperketonemia is increasingly being identified in type 2 diabetic patients in addition to those with type 1 diabetes. No previous study has examined the effect of hyperketonemia and trivalent chromium on cellular mitochondrial membrane potential (MMP) in any cell type. Using a U937 monocyte cell culture model, this study examined the hypothesis that hyperketonemia decreases and trivalent chromium normalizes the cellular MMP level. Cells were cultured with control and ketone bodies [acetoacetate (AA), β-hydroxybutyrate (BHB)] in the absence or the presence (0.5–100 μM) of Cr3+ at 37°C for 24 h. The MMP was determined using DiOC6 and flow cytometry. The results show a significant decrease in MMP in cells treated with AA, but not in the cells treated with BHB. The effect of AA on cellular MMP was prevented in chromium (III)-pretreated cells. Thus, hyperketonemia decreases the MMP, and supplementation with chromium (III) normalizes altered MMP, which may play a role in the improvement in glucose metabolism seen after chromium (III) supplementation in some studies with diabetic animals and patients.



Similar content being viewed by others
References
Kim J-a, Wei Y, Sowers JR (2008) Role of mitochondrial dysfunction in insulin resistance. Circ Res 102(4):401–414
Turner N, Heilbronn LK (2008) Is mitochondrial dysfunction a cause of insulin resistance? Trends Endocrinol Metab 19(9):324–330
Morino K, Petersen Kitt Falk, Dufour Sylvie, Befroy Douglas, Frattini Jared, Shatzkes Nadine, Neschen Susanne, White Morris F, Bilz Stefan, Sono Saki, Pypaert Marc, Shulman Gerald I (2005) Reduced mitochondrial density and increased IRS-1 serine phosphorylation in muscle of insulin-resistant offspring of type 2 diabetic parents. J Clin Invest 115(12):3587–3593
Szendroedi J, Schmid AI, Meyerspeer M, Cervin C, Kacerovsky M, Smekal G, Graser-Lang S, Groop L, Roden M (2009) Impaired mitochondrial function and insulin resistance of skeletal muscle in mitochondrial diabetes. Diabetes Care 32(4):677–679
Mogensen M, Sahlin K, Fernstrom M, Glintborg D, Vind BF, Beck-Nielsen H, Hojlund K (2007) Mitochondrial respiration is decreased in skeletal muscle of patients with type 2 diabetes. Diabetes 56(6):1592–1599
Duchen MR (2004) Roles of mitochondria in health and disease. Diabetes 53(90001):S96–102
Lowell BB, Shulman GI (2005) Mitochondrial dysfunction and type 2 diabetes. Science 307(5708):384–387
Lim J, Lee J, Suh Y, Kim W, Song J, Jung M (2006) Mitochondrial dysfunction induces aberrant insulin signalling and glucose utilisation in murine C2C12 myotube cells. Diabetologia 49(8):1924–1936
Pieczenik SR, Neustadt J (2007) Mitochondrial dysfunction and molecular pathways of disease. Exp Mol Pathol 83(1):84–92
Sirikul B, Gower BA, Hunter GR, Larson-Meyer DE, Newcomer BR (2006) Relationship between insulin sensitivity and in vivo mitochondrial function in skeletal muscle. Am J Physiol Endocrinol Metab 291(4):E724–E728
Anderson RACN, Bryden NA, Polansky MM, Cheng N, Chi J, Feng J (1997) Elevated intakes of supplemental chromium improve glucose and insulin variables in individuals with type 2 diabetes. Diabetes 46:1786–1791
Ding W, Chai Z, Duan P, Feng W, Qian Q (1998) Serum and urine chromium concentrations in elderly diabetics. Biol Trace Elem Res 63(3):231–237
Ekmekcioglu C, Prohaska C, Pomazal K, Steffan I, Schernthaner G, Marktl W (2001) Concentrations of seven trace elements in different hematological matrices in patients with type 2 diabetes as compared to healthy controls. Biol Trace Elem Res 79(3):205–219
Pineau A, Guillard O, Risse J (1992) A study of chromium in human cataractous lenses and whole blood of diabetics, senile, and normal population. Biol Trace Elem Res 32(1):133–138
Cefalu WT, Wang ZQ, Zhang XH, Baldor LC, Russell JC (2002) Oral Chromium picolinate improves carbohydrate and lipid metabolism and enhances skeletal muscle Glut-4 translocation in obese, hyperinsulinemic (JCR-LA Corpulent) Rats. J Nutr 132(6):1107–1114
Lamson D, Plaza S (2002) The safety and efficacy of high-dose chromium. Alternative Medicine Review J Clin Ther 7(3):218–235
Lee NA, Reasner CA (1994) Beneficial effect of chromium supplementation on serum triglyceride levels in NIDDM. Diabetes Care 17(12):1449–1452
Offenbacher EG, Pi-Sunyer FX (1980) Beneficial effect of chromium-rich yeast on glucose tolerance and blood lipids in elderly subjects. Diabetes 28:919–925
Vincent JB (1999) Mechanisms of chromium action: low-molecular-weight chromium-binding substance. J Am Coll Nutr 18(1):6–12
Jeejeebhoy KN (1999) Chromium and parenteral nutrition. J Trace Elem Exp Med 12(2):85–89
Jain SK, Rains JL, Croad JL (2007) Effect of chromium niacinate and chromium picolinate supplementation on lipid peroxidation, TNF-[alpha], IL-6, CRP, glycated hemoglobin, triglycerides, and cholesterol levels in blood of streptozotocin-treated diabetic rats. Free Radic Biol Med 43(8):1124–1131
Candiloros H MS, Zeghari N, Donner M, Drouin P, Ziegler O (1995) Decreased erythrocyte membrane fluidity in poorly controlled IDDM. Influence of ketone bodies. Diabetes Care 18(4):549–551
Newton CA, Raskin P (2004) Diabetic ketoacidosis in type 1 and type 2 diabetes mellitus: clinical and biochemical differences. Arch Intern Med 164(17):1925–1931
Umpierrez G, Kitabchi A (2003) Diabetic ketoacidosis: risk factors and management strategies. Treat Endocrinol 2(2):95–108
Yared Z, Chiasson J (2003) Ketoacidosis and the hyperosmolar hyperglycemic state in adult diabetic patients. Diagnosis and treatment. Minervia Med 94(6):409–418
Jain SK, Kannan K (2001) Chromium chloride inhibits oxidative stress and tnf-[alpha] secretion caused by exposure to high glucose in cultured U937 monocytes. Biochem Biophys Res Commun 289(3):687–691
Kannan K, Jain SK (2004) Effect of vitamin B6 on oxygen radicals, mitochondrial membrane potential, and lipid peroxidation in H2O2-treated U937 monocytes. Free Radic Biol Med 36(4):423–428
Jain SK, Rains JL, Croad JL (2007) High glucose and ketosis (acetoacetate) increases, and chromium niacinate decreases, IL-6, IL-8, and MCP-1 secretion and oxidative stress in U937 monocytes. Antioxid Redox Signal 9(10):1581–1590
Jennifer M, Stephens MJS, Watkins PJ (1971) Relationship of blood acetoacetate and 3-hydroxybutyrate in diabetes. Diabetes 20(7):485–489
Lori L (1999) Ketone bodies: a review of physiology, pathophysiology and application of monitoring to diabetes. Diabetes Metab Res Rev 15(6):412–426
Abdelmegeed MA, Kim SK, Woodcroft KJ, Novak RF (2004) Acetoacetate activation of extracellular signal-regulated kinase 1/2 and p38 mitogen-activated protein kinase in primary cultured rat hepatocytes: role of oxidative stress. J Pharmacol Exp Ther 310(2):728–736
Hoffman WH, Cheng C, Passmore GG, Carroll JE, Hess D (2002) Acetoacetate increases expression of intercellular adhesion molecule-1 (ICAM-1) in human brain microvascular endothelial cells. Neurosci Lett 334(2):71–74
Jain SK, Kannan K, Lim G (1998) Ketosis (acetoacetate) can generate oxygen radicals and cause increased lipid peroxidation and growth inhibition in human endothelial cells. Free Radic Biol Med 25(9):1083–1088
Jain SK, Kannan K, Lim G, Matthews-Greer J, McVie R, Bocchini JA Jr (2003) Elevated blood interleukin-6 levels in hyperketonemic type 1 diabetic patients and secretion by acetoacetate-treated cultured U937 monocytes. Diabetes Care 26(7):2139–2143
Jain SK, Kannan K, Lim G, McVie R, Bocchini JA Jr (2002) Hyperketonemia increases tumor necrosis factor-{alpha} secretion in cultured u937 monocytes and type 1 diabetic patients and is apparently mediated by oxidative stress and cAMP deficiency. Diabetes 51(7):2287–2293
Stentz FB, Umpierrez GE, Cuervo R, Kitabchi AE (2004) Proinflammatory cytokines, markers of cardiovascular risks, oxidative stress, and lipid peroxidation in patients with hyperglycemic crises. Diabetes 53(8):2079–2086
Jain SK, McVie R (1999) Hyperketonemia can increase lipid peroxidation and lower glutathione levels in human erythrocytes in vitro and in type 1 diabetic patients. Diabetes 48(9):1850–1855
Mossop R (1983) Effects of chromium III on fasting blood glucose, cholesterol and cholesterol HDL levels in diabetics. Cent Afr J Med 29(4):80–82
Vincent JB (2004) Recent advances in the nutritional biochemistry of trivalent chromium. Proc Nutr Soc 63(01):41–47
Office of dietary supplements chromium and diabetes workshop summary (1999) http://odsodnihgov/pubs/conferences/chromium_diabetes.html Natcher Conference Center, National Institutes of Health
Vincent JB (2000) Quest for the molecular mechanism of chromium action and its relationship to diabetes. Nutr Rev 58(3):67–72
Jain SK, Croad JL, Velusamy T, Rains JL, Bull R (2010) Chromium dinicocysteinate supplementation can lower blood glucose, CRP, MCP-1, ICAM-1, creatinine, apparently mediated by elevated blood vitamin C and adiponectin and inhibition of NFkB, Akt, and Glut-2 in livers of zucker diabetic fatty rats. Mol Nutr Food Res 54(9):1371–1380
Acknowledgments
The authors are supported by grants from NIDDK and the Office of Dietary Supplements of the National Institutes of Health (RO1 DK072433) and the Malcolm Feist Endowed Chair in Diabetes. The authors thank Ms Georgia Morgan for excellent editing of this manuscript.
Conflict of interest
None of the authors has any financial interest in the publication of this manuscript, nor have they received any money from any other sources except the NIH or LSUHSC.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rains, J.L., Jain, S.K. Hyperketonemia decreases mitochondrial membrane potential and its normalization with chromium (III) supplementation in monocytes. Mol Cell Biochem 349, 77–82 (2011). https://doi.org/10.1007/s11010-010-0662-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-010-0662-8