Abstract
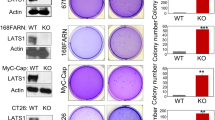
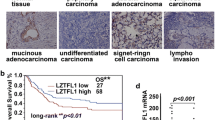
β-catenin is a component of the adhesion complex linking cadherin and actin cytoskeleton, as well as a major mediator of the Wnt pathway, which is a critical signal cascade regulating embryonic development, cell polarity, carcinogenesis, and stem cell function. NF-κB functions as a key regulator of immune responses and apoptosis, and mutations in NF-κB signaling can lead to immune diseases and cancers. We previously showed that NF-κB-mediated modulation of β-catenin/Tcf signaling is mediated by leucine zipper tumor suppressor 2 (Lzts2) and that lzts2 expression is differentially regulated in various cancer cells. Its functional significances, however, are poorly understood. We showed that NF-κB-induced modulation of β-catenin/Tcf pathway is regulated by lzts2 expression in mesenchymal stem cells (MSCs) and several cancer cells, and that NF-κB-induced lzts2 expression is differentially regulated among cancer cell types. Here, using a promoter–reporter assay and EMSA, we demonstrate that NF-κB regulates lzts2 transcription by directly binding to the lzts2 promoter, and that NF-κB-induced lzts2 transcription differs by cell types. Modulation of lzts2 expression by lentiviral techniques affected proliferation and tumorigenicity of several cancer cell lines such as breast, colon, prostate cancer, and glioma, but did not affect cisplatin sensitivity or cell migration. Our data indicate that lzts2 expression is transcriptionally regulated by NF-κB activities, and the modulation of lzts2 expression affects cell proliferation and tumor growth through the Wnt/β-catenin pathway in various cancer cell lines.








Similar content being viewed by others
References
Logan CY, Nusse R (2004) The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol 20:781–810
MacDonald BT, Tamai K, He X (2009) Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell 17:9–26
Bian YS, Osterheld MC, Bosman FT, Fontolliet C, Benhattar J (2000) Nuclear accumulation of beta-catenin is a common and early event during neoplastic progression of Barrett esophagus. Am J Clin Pathol 114:583–590
Eberhart CG, Tihan T, Burger PC (2000) Nuclear localization and mutation of beta-catenin in medulloblastomas. J Neuropathol Exp Neurol 59:333–337
Li Q, Verma IM (2002) NF-κB regulation in the immune system. Nat Rev Immunol 2:725–734
Lin A, Karin M (2003) NF-κB in cancer: a marked target. Semin Cancer Biol 13:107–114
Mayo MW, Baldwin AS (2000) The transcription factor NF-κB: control of oncogenesis and cancer therapy resistance. Biochim Biophys Acta 1470:M55–M62
Prasad S, Ravindran J, Aggarwal BB (2010) NF-kappaB and cancer: how intimate is this relationship. Mol Cell Biochem 336:25–37
Chang AA, Van Waes C (2005) Nuclear factor-kappaB as a common target and activator of oncogenes in head and neck squamous cell carcinoma. Adv Otorhinolaryngol 62:92–102
Pepper C, Hewamana S, Brennan P, Fegan C (2009) NF-kappaB as a prognostic marker and therapeutic target in chronic lymphocytic leukemia. Future Oncol 5:1027–1237
Deng J et al (2002) Beta-catenin interacts with and inhibits NF-kappa B in human colon and breast cancer. Cancer Cell 2:323–334
Polakis P (2000) Wnt signaling and cancer. Genes Dev 14:1837–1851
Hoeflich KP, Luo J, Rubie EA, Tsao MS, Jin O, Woodgett JR (2000) Requirement for glycogen synthase kinase-3beta in cell survival and NF-kappaB activation. Nature 406:86–90
Wang X, Adhikari N, Li Q, Guan Z, Hall JL (2004) The role of [beta]-transducin repeat containing protein ([beta]-TrCP) in the regulation of NF-[kappa]B in vascular smooth muscle cells. Arterioscler Thromb Vasc 24:85–90
Cho HH, Joo HJ, Song JS, Bae YC, Jung JS (2007) Crossregulation of β-catenin/Tcf pathway by NF-κB is mediated by lzts2 in human adipose tissue-derived mesenchymal stem cells. Biochim Biophys Acta 26:8857–8867
Thyssen G, Li TH, Lehmann L, Zhuo M, Sharma M, Sun Z (2006) LZTS2 is a novel beta-catenin-interacting protein and regulates the nuclear export of beta-catenin. Mol Cell Biol 26:8857–8867
Cho HH, Song JS, Yu JM, Yu SS, Choi SJ, Kim DH, Jung JS (2008) Differential effect of NF-kappaB activity on beta-catenin/Tcf pathway in various cancer cells. FEBS Lett 582:616–622
Bours V, Bentires-Alj M, Hellin AC, Viatour P, Robe P, Delhalle S, Benoit V, Merville MP (2000) Nuclear factor-kappa B, cancer, and apoptosis. Biochem Pharmacol 60:1085–1089
Zhu J, He F, Hu S, Yu J (2008) On the nature of human housekeeping genes. Trends Genet 24:481–484
Goodrich JA, Cutler G, Tjian R (1996) Contacts in context: promoter specificity and macromolecular interactions in transcription. Cell 84:825–830
Janknecht R, Hunter T (1996) Transcription. A growing coactivator network. Nature 383:22–23
Sun H, Chung WC, Ryu SH, Ju Z, Tran HT, Kim E, Kurie JM, Koo JS (2008) Cyclic AMP-responsive element binding protein- and nuclear factor-{kappa}B-regulated CXC chemokine gene expression in lung carcinogenesis. Cancer Prev Res 1:316–328
Yabe T, Sanagi T, Schwartz JP, Yamada H (2005) Pigment epithelium-derived factor induces pro-inflammatory genes in neonatal astrocytes through activation of NF-kappa B and CREB. Glia 50:223–234
Parry GC, Mackman N (1997) Role of cyclic AMP response element-binding protein in cyclic AMP inhibition of NF-kappaB-mediated transcription. J Immunol 159:5450–5456
Delgado M, Munoz-Elias EJ, Kan Y, Gozes I, Fridkin M, Brenneman DE, Gomariz RP, Ganea D (1998) Vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide inhibit tumor necrosis factor alpha transcriptional activation by regulating nuclear factor-kB and cAMP response element-binding protein/c-Jun. J Biol Chem 273:31427–31436
López-Knowles E, Zardawi SJ, McNeil CM, Millar EK, Crea P, Musgrove EA, Sutherland RL, O’Toole SA (2010) Cytoplasmic localization of beta-catenin is a marker of poor outcome in breast cancer patients. Cancer Epidemiol Biomark Prev 19:301–309
Sethi G, Sung B, Aggarwal BB (2008) Nuclear factor-kappaB activation: from bench to bedside. Exp Biol Med (Maywood) 233:21–31
Nakanishi C, Toi M (2005) Nuclear factor-kappaB inhibitors as sensitizers to anticancer drugs. Nat Rev Cancer 5:297–309
Wang CY, Mayo MW, Baldwin AS Jr (1996) TNF- and cancer therapy-induced apoptosis: potentiation by inhibition of NF-kappaB. Science 274:784–787
Ji D, Deeds SL, Weinstein EJ (2007) A screen of shRNAs targeting tumor suppressor genes to identify factors involved in A549 paclitaxel sensitivity. Oncol Rep 18:1499–1505
Saldanha G, Ghura V, Potter L, Fletcher A (2004) Nuclear β-catenin in basal cell carcinoma correlates with increased proliferation. Br J Dermatol 151:157–164
Acknowledgments
This study was supported by a grant (20080463000) of National Research Foundation (NRF).
Author information
Authors and Affiliations
Corresponding author
Additional information
Jong Myung Kim, Ji Sun Song are the two authors contributed equally to this study.
An erratum to this article can be found at http://dx.doi.org/10.1007/s11010-011-1177-7
Rights and permissions
About this article
Cite this article
Kim, J.M., Song, J.S., Cho, H.H. et al. Effect of the modulation of leucine zipper tumor suppressor 2 expression on proliferation of various cancer cells functions as a tumor suppressor. Mol Cell Biochem 346, 125–136 (2011). https://doi.org/10.1007/s11010-010-0599-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-010-0599-y