Abstract
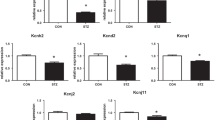
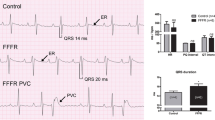
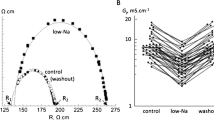
Previous studies in isolated perfused heart and in atrial preparations have demonstrated significant reductions in beating rate in STZ-induced diabetic rats, which suggests that sinus arrhythmias in diabetes mellitus may be partly caused by intrinsic alteration of sino-atrial node (SAN) function. The effects of diabetes on electrical activity and expression levels of mRNA for gap junction proteins in the SAN have been investigated. Diabetes was induced by a single intraperitoneal injection of STZ (60 mg/kg) administered to young male Wistar rats (200–250 g). Experiments were performed 8–10 weeks after treatment. Conduction time and pacemaker cycle length were measured in sino-atrial node preparations with extracellular electrodes. Expression levels of mRNA for Gja5 (C×40), Gja1 (C×43) and Gja7 (C×45) were measured in SAN and compared with right atrium and right ventricle with real-time quantitative reverse transcription-polymerase chain reaction. Diabetes was confirmed by a significant elevation of blood glucose (356 ± 21 mg/dl) compared to age-matched controls (66 ± 2 mg/dl). Pacemaker cycle length was significantly prolonged in diabetic heart (415 ± 43 ms, n = 6) compared to controls (255 ± 7 ms, n = 6). Sino-atrial conduction time was also significantly prolonged in diabetic hearts (12 ± 2 ms) compared to controls (7 ± 1 ms). Expression levels of mRNA for Gja5 (C×40) and Gja1 (C×43) were moderately increased and for Gja7 (C×45) was significantly increased in SAN from diabetic heart compared to controls. Expression levels for gap junction connexin proteins were not significantly altered in right atrium or right ventricle from diabetic heart compared to controls. Structural remodelling of gap junction connexin proteins may partly underlie electrophysiological defects in STZ-induced diabetic rat SAN.


Similar content being viewed by others
References
Veglio M, Chinaglia A, Cavallo PP (2000) The clinical utility of QT interval assessment in diabetes. Diabetes Nutr Metab 13:356–365
Park HW, Cho JG, Yum JH, Hong YJ, Lim JH, Kim HG, Kim JH, Weon-Kim, Ahn YK, Jeong MH, Park JC, Kang JC (2004) Clinical characteristics of hypervagotonic sinus node dysfunction. Korean J Intern Med 19:155–159
Podlaha R, Falk A (1992) The prevalence of diabetes mellitus and other risk factors of atherosclerosis in bradycardia requiring pacemaker treatment. Horm Metab Res Suppl 26:84–87
Linnemann B, Janka HU (2003) Prolonged QTc interval and elevated heart rate identify the type 2 diabetic patient at high risk for cardiovascular death. The Bremen diabetes study. Exp Clin Endocrinol Diabetes 111:215–222
Howarth FC, Jacobson M, Shafiullah M, Adeghate E (2006) Effects of insulin treatment on heart rhythm, body temperature and physical activity in streptozotocin-induced diabetic rat. Clin Exp Pharmacol Physiol 33:327–331
Howarth FC, Jacobson M, Shafiullah M, Adeghate E (2005) Long-term effects of streptozotocin-induced diabetes on the electrocardiogram, physical activity and body temperature in rats. Exp Physiol 90:827–835
Hicks KK, Seifen E, Stimers JR, Kennedy RH (1998) Effects of streptozotocin-induced diabetes on heart rate, blood pressure and cardiac autonomic nervous control. J Auton Nerv Syst 69:21–30
Howarth FC, Qureshi MA (2006) Effects of carbenoxolone on heart rhythm, contractility and intracellular calcium in streptozotocin-induced diabetic rat. Mol Cell Biochem 289:21–29
Kofo-Abayomi A, Lucas PD (1988) A comparison between atria from control and streptozotocin-diabetic rats: the effects of dietary myoinositol. Br J Pharmacol 93:3–8
Betsuyaku T, Nnebe NS, Sundset R, Patibandla S, Krueger CM, Yamada KA (2006) Overexpression of cardiac connexin45 increases susceptibility to ventricular tachyarrhythmias in vivo. Am J Physiol 290:H163-H171
Yamada KA, Rogers JG, Sundset R, Steinberg TH, Saffitz JE (2003) Up-regulation of connexin45 in heart failure. J Cardiovasc Electrophysiol 14:1205–1212
Yamamoto M, Honjo H, Niwa R, Kodama I (1998) Low-frequency extracellular potentials recorded from the sinoatrial node. Cardiovasc Res 39:360–372
Lei M, Jones SA, Liu J, Lancaster MK, Fung SS, Dobrzynski H, Camelliti P, Maier SK, Noble D, Boyett MR (2004) Requirement of neuronal- and cardiac-type sodium channels for murine sinoatrial node pacemaking. J Physiol 559:835–848
Marionneau C, Couette B, Liu J, Li H, Mangoni ME, Nargeot J, Lei M, Escande D, Demolombe S (2005) Specific pattern of ionic channel gene expression associated with pacemaker activity in the mouse heart. J Physiol 562:223–234
Loganathan R, Bilgen M, Al Hafez B, Smirnova IV (2006) Characterization of alterations in diabetic myocardial tissue using high resolution MRI. Int J Cardiovasc Imaging 22:81–90
Ruzicska E, Foldes G, Lako-Futo Z, Sarman B, Wellmann J, Szenasi G, Tulassay Z, Ruskoaho H, Toth M, Somogyi A (2004) Cardiac gene expression of natriuretic substances is altered in streptozotocin-induced diabetes during angiotensin II-induced pressure overload. J Hypertens 22:1191–1200
Grimm D, Jabusch HC, Kossmehl P, Huber M, Fredersdorf S, Griese DP, Kramer BK, Kromer EP (2002) Experimental diabetes and left ventricular hypertrophy: effects of beta-receptor blockade. Cardiovasc Pathol 11:229–237
Stadler K, Jenei V, Somogyi A, Jakus J (2004) Aminoguanidine prevents peroxynitrite production and cardiac hypertrophy in streptozotocin-induced diabetic rats. Orv Hetil 145:2491–2496
Hajinazarian M, Cosio FG, Nahman NSJ, Mahan JD (1994) Angiotensin-converting enzyme inhibition partially prevents diabetic organomegaly. Am J Kidney Dis 23:105–117
Borges GR, de Oliveira M, Salgado HC, Fazan R Jr (2006) Myocardial performance in conscious streptozotocin diabetic rats. Cardiovasc Diabetol 5:26
Zhong Y, Ahmed S, Grupp IL, Matlib MA (2001) Altered SR protein expression associated with contractile dysfunction in diabetic rat hearts. Am J Physiol 281:H1137–H1147
Tamada A, Hattori Y, Houzen H, Yamada Y, Sakuma I, Kitabatake A, Kanoo M (1998) Effects of beta-adrenoceptor stimulation on contractility, [Ca2+](i), and Ca2+ current in diabetic rat cardiomyocytes. Am J Physiol 43:H1849–H1857
Kreuzberg MM, Schrickel JW, Ghanem A, Kim JS, Degen J, Janssen-Bienhold U, Lewalter T, Tiemann K, Willecke K (2006) Connexin30.2 containing gap junction channels decelerate impulse propagation through the atrioventricular node. Proc Natl Acad Sci USA 103:5959–5964
Goodenough DA, Goliger JA, Paul DL (1996) Connexins, connexons, and intercellular communication. Annu Rev Biochem 65:475–502
John S, Cesario D, Weiss JN (2003) Gap junctional hemichannels in the heart. Acta Physiol Scand 179:23–31
Nicholson BJ, Weber PA, Cao F, Chang H, Lampe P, Goldberg G (2000) The molecular basis of selective permeability of connexins is complex and includes both size and charge. Braz J Med Biol Res 33:369–378
Muller DJ, Hand GM, Engel A, Sosinsky GE (2002) Conformational changes in surface structures of isolated connexin 26 gap junctions. EMBO J 21:3598–3607
John SA, Kondo R, Wang SY, Goldhaber JI, Weiss JN (1999) Connexin-43 hemichannels opened by metabolic inhibition. J Biol Chem 274:236–240
Lampe PD, Cooper CD, King TJ, Burt JM (2006) Analysis of Connexin43 phosphorylated at S325, S328 and S330 in normoxic and ischemic heart. J Cell Sci 119:3435–3442
Bagwe S, Berenfeld O, Vaidya D, Morley GE, Jalife J (2005) Altered right atrial excitation and propagation in connexin40 knockout mice. Circulation 112:2245–2253
Danik SB, Liu F, Zhang J, Suk HJ, Morley GE, Fishman GI, Gutstein DE (2004) Modulation of cardiac gap junction expression and arrhythmic susceptibility. Circ Res 95:1035–1041
van Rijen HV, Eckardt D, Degen J, Theis M, Ott T, Willecke K, Jongsma HJ, Opthof T, de Bakker JM (2004) Slow conduction and enhanced anisotropy increase the propensity for ventricular tachyarrhythmias in adult mice with induced deletion of connexin43. Circulation 109:1048–1055
Lee PJ, Pogwizd SM (2006) Micropatterns of propagation. Adv Cardiol 42:86–106
Gollob MH (2006) Cardiac connexins as candidate genes for idiopathic atrial fibrillation. Curr Opin Cardiol 21:155–158
Boyett MR, Honjo H, Kodama I (2000) The sinoatrial node, a heterogeneous pacemaker structure. Cardiovasc Res 47:658–687
Boyett MR, Inada S, Yoo S, Li J, Liu J, Tellez J, Greener ID, Honjo H, Billeter R, Lei M, Zhang H, Efimov IR, Dobrzynski H (2006) Connexins in the sinoatrial and atrioventricular nodes. Adv Cardiol 42:175–197
Nishii K, Kumai M, Egashira K, Miwa T, Hashizume K, Miyano Y, Shibata Y (2003) Mice lacking connexin45 conditionally in cardiac myocytes display embryonic lethality similar to that of germline knockout mice without endocardial cushion defect. Cell Commun Adhes 10:365–369
Egashira K, Nishii K, Nakamura K, Kumai M, Morimoto S, Shibata Y (2004) Conduction abnormality in gap junction protein connexin45-deficient embryonic stem cell-derived cardiac myocytes. Anat Rec A Discov Mol Cell Evol Biol 280:973–979
Acknowledgements
This work was funded by grants from the UAE University (Chris Howarth), Welcome Trust (Ming Lei) and by a travel bursary from the British Council (Chris Howarth), Abu Dhabi, UAE.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Howarth, F.C., Nowotny, N., Zilahi, E. et al. Altered expression of gap junction connexin proteins may partly underlie heart rhythm disturbances in the streptozotocin-induced diabetic rat heart. Mol Cell Biochem 305, 145–151 (2007). https://doi.org/10.1007/s11010-007-9537-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-007-9537-z