Abstract
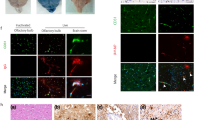
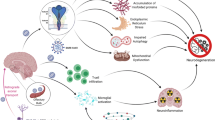
The neuropathogenesis of influenza-associated encephalopathy in children and Reye's syndrome remains unclear. A surveillance effort conducted during 2000-2003 in South-West Japan reveals that almost all fatal and handicapped influenza-associated encephalopathy patients exhibit a disorder of mitochondrial β-oxidation with elevated serum acylcarnitine ratios (C16:0+C18:1)/C2. Here we show invasion by a non-neurotropic epidemic influenza A H3N2 virus in cerebral capillaries with progressive brain edema after intranasal infection of mice having impaired mitochondrial β-oxidation congenitally or posteriorly in the newborn/ suckling periods. Mice genetically lacking of carnitine transporter OCTN2, resulting in carnitine deficiency and impaired β-oxidation, exhibited significant higher virus-genome numbers in the brain, accumulation of virus antigen exclusively in the cerebral capillaries and increased brain vascular permeability compared to in wild type mice. Mini-plasmin, which proteolytically potentiates influenza virus multiplication in vivo and destroys the blood-brain barrier, accumulated with virus antigen in the brain capillaries of OCTN2-deficient mice but only a little in wild-type mice. These results suggest that the impaired mitochondrial β-oxidation changes the susceptibility to a non-neurotropic influenza A virus as to multiplication in the brain capillaries and to cause brain edema. These pathological findings in the brain of mice having impaired mitochondrial β-oxidation after influenza virus infection may have implications for human influenza-associated encephalopathy.
Similar content being viewed by others
References
Kim HW, Brandt CD, Arrobio JO, Murphy B, Chanock RM, Parrott RH: Influenza A and B virus infection in infants and young children during the years 1957–1976. Am J Epidemiol 109: 464–479, 1979
Fujimoto S, Kobayashi M, Uemura O, Iwasa M, Ando T, Katoh T, Nakamura C, Maki N, Togari H, Wada Y: PCR on cerebrospinal fluid to show influenza-associated acute encephalopathy or encephalitis. Lancet 352: 873–875, 1998
Davis LE, Kornfeld M, Daniels RS, Skehel JJ: Experimental influenza causes a non-permissive viral infection of brain, liver and muscle. J Neurovirol 6: 529–536, 2000
Kilbourne ED: Influenza. Plenum Press: New York, pp. 157–218, 1987
Dolorme L, Middleton PJ: Influenza A virus associated with acute encephalopathy. Am J Dis Child 133: 822–824, 1979
Menninger KA: Influenza and schizophrenia. An analysis of post-influenza “dementia precox” as of 1918 and five years later. Am J Psychiatr 5: 469–529, 1926
Ward AC: Neurovirulence of influenza A virus. J Neurovirol 2: 139–151, 1996
Reye RDK, Morgan G, Baral J: Encephalopathy and fatty degeneration of the viscera: a disease entity in childhood. Lancet 2: 749–752, 1963
Rogers MF, Schonberger LB, Hurwitz ES, Rowley DL: National Reye syndrome surveillance, 1982. Pediatics 75: 260–264, 1985
Remington PL, Rowley D, McGee H, Hall WN, Monto AS: Decreasing trends in Reye Syndrome and aspirin use in Michigan, 1979 to 1984. Pediatics 77: 93–98, 1986
Klenk HD, Garten W: Host cell processing proteases controlling virus pathogenicity. Trends Microbiol 2: 39–42, 1994
Kido H, Yokogoshi Y, Sakai K, Rashiro M, Kishino Y, Fukutomi A, Katunuma N: Isolation and characterization of a novel trypsin-like protease found in rat bronchiolar epithelial Clara cells. A possible activator of the viral fusion glycoprotein. J Biol Chem 267: 13573–13579, 1992
Towatari T, Ide M, Ohba K, Chiba Y, Murakami M, Shiota M, Kawachi M, Yamada H, Kido H: Identification of ectopic anionic trypsin I in rat lungs potentiating pneumotropic virus infectivity and increased enzyme level after virus infection. Eur J Biochem 269: 1–9, 2002
Murakami M, Towatari T, Ohuchi M, Shiota M, Akao M, Okumura Y, Parry MAA, Kido H: Mini-plasmin found in the epithelial cells of bronchioles triggers infection by broad-spectrum influenza A viruses and Sendai virus. Eur J Biochem 268: 2847–2855, 2001
Kido H, Murakami M, Oba K, Chen Y, Towatari T: Cellular proteinases trigger the infectivity of influenza A and Sendai viruses. Mol Cells 9: 235–244, 1999
Morishima T, Togashi T, Yokota S, Okuno Y, Miyazaki C, Tashiro M, Okabe N: Encephalitis and encephalopathy associated with an influenza epidemic in Japan. Clin Infect Dis 35: 512–517, 2000
Yokota S, Imagawa T, Miyamae, Ito S, Nakajima S, Nezu A, Mori M: Hypothetical pathophysiology of acute encephalopathy and encephalitis related to influenza virus infection and hypothermia therapy. Pedatr Int 42: 197–203, 2000
Smith TCG: Reye's syndrome and the use of aspirin. Scott Med J 41: 409, 1996
Clark I, Whitten R, Molyneux M, Taylor T: Salicylates, nitric oxide, malaria, and Reye's syndrome. Lancet 357: 625–627, 2001
Murphy MG, Crocker JFS, Her H: Abnormalities in hepatic fatty-acid metabolism in a surfactant/influenza B virus mouse model for acute encephalopathy. Biochim Biophys Acta 1315: 208–216, 1996
Crocker JFS, Digout SC, Lee SH, Rozee KR, Renton K, Field CA, Acott P, Murphy MG: Effect of antipyretics on mortality due to influenza B virus in a mouse model of Reye's syndrome. Clin Invest Med 21: 192–202, 1998
Visentin M, Bellasio R, Tacconi MT: Reye syndrome model in rats: protection against liver abnormalities by L-carnitine and acetyl-L-carnitine. J Pharmacol Exp Ther 275: 1069–1075, 1995
Deschamps D, Fisch C, Fromenty B, Berson A, Degott C, Pessayre D: Inhibition by salicylic acid of the activation and thus oxidation of long chain fatty acids, possible role in the development of Reye's syndrome. J Pharmacol Exp Ther 259: 894–904, 1991
Trauner DA, Horvath E, Davis LE: Inhibition of fatty acid beta oxidation by influenza B virus and salicylic acid in mice: implications for Reye's syndrome. Neurology 38: 239–241, 1988
Kerner J, Hoppel C: Genetic disorders of carnitine metabolism and their nutritional management. Annu Rev Nutr 18: 179–206, 1998
Pons R, De Vivo DC: Primary and secondary carnitine deficiency syndrome. J Chil Neurol 10(Suppl 2): 2S8–2S24, 1995
Okita K, Tokino T, Nishimori H, Miura K, Nikaido H, Hayakawa J, Ono A, Kuwajima M, Matsuzawa Y, Nakamura Y: Definition of the locus responsible for systemic carnitine deficiency within a 1.6-cm region of mouse chromosome 11 by detailed linkage analysis. Genomics 33: 289–291, 1996
Kuwajima M, Harashima H, Hayashi M, Ise S, Sei M, Lu KM, Kiwada H, Sugiyama Y, Shima K: Pharmacokinetic analysis of the cardioprotective effect of 3-(2,2,2-trimethylhydrazinium) propionate in mice: inhibition of carnitine transport in kidney. J Pharmacol Exp Ther 289: 93–102, 1999
Kuwajima M, Kono N, Horiuchi M, Imamura Y, Ono A, Inui Y, Kawata S, Koizumi T, Hayakawa J, Saheki T, Tarui S: Animal model of systemic carnitine deficiency: analysis in C3H-H2∘ strain of mouse associated with juvenile visceral steatosis. Biochem Biophy Res Commun 174: 1090–1094, 1991
Gething MJ, Bye J, Skehel J, Waterfield M: Cloning and DNA sequence of double-stranded copies of haemagglutinin genes from H2 and H3 strains elucidates antigenic shift and drift in human influenza virus. Nature 287: 301–306, 1980
Butler SL, Hansen MST, Bushman FD: A quantitative assay for HIV DNA integration in vivo. Nature Med 7: 631–634, 2001
Kakinuma Y, Hama H, Sugiyama F, Yagami KI, Goto K, Murakami K, Fukamizu A: Impaired blood-brain barrier function in angiotensinogen-deficient mice. Nature Med 4: 1078–1080, 1998
Chaturvedi UC, Dhawan R, Khanna M, Mathur A. Breakdown of the blood-brain barrier during dengue virus infection of mice. J Gen Virol 72: 859–866, 1991
Tamai I, Ohashi R, Nezu J, Sai Y, Kobayashi D, Oku A, Shimane M, Tsuj A: Molecular and functional characterization of organic cation/carnitine transporter family in mice. J Biol Chem 275: 40064–40072, 2000
Nezu JI, Tamai I, Oku A, Ohashi R, Yabuuchi H, Hashimoto N, Nikaido H, Sai Y, Koizumi A, Shoji Y, Takada G, Matsuishi T, Yoshino M, Kato H, Ohura T, Tsujimoto G, Hayakawa J, Shimane M, Tsuji, A: Primary systemic carnitine deficiency is caused by mutations in a gene encoding sodium ion-dependent carnitine transporter. Nature Genet 21: 91–94, 1999
Togashi T, Matsuzono Y, Narita M, Morishima T: Influenza-associated acute encephalopathy in Japanese children in 1994–2002. Virus Res 103: 75–78, 2004
Nagy Z, Kolev K, Csonka E, Vastag M, Machovic R: Perturbation of the integrity of the blood-brain barrier by fibrinolytic enzymes. Blood Coag. Fibrinol. 1: 79–90, 1990
Suzuki Y: Gangliosides as influenza virus receptors. Variation of influenza viruses and their recognition of the receptor sialo-sugar chains. Prog Lipid Res 33: 429–457, 1994
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yao, D., Kuwajima, M., Chen, Y. et al. Impaired long-chain fatty acid metabolism in mitochondria causes brain vascular invasion by a non-neurotropic epidemic influenza A virus in the newborn/suckling period: implications for influenza-associated encephalopathy. Mol Cell Biochem 299, 85–92 (2007). https://doi.org/10.1007/s11010-005-9046-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-005-9046-x