Abstract
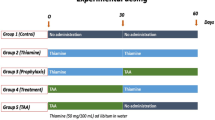
Serine dehydratase (SerDH) is a gluconeogenic enzyme involved in the catabolism of serine, which is regulated by the composition of their diet and their hormonal status in rats. This study examines how chronic injury caused to the liver of rats by the ingestion of thioacetamide (TAA) affects SerDH protein, mRNA levels, enzyme kinetics and its tissue location. After 97 days’ oral intake of TAA, the activity of SerDH at all substrate concentrations assayed was about 60% lower than in controls. No significant differences in Km values were found between the treated group and controls. Immunoblotting and immunohistochemistry revealed a significant reduction in the level of SerDH protein in the livers of the treated rats. SerDH was detected specifically in the periportal zone of the hepatic acinus and this location did not change in response to TAA treatment. The level of SerDH mRNA, quantified by reverse transcription and polymerase chain reaction, was significantly lower in treated rats than in the controls. The present findings suggest that the SerDH expression is rendered to be down regulatory during chronic liver injury induced by TAA. These results enhance our understanding about the biochemical mechanisms implied in the control and integration of serine catabolism during liver injury in rat. (Mol Cell Biochem 268: 33–43, 2005)
Similar content being viewed by others
Abbreviations
- SerDH:
-
serine dehydratase
- TAA:
-
thioacetamide
- Km:
-
Michaelis constant
- cAMP:
-
3′,5′-cyclic adenosine monophosphate
- PLP:
-
pyridoxal 5′-phosphate
- ASAT:
-
aspartate aminotransferase
- ALAT:
-
alanine aminotransferase
- AP:
-
alkaline phosphatase
- γ-GT:
-
gamma-glutamyl transferase
- rGST-7-7:
-
glutathione transferase isoenzyme 7-7
- Vmax:
-
maximum velocity
- SDS-PAGE:
-
polyacrylamide-gel electrophoresis with sodium dodecyl sulphate
- TBS:
-
Tris buffer solution
- PBS:
-
phosphate buffer solution
- RT-PCR:
-
reverse transcription and polymerase chain reaction
- dNTP:
-
deoxynucleotide triphosphate
- AMV-RT:
-
avian-myeloblastosis virus reverse transcriptase
- cDNA:
-
complementary DNA
- Tm:
-
melting temperature
- SDH3:
-
SerDH cDNA probe
- S.E.M.:
-
standard error of the mean
- bp:
-
base pair(s)
References
Snell K: Mitochondrial-cytosolic interrelationships involved in gluconeogenesis from serine in rat liver. FEBS Lett 55: 202–205, 1975
Ogawa H, Kawamata S: Periportal expression of the serine dehydratase gene in rat liver. Histochem J 27: 380–387, 1995
Inoue H, Kasper CB, Pitot HC: Studies of the induction and repression of enzymes in rat liver. VI. Some properties and the metabolic regulation of two isozymic forms of serine dehydratase. J Biol Chem 246: 2626–2632, 1971
Snell K: Enzymes of serine metabolism in normal, developing and neoplastic rat tissues. Adv Enzyme Regul 22: 325–400, 1984
Pitot HC, Peraino C: Studies on the induction and repression of enzymes in rat liver. I. Induction of threonine dehydratase and ornithine transaminase by oral intubation of casein hydrolysate. J Biol Chem 239: 1783–1788, 1964
Ishikawa E, Ninagawa T, Suda M: Hormonal and dietary control of serine dehydratase in rat. J Biochem (Tokyo) 57: 506–513, 1965
Jost JP, Khairallah EA, Pitot HC: Studies on the induction and repression of enzymes in rat liver. V. Regulation of the rate of synthesis and degradation of serine dehydratase by dietary amino acids and glucose. J Biol Chem 243: 3057–3066, 1968
Kanamoto R, Su Y, Pitot HC: Effects of glucose, insulin and cAMP on transcription of the serine dehydratase gene in rat liver. Arch Biochem Biophys 288: 562–566, 1991
Ogawa H, Fujioka M, Su Y, Kanamoto R, Pitot HC: Nutritional regulation and tissue-specific expression of the serine dehydratase gene in rat. J Biol Chem 266: 20412–20417, 1991
Haas MJ, Pitot HC: Glucocorticoids stimulate CREB binding to a cyclic-AMP response element in the rat serine dehydratase gene. Arch Biochem Biophys 362: 317–324, 1999
Imai S, Yagi I, Saeki T, Kotaru M, Iwami K: Quantity as well as quality of dietary protein affects serine dehydratase gene expression in rat liver. J Nutr Sci Vitaminol 49: 33–39, 2003
Jean C, Rome S, Mathe V, Huneau JF, Aattouri N, Fromentin G, Achagiotis CL, Tome D: Metabolic evidence for adaptation to a high protein diet in rats. J Nutr 131: 91–98, 2001
Koning TJ, Snell K, Duran M, Berger R, Poll-The B-T, Surtees R: L-Serine in disease and development. Biochem J 371: 653–661, 2003
Sugimoto S, Mitaka T, Ikeda S, Harada K, Ikai I, Yamaoka Y, Mochizuki Y: Morphological changes induced by extracellular matrix are correlated with maturation of rat small hepatocytes. J Cell Biochem 87: 16–28, 2002
Korekane H, Nishikawa A, Imamura K: Mechanisms mediating metabolic abnormalities in the livers of Ehrlich ascites tumor-bearing mice. Arch Biochem Biophys 412: 216–222, 2003
Lautz HU, Selberg O, Körber J, Bürger M, Müller MJ: Protein-calorie malnutrition in liver cirrhosis. Clin Invest 70: 478–486, 1992
Bugianesi E, Kalhan S, Burkett E, Marchesini G, McCullough A: Quantification of gluconeogenesis in cirrhosis: Response to glucagon. Gastroenterol 115: 1530–1540, 1998
Krahenbuhl S, Reichen J: Decreased hepatic glucose production in rats with carbon tetrachloride-induced cirrhosis. J Hepatol 19: 64–70, 1993
Xue HH, Fujie M, Sakaguchi T, Oda T, Ogawa H, Kneer NM, Lardy HA, Ichiyama A: Flux of the l-serine metabolism in rat liver. J Biol Chem 274: 16020–16027, 1999
Hunter AL, Holschen MA, Neal RA: Thioacetamide-induced hepatic necrosis. I. Involvement of the mixed function oxidase system. J Pharmacol Exp Ther 200: 439–448, 1977
Dasgupta A, Chatterjee R, Chowdhury JR: Thioacetamide-induced hepatocarcinoma in rats. Oncol 34: 249–253, 1981
Conseil of European Communities: Aproximación de las disposiciones legales, reglamentarias y administrativas de los Estados miembros respecto a la protección de los animales utilizados para experimentación y otros fines científicos. Diario Oficial de las Comunidades Europeas 358: 1–8, 1986
Ministerio de Agricultura, Pesca y Alimentación: Protección de los animales utilizados para experimentación y otros fines científicos. Boletín Oficial del Estado 67: 8509–8512, 1988
Europe Conseil: Convenio europeo sobre protrección de los animales vertebrados utilizados con fines experimentales y otros fines científicos. Boletín Oficial del Estado 256: 2187–2190, 1990
Ito N, Tsuda H, Tatematsu H, Inoue T, Tagawa Y, Aoki T, Uwagawa H, Ogiso T, Hasui T, Imaida K, Fukushima S, Asamoto M: Enhancing effect of various hepatocarcinogens on preneoplastic GST placental form positive foci in rats – An approach for a new medium – Term bioassay system. Carcinogen 9: 387–394, 1988
Sandoval IV, Sols A: Gluconeogenesis from serine by the serine-dehydratase-dependent pathway in rat liver. Eur J Biochem 43: 609–616, 1974
Bradford M: A rapid and sensitive method for the quantitation of microgram quantities of protein using the principle of protein-dye binding. Anal Biochem 72: 248–254 1976
JB Barroso, J Peragón, C Contreras-Jurado, L García-Salguero, FJ Corpas, FJ Esteban, MA Peinado, De la Higuera M, JA Lupiáñez, Impact of starvation-refeeding on kinetics and protein expression of trout liver NADPH-production systems. Am J Physiol 274: R1578–R1587
Ogawa H, Pitot HC, Fujioka M: Diurnal variation of the serine dehydratase mRNA level in rat liver. Arch Biochem Biophys 308: 285–291, 1994
Ogawa H, Kawamata S, Gomi T, Ansai Y, Karaki Y: Laparatomy causes a transient induction of rat liver serine dehydratase mRNA. Arch Biochem Biophys 316: 844–850, 1995
Ogawa H, Takusagawa F, Wakaki K, Kishi H, Eskandarian MR, Kobayashi M, Date T, Huh N-H, Pitot HC: Rat liver serine dehydratase. Bacterial expression and two folding domains as revealed by limited proteolysis. J Biol Chem 274: 12855–12860, 1999
Shu SY, Ju G, Fan LZ: The glucose-oxidase-DAB-nickel method in peroxidase histochemistry of the nevous system. Neurosci Lett 85: 169–171, 1988
Chomczynski P, Sacchi N: Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162: 156–159, 1987
Ogawa H, Fujioka M, Date T, Mueckler M, Su Y, Pitot HC: Rat serine dehydratase gene codes for two species of mRNA of which only one is translated into serine dehydratase. J Biol Chem 265: 14407–14413, 1990
Zimmermann T, Franke H, Dargel R: Studies on lipid and lipoprotein metabolism in rat liver cirrhosis induced by different regimens of thioacetamide administration. Exp Pathol 30: 109–117, 1986
Moreira E, Fontana L, Periago JL, Sánchez de Medina F, Gil A: Changes in fatty acid composition of plasma, liver microsomes and erythrocytes in liver cirrhosis induced by oral intake of thioacetamide in rats. Hepatol 21: 199–206, 1995
Fontana L, Moreira E, Torres MI, Fernández MI, Rios A, Sánchez de Medina F, Gil A: Serum amino acid changes in rats with thioacetamide-induced liver cirrhosis. Toxicol 106: 197–206, 1996
Nolte W, Hartmann H, Ramadori G: Glucose metabolism and liver cirrhosis. Exp Clin Endocrinol 103: 63–74, 1995
Cascales C, Martín-Sanz P, Pittner RA, Hopewell R, Brindley DN, Cascales M: Effects of an antitumoural rhodium complex on thioacetamide-induced liver tumour in rats. Changes in the activities of ornithine decarboxylase, tyrosine aminotransferase and of enzymes involved in fatty acid and glycerolipids synthesis. Biochem Pharmacol 35: 2655–2661, 1986
Pepplinkhuizen L, Bruinvels J, Blom W, Moleman P: Schizophrenia-like psychosis caused by a metabolic disorder. Lancet 1: 454–456, 1980
Jungermann K, Katz N: Functional specialization of different hepatocyte populations. Physiol Rev 69: 708–764, 1989
Kanamoto R, Su Y, Pitot HC: Hormonal regulation of serine dehydratase gene expression in liver and kidney of the adrenalectomized rat. Mol Endocrinol 5: 1661–1668, 1991
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
López-Flores, I., Barroso, J.B., Valderrama, R. et al. Serine dehydratase expression decreases in rat livers injured by chronic thioacetamide ingestion. Mol Cell Biochem 268, 33–43 (2005). https://doi.org/10.1007/s11010-005-2995-2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11010-005-2995-2