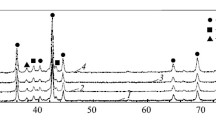
By the method of X-ray photoelectron spectroscopy, it is shown that, as a result of oxidation of the Zr–Mn–Ni–Cr–V alloy powder in air for 15 days, the most pronounced changes are observed in the spectrum of Ni2 p -electrons recorded on the surface of alloy in the charge state of Ni2+ and Ni0 in almost equal proportions. In this case, the relative intensity of the spectra of inner electrons of zirconium, nickel, manganese, and chromium decreases and, parallel with Cr0, Ni0 , and Mn0 , we reveal a certain amount of their atoms in the degree of oxidations M2+ (M = Cr, Ni, Mn). By the method of X-ray diffractometry, it was discovered that, after holding of an ingot of alloy in air for 5–6 months, as well as after the oxidation of alloy powder for 15 days, the phase composition remains unchanged. In this case, the electrode compressed from the alloy powder oxidized for 7 days is more resistant under cyclic loads.





Similar content being viewed by others
References
K. Hong, МКIS S22s30/00. Hydrogen-Accumulating Material and Its Application in Nickel-Hydride Batteries (UK Patent No. 2301115), Pr. 20.05.95, No. 9510225.7, Publ. on 23.11.96; NKI S7А.
L. Le Guenne, Ch. Argueu, and O. Arnaud, МPK7N01М 4/48. Matiere Electrochimiguement Active Pour Electrode Negative de Generateur Electrochimigue Secondaire a Electrolyte Alcalin (Patent of France), Publ. on 12.11.2004.
Y. Moriwaki, T. Gamo, H. Seri, and T. Iwari, “Electrode characteristics of C15-type Laves phase alloys,” J. Less-Common Metals, 172–174, 1211–1219 (1991).
S.–M. Lee, S.-H. Kim, and J.-Y. Lee, “A study on the electrode characteristics of Zr-based alloy by ball-milling process as an anode material for Ni–MH rechargeable batteries,” in: Internat. Symp. on Metal-Hydrogen Systems, Fundamentals & Applications (MH 2000) (Noosa Heads, Oct. 1–6, 2000), J. Alloys & Compounds, 330–332, 796–801 (2002).
Yu. M. Solonin, L. L. Kolomiets, S. M. Solonin, and V. V. Skorokhod, “Development of powder hydrogenated alloys for the electrodes of alkaline accumulators. 1. Principles of doping of the alloys with reversible sorption of hydrogen,” Poroshk. Metallurg., No. 7–8, 53–59 (2003).
Z. P. Li, B. H. Liu, K. Hitaka, and S. Suda, “Effects of surface structure of fluorinated AB2 alloys on their electrodes and battery performances,” J. Alloys & Compounds, 330–332, 7 (2002).
V. A. Shvets, O. Z. Galii, V. A. Lavrenko, Yu. M. Solonin, and Yu. A. Rudenko, “Corrosion-electrochemical behavior of the hydride-forming alloy of the Zr–Mn–Ni–Cr–Al system depending on the composition and time of surface oxidation in air,” Fiz.-Khim. Mekh. Mater., Special issue, Vol. 2, No. 9, 410–414 (2012).
V. D. Dobrovolsky, Yu. M. Solonin, V. V. Skorokhod, and O. Y. Khyzhun, “XPS and transmission electron microscopy of the multicomponent hydride-forming alloys,” Int. J. Hydrogen Energy, 24, 195–201 (1999).
A. A. Lavrentyev, B. V. Gabrelian, P. N. Shkumat, I. Y. Nikiforov, I. Y. Zavaliy, and O. Y. Khyzhun, “First-principles APW+LO calculations and X-ray spectroscopy studies of the electronic structure of Zr6FeAl2 ,” Mater. Chem. Phys., 136, 980–984 (2012).
M. V. Karpets, O. A. Gnitetskii, S. V. Sirichenko, and Yu. M. Solonin, “Phase composition and electrochemical properties of the zirconium-based hydride-forming alloy,” in: Proc. 7th Internat. Conf. on Hydrides (Alushta, 2001), p. 108.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Fizyko-Khimichna Mekhanika Materialiv, Vol. 53, No. 2, pp. 24–29, March–April, 2017.
Rights and permissions
About this article
Cite this article
Solonin, Y.М., Galii, О.Z., Graivoronskaya, Е.А. et al. Effect of Oxidation on the State of the Surface and the Electrode Capacitance of Zr–Mn–Ni–Cr–V Alloy. Mater Sci 53, 151–157 (2017). https://doi.org/10.1007/s11003-017-0056-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11003-017-0056-0