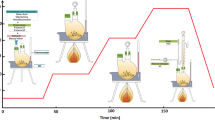
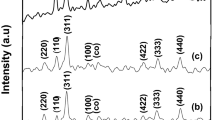
For the first time, we experimentally develop and propose a procedure of synthesis of Fe–Co–Ni salts in the nanodispersed state with prescribed properties and structure by means of their chemical precipitation. We establish the influence of the concentrations of components in the reaction mixture, introduction of the admixtures, and temperature and viscosity of the medium on the size and morphology of particles of metal salts obtained as a result.


Similar content being viewed by others
References
S. I. Koryagin, I. V. Pimenov, and V. K. Kudyakov, Methods of Treatment of the Materials [in Russian], Izd. KGU, Kaliningrad (2000).
I. T. Kazarmshchikov, Production of Metallic Structural Materials [in Russian], GOU OGU, Orenburg (2004).
T. M. Shvets’, Physical Chemistry of Ultradispersed Systems [in Russian], Nauka, Moscow (1987).
Yu. I. Krasnokutskii, Production of Refractory Compounds in Plasma [in Russian], Vyshcha Shkola, Kiev (1997).
É. T. Denisenko, O. P. Kulik, and T. V. Eremina, “Dispersed crystalline powders,” Poroshk. Metallurg., No. 4, 4–25 (1983).
K. Wegner, B. Walker, et al., “Design of metal nanoparticle synthesis by vapor flow condensation,” Chem. Eng. Sci., 57, No. 10, 1753–1762 (2002).
L. I. Boguslavskii, “Methods for the production of nanoparticles and their physical parameters sensitive to dimensions,” Vestn. MITKhT, 5, No. 5, 3–15 (2010).
R. Arbain and M. Othman, “Preparation of iron oxide nanoparticles by mechanical milling,” Minerals Eng., 24, No. 1, 1–9 (2011).
M. Marcinek, L. J. Hardwick, et al., “Microwave plasma chemical vapor deposition of nanocomposite,” J. Power Sources, 173, No. 2, 965–972 (2007).
N. G. Semaltionosa, “Nanoparticles by laser ablation,” Crit. Rev. Solid State Mater. Sci., 35, No. 2, 105–124 (2010).
A. M. Panfilov, Kinetics of Homogeneous and Heterogeneous Chemical Reactions [in Russian], GOU VPO UGPU-UPI, Ekaterinburg (2005).
M. Volmer, Kinetik der Phasenbildung, Steinkopf, Leipzig (1939).
G. A. Krestov, Thermodynamics of Ionic Processes in Solutions [in Russian], Khimiya, Moscow (1984).
G. D. Sergeev, Nanochemistry [in Russian], MGU, Moscow (2003).
A. P. Shpak, Yu. A. Kunyts’kyi, and M. I. Zakharenko, Magnetism of Amorphous and Nanocrystalline Systems [in Ukrainian], Akademperiodyka, Kyiv (2003).
G. A. Krestov, V. I. Vinogradov, and Yu. M. Kessler, Contemporary Problems of the Chemistry of Solutions [in Russian], Nauka, Moscow (1986).
G. A. Krestov, N. N. Novoselov, and I. S. Perelygin, Problems of the Chemistry of Solutions: Ionic Solvation [in Russian], Nauka, Moscow (1987).
A. V. Pakoev and Yu. V. Osinskaya, Physical Properties of Crystals [in Russian], Izd. “Samara,” Samara, (2007).
A. K. Dudchenko and A. S. Kuzema, A Method for the Preparation of Compositions of Ferromagnetic Substances by Electron Microscopy and the Devices for Its Realization [in Russian], USSR Author’s Certificate No. 1033904, Publ. 10.07.83, Bull. No. 29.
GOST 23401–90 (ST SГV 9746-890). Metallic Powders. Catalysts and Carriers. Evaluation of the Specific Surface [in Russian], Izd. Standartov, Moscow (1991).
The present work work was financially supported within the framework of the Program of Ukrainian-Vietnamese Collaboration (Project No. M/39-2013).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Fizyko-Khimichna Mekhanika Materialiv, Vol. 51, No. 1, pp. 58–63, January–February, 2015.
Rights and permissions
About this article
Cite this article
Kushchevs’ka, N.F., Shakhnin, D.B. & Malyshev, V.V. Creation of Ferromagnetic Nanocomposites. I. Chemical Synthesis of Fe–Co–Ni Salts. Mater Sci 51, 61–67 (2015). https://doi.org/10.1007/s11003-015-9810-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11003-015-9810-3