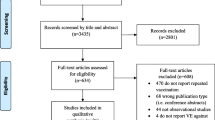
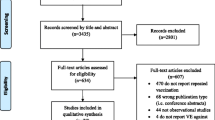
Abstract
Objective
Pregnant women have a high risk for complications from influenza infection, but vaccination rates within this group remain low in the US and other countries. The efficacy and effectiveness of the influenza vaccine are a key determinant of vaccine uptake. This review aimed to synthesize the available evidence on the protection of both seasonal and monovalent pandemic H1N1 (pdmH1N1) vaccine against laboratory-confirmed influenza (LCI), influenza-like illness (ILI), and respiratory illness (RI).
Methods
A search of the literature was undertaken from Pubmed, Embase, ClinicalTrials.gov, and Cochrane Central Register of Controlled Trials up to Aug 1, 2018. Both observational studies and clinical trials were considered.
Results
Nineteen studies were identified from 11 countries. Women with pdmH1N1 vaccination had a lower risk of getting LCI (Relative risk [RR] 0.3, 95% confidence interval [CI] 0.26–0.35) and ILI (RR 0.15, 95% CI 0.06–0.36)). The pooled estimate from three randomized clinical trials (RR 0.47, 95% CI 0.31–0.71) and two case control studies (OR 0.37, 95% CI 0.23–0.61) showed that the seasonal vaccine was protective against LCI. The seasonal vaccine was not protective against ILI (RR 0.95, 95% CI 0.88–1.03). This association was similar for the outcome of RI (RR 0.81, 95% CI 0.55–1.20).
Conclusion
This analysis bolsters existing evidence that influenza vaccines are effective among pregnant women. Additional public health efforts are needed to promote physician recommendations of influenza vaccination in pregnancy.




Similar content being viewed by others
References
Ahrens, K. A., Louik, C., Kerr, S., Mitchell, A. A., & Werler, M. M. (2014). Seasonal influenza vaccination during pregnancy and the risks of preterm delivery and small for gestational age birth. Paediatric and Perinatal Epidemiology,28(6), 498–509.
Akl, E. A., Sun, X., Busse, J. W., Johnston, B. C., Briel, M., Mulla, S., et al. (2012). Specific instructions for estimating unclearly reported blinding status in randomized trials were reliable and valid. Journal of Clinical Epidemiology,65(3), 262–267.
Black, S. B., Shinefield, H. R., France, E. K., Fireman, B. H., Platt, S. T., & Shay, D. (2004). Effectiveness of influenza vaccine during pregnancy in preventing hospitalizations and outpatient visits for respiratory illness in pregnant women and their infants. American Journal of Perinatology,21(06), 333–339.
Brien, S., Kwong, J. C., & Buckeridge, D. L. (2012). The determinants of 2009 pandemic A/H1N1 influenza vaccination: A systematic review. Vaccine,30(7), 1255–1264.
Cheng, A. C., Holmes, M., Irving, L. B., Brown, S. G., Waterer, G. W., Korman, T. M., et al. (2013). Influenza vaccine effectiveness against hospitalisation with confirmed influenza in the 2010–11 seasons: A test-negative observational study. PLoS ONE,8(7), e68760.
Cox, N. J., & Subbarao, K. (1999). Influenza. The Lancet,354(9186), 1277–1282. https://doi.org/10.1016/S0140-6736(99)01241-6.
Demicheli, V., Di Pietrantonj, C., Jefferson, T., Rivetti, A., & Rivetti, D. (2007). Vaccines for preventing influenza in healthy adults. Cochrane Database Systematic Review,2, CD001269.
DerSimonian, R., & Laird, N. (1986). Meta-analysis in clinical trials. Controlled Clinical Trials,7(3), 177–188.
Ding, H., Black, C. L., Ball, S., Fink, R. V., Williams, W. W., Fiebelkorn, A. P., et al. (2017). Influenza vaccination coverage among pregnant women-United States, 2016-17 influenza season. MMWR. Morbidity and Mortality Weekly Report,66(38), 1016–1022.
Doyle, T. J., Goodin, K., & Hamilton, J. J. (2013). Maternal and neonatal outcomes among pregnant women with 2009 pandemic influenza A (H1N1) illness in Florida, 2009-2010: A population-based cohort study. PLoS ONE,8(10), e79040.
Ellington, S. R., Hartman, L. K., Acosta, M., Martinez-Romo, M., Rubinson, L., Jamieson, D. J., et al. (2011). Pandemic 2009 influenza A (H1N1) in 71 critically ill pregnant women in California. American Journal of Obstetrics and Gynecology,204(6), S21–S30.
Emeruwa, U. N., Nour, N. M., & Diouf, K. (2018). Outcomes associated with influenza-like illness in the peripartum period. Paper presented at the reproductive sciences.
Fabiani, M., Bella, A., Rota, M. C., Clagnan, E., Gallo, T., D’Amato, M., et al. (2015). A/H1N1 pandemic influenza vaccination: A retrospective evaluation of adverse maternal, fetal and neonatal outcomes in a cohort of pregnant women in Italy. Vaccine,33(19), 2240–2247.
Frey, S., Vesikari, T., Szymczakiewicz-Multanowska, A., Lattanzi, M., Izu, A., Groth, N., et al. (2010). Clinical efficacy of cell culture-derived and egg-derived inactivated subunit influenza vaccines in healthy adults. Clinical Infectious Diseases,51(9), 997–1004.
Getahun, D., Fassett, M. J., Peltier, M. R., Chiu, V., Wing, D. A., & Jacobsen, S. J. (2017). 499: Association between influenza immunization during pregnancy and perinatal outcomes. American Journal of Obstetrics and Gynecology,216(1), S295.
Grohskopf, L. A., Sokolow, L. Z., Broder, K. R., Walter, E. B., Fry, A. M., & Jernigan, D. B. (2018). Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2018–19 influenza season. MMWR Recommendations and Reports,67(3), 1.
Håberg, S. E., Trogstad, L., Gunnes, N., Wilcox, A. J., Gjessing, H. K., Samuelsen, S. O., et al. (2013). Risk of fetal death after pandemic influenza virus infection or vaccination. New England Journal of Medicine,368(4), 333–340.
Higgins, J. P., Altman, D. G., Gøtzsche, P. C., Jüni, P., Moher, D., Oxman, A. D., et al. (2011). The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ,343, d5928.
Hulka, J. F. (1964). Effectiveness of polyvalent influenza vaccine in pregnancy: Report of a controlled study during an outbreak of Asian influenza. Obstetrics & Gynecology,23(6), 830–837.
Luo, Y., Johnson, D. L., Xu, R., & Chambers, C. D. (2013). Comparison of the incidence of influenza like illness in pregnant women with rheumatoid arthritis and women without rheumatoid arthritis who receive an influenza vaccination. Paper presented at the arthritis and rheumatism.
Ma, F., Zhang, L., Jiang, R., Zhang, J., Wang, H., Gao, X., et al. (2014). Prospective cohort study of the safety of an influenza A (H1N1) vaccine in pregnant Chinese women. Clinical and Vaccine Immunology,21(9), 1282–1287.
Madhi, S. A., Cutland, C. L., Kuwanda, L., Weinberg, A., Hugo, A., Jones, S., et al. (2014). Influenza vaccination of pregnant women and protection of their infants. New England Journal of Medicine,371(10), 918–931.
Matias, G., Taylor, R., Haguinet, F., Schuck-Paim, C., Lustig, R., & Shinde, V. (2014). Estimates of mortality attributable to influenza and RSV in the U nited S tates during 1997–2009 by influenza type or subtype, age, cause of death, and risk status. Influenza and Other Respiratory Viruses,8(5), 507–515.
Matias, G., Taylor, R., Haguinet, F., Schuck-Paim, C., Lustig, R., & Shinde, V. (2017). Estimates of hospitalization attributable to influenza and RSV in the US during 1997–2009, by age and risk status. BMC Public Health,17(1), 271.
Mosby, L. G., Rasmussen, S. A., & Jamieson, D. J. (2011). 2009 pandemic influenza A (H1N1) in pregnancy: A systematic review of the literature. American Journal of Obstetrics and Gynecology,205(1), 10–18.
Munoz, F. M., Greisinger, A. J., Wehmanen, O. A., Mouzoon, M. E., Hoyle, J. C., Smith, F. A., et al. (2005). Safety of influenza vaccination during pregnancy. American Journal of Obstetrics and Gynecology,192(4), 1098–1106.
Mutsaerts, E., Madhi, S. A., Cutland, C. L., Jones, S., Hugo, A., Trenor, S., et al. (2016). Influenza vaccination of pregnant women protects them over two consecutive influenza seasons in a randomized controlled trial. Expert Review of Vaccines,15(8), 1055–1062.
Nunes, M. C., Aqil, A. R., Omer, S. B., & Madhi, S. A. (2016). The effects of influenza vaccination during pregnancy on birth outcomes: A systematic review and meta-analysis. American Journal of Perinatology,33(11), 1104–1114.
Nunes, M. C., & Madhi, S. A. (2018). Influenza vaccination during pregnancy for prevention of influenza confirmed illness in the infants: A systematic review and meta-analysis. Human Vaccines & Immunotherapeutics,14(3), 758–766.
Ohfuji, S., Deguchi, M., Tachibana, D., Koyama, M., Takagi, T., Yoshioka, T., et al. (2017). Protective effect of maternal influenza vaccination on influenza in their infants: A prospective cohort study. The Journal of Infectious Diseases,217(6), 878–886.
Olsen, S. J., Mirza, S. A., Vonglokham, P., Khanthamaly, V., Chitry, B., Pholsena, V., et al. (2016). The effect of influenza vaccination on birth outcomes in a cohort of pregnant women in Lao PDR, 2014–2015. Reviews of Infectious Diseases,63(4), 487–494.
Regan, A. K., de Klerk, N., Moore, H. C., Omer, S. B., Shellam, G., & Effler, P. V. (2016). Effectiveness of seasonal trivalent influenza vaccination against hospital-attended acute respiratory infections in pregnant women: A retrospective cohort study. Vaccine,34(32), 3649–3656.
Richards, J. L., Hansen, C., Bredfeldt, C., Bednarczyk, R. A., Steinhoff, M. C., Adjaye-Gbewonyo, D., et al. (2013). Neonatal outcomes after antenatal influenza immunization during the 2009 H1N1 influenza pandemic: Impact on preterm birth, birth weight, and small for gestational age birth. Clinical Infectious Diseases,56(9), 1216–1222.
Steinhoff, M. C., Katz, J., Englund, J. A., Khatry, S. K., Shrestha, L., Kuypers, J., et al. (2017). Year-round influenza immunisation during pregnancy in Nepal: A phase 4, randomised, placebo-controlled trial. The Lancet Infectious Diseases,17(9), 981–989.
Steinhoff, M. C., Omer, S. B., Roy, E., El Arifeen, S., Raqib, R., Dodd, C., et al. (2012). Neonatal outcomes after influenza immunization during pregnancy: A randomized controlled trial. Canadian Medical Association Journal: CMAJ,184(6), 645–653.
Tapia, M. D., Sow, S. O., Tamboura, B., Tégueté, I., Pasetti, M. F., Kodio, M., et al. (2016). Maternal immunisation with trivalent inactivated influenza vaccine for prevention of influenza in infants in Mali: A prospective, active-controlled, observer-blind, randomised phase 4 trial. The Lancet Infectious Diseases,16(9), 1026–1035.
Tempia, S., Walaza, S., Cohen, A. L., von Mollendorf, C., Moyes, J., McAnerney, J. M., et al. (2015). Mortality associated with seasonal and pandemic influenza among pregnant and nonpregnant women of childbearing age in a high-HIV-prevalence setting—South Africa, 1999–2009. Clinical Infectious Diseases,61(7), 1063–1070.
Thompson, M. G., Li, D.-K., Shifflett, P., Sokolow, L. Z., Ferber, J. R., Kurosky, S., et al. (2013). Effectiveness of seasonal trivalent influenza vaccine for preventing influenza virus illness among pregnant women: A population-based case-control study during the 2010–2011 and 2011–2012 influenza seasons. Clinical Infectious Diseases,58(4), 449–457.
Wells, G., Shea, B., O’Connell, D., Peterson, J., Welch, V., Losos, M., et al. (2012). The Newcastle-Ottawa Scale (NOS) for assessing the quality of non-randomized studies in meta-analysis. Ottawa: Ottawa Health Research Institute.
WHO. (2012). Vaccines against influenza WHO position paper—November 2012. Weekly Epidemiological Record = Relevé épidémiologique hebdomadaire,87(47), 461–476.
Wong, V. W., Lok, K. Y., & Tarrant, M. (2016). Interventions to increase the uptake of seasonal influenza vaccination among pregnant women: A systematic review. Vaccine,34(1), 20–32.
Wong, V. W., & Tarrant, M. (2015). Influenza-like illness among Hong Kong Chinese pregnant women. The Journal of Maternal-Fetal & Neonatal Medicine,28(1), 116–119.
Yamada, T., Kawakami, S., Yoshida, Y., Kawamura, H., Ohta, S., Abe, K., et al. (2016). Influenza 2014-2015 among pregnant Japanese women: Primiparous vs multiparous women. European Journal of Clinical Microbiology & Infectious Diseases,35(4), 665–671.
Yamada, T., Yamada, T., Morikawa, M., Cho, K., Endo, T., Sato, S. S., et al. (2012). Pandemic (H1N1) 2009 in pregnant Japanese women in Hokkaido. Journal of Obstetrics and Gynaecology Research,38(1), 130–136.
Zaman, K., Roy, E., Arifeen, S. E., Rahman, M., Raqib, R., Wilson, E., et al. (2008). Effectiveness of maternal influenza immunization in mothers and infants. New England Journal of Medicine,359(15), 1555–1564. https://doi.org/10.1056/NEJMoa0708630.
Zhang, P., Li, X.-L., Cao, B., Yang, S.-G., Liang, L.-R., Gu, L., et al. (2012). Clinical features and risk factors for severe and critical pregnant women with 2009 pandemic H1N1 influenza infection in China. BMC Infectious Diseases,12(1), 29.
Zhang, C., Wang, X., Liu, D., Zhang, L., & Sun, X. (2018). A systematic review and meta-analysis of fetal outcomes following the administration of influenza A/H1N1 vaccination during pregnancy. International Journal of Gynecology & Obstetrics,141(2), 141–150.
Acknowledgements
We would like to thank the University of Georgia’s College of Public Health Epidemiology and Biostatistics Department. Additionally, we would like to acknowledge support from the University of Georgia’s Global Health Institute. We are grateful for the valuable suggestions made by Dr. Mark Ebell, UGA Professor of Epidemiology & Biostatistics. No funding was received to conduct this review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
TQ and NAM declare that they have no conflict of interest. Dr. Cordero reports consulting for Sanofi Pasteur, outside the submitted work. All authors have submitted the ICMJE form for Disclosure of Potential Conflicts of Interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Quach, T.H.T., Mallis, N.A. & Cordero, J.F. Influenza Vaccine Efficacy and Effectiveness in Pregnant Women: Systematic Review and Meta-analysis. Matern Child Health J 24, 229–240 (2020). https://doi.org/10.1007/s10995-019-02844-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10995-019-02844-y