Abstract
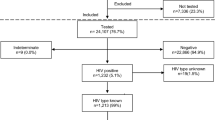
Objective To identify factors associated with maternal hepatitis C virus (HCV) seroprevalence and transmission of HCV as identified by qualitative HCV ribonucleic acid (RNA) in the infants of human immunodeficiency virus (HIV) infected women delivering in New York State (NYS) in 2006. Study Design In this retrospective cohort study of HIV-exposed infants born in NYS, leftover infant plasma from HIV diagnostic testing was de-identified and tested for HCV. If HCV antibodies were detected, a second specimen collected when the infant was >2 months old was tested for HCV qualitative RNA. Multivariate logistic regression was used to identify factors associated with HCV seropositivity. Results In a final sample of 553 live birth events with perinatal HIV exposure, 21 (3.8 %) of tested infant specimens had HCV antibodies indicative of maternal HCV seropositivity. Maternal age at delivery of >35 years, Hispanic ethnicity, white race and injection drug use (IDU) were significantly associated with HCV seropositivity in multivariate analysis. No cases of HCV vertical transmission were identified among HCV exposed infant specimens. Conclusions This statewide population-based study of HIV-infected childbearing women shows HCV seroprevalence of 3.8 %. Maternal age of >35 years and IDU are the strongest predictors of HCV seropositivity. Although no viral transmission was documented, more comprehensive longitudinal testing would be required to conclude that HCV transmission did not occur.
Similar content being viewed by others
Abbreviations
- HCV:
-
Hepatitis C virus
- HIV:
-
Human immunodeficiency virus
- IDU:
-
Injection drug use
- NHANES:
-
National Health and Nutrition Examination Survey
- NYS:
-
New York State
- NYSDOH:
-
New York State Department of Health
- RNA:
-
Ribonucleic acid
References
Denniston, M. M., et al. (2014). Chronic hepatitis C virus infection in the United States, National Health and Nutrition Examination Survey 2003–2010. Annals of Internal Medicine, 160(5), 293–300.
Hellard, M., Sacks-Davis, R., & Gold, J. (2009). Hepatitis C treatment for injection drug users: A review of the available evidence. Clinical Infectious Diseases, 49(4), 561–573.
Grebely, J., & G.J. Dore, G. J. (2011). What is killing people with hepatitis C virus infection? In Seminars in Liver Diseases.
Trooskin, S. B., et al. (2015). Results from a Geographically Focused, Community-Based HCV Screening, Linkage-to-Care and Patient Navigation Program. Journal of General Internal Medicine, 30(7), 950–957.
Alter, M. J., et al. (1999). The prevalence of hepatitis C virus infection in the United States, 1988 through 1994. New England Journal of Medicine, 341(8), 556–562.
Arshad, M., El-Kamary, S., & Jhaveri, R. (2011). Hepatitis C virus infection during pregnancy and the newborn period–are they opportunities for treatment? Journal of Viral Hepatitis, 18(4), 229–236.
Yeung, L. T., King, S. M., & Roberts, E. A. (2001). Mother-to-infant transmission of hepatitis C virus. Hepatology, 34(2), 223–229.
Dunkelberg, J., et al. (2014). Hepatitis B and C in pregnancy: A review and recommendations for care. Journal of Perinatology, 34, 882–891.
Roberts, E. A., & Yeung, L. (2002). Maternal-infant transmission of hepatitis C virus infection. Hepatology, 36(5B), s106–s113.
Thaler, M., et al. (1991). Vertical transmission of hepatitis C virus. The Lancet, 338(8758), 17–18.
Zanetti, A. R., et al. (1995). Mother-to-infant transmission of hepatitis C virus. Lombardy study group on vertical HCV transmission. Lancet, 345(8945), 289–291.
Birkhead, G. S., et al. (2010). Progress in prevention of mother-to-child transmission of HIV in New York State: 1988–2008. Journal of Public Health Management and Practice, 16(6), 481–491.
CDC. (2013). Testing for HCV infection: An update of guidance for clinicians and laboratorians. Morbidity and Mortality Weekly Report, 62(18), 362.
Heinze, G., & Schemper, M. (2002). A solution to the problem of separation in logistic regression. Statistics in Medicine, 21(16), 2409–2419.
Frederick, T., et al. (2009). Factors associated with prevalent hepatitis C infection among HIV-infected women with no reported history of injection drug use: The Women’s Interagency HIV Study (WIHS). AIDS patient care and STDs, 23(11), 915–923.
Hagan, H., Thiede, H., & Des, D. C. (2005). Jarlais, HIV/hepatitis C virus co-infection in drug users: Risk behavior and prevention. AIDS, 19, S199–S207.
AASLD/IDSA HCV Guidance Panel. (2015). Hepatitis C guidance: AASLD-IDSA recommendations for testing, managing, and treating adults infected with hepatitis C virus. Hepatology, 62(3), 932–954.
Alter, M. J., et al. (1998). Recommendations for prevention and control of hepatitis C virus (HCV) infection and HCV-related chronic disease. MMWR. Morbidity and Mortality Weekly Report, 47(1), 1–39.
Smith, B. D., et al. (2012). Recommendations for the identification of chronic hepatitis C virus infection among persons born during 1945–1965. MMWR Recommendations and Reports, 61(RR-4), 1–32.
Santiago-Munoz, P., et al. (2005). Prevalence of hepatitis B and C in pregnant women who are infected with human immunodeficiency virus. American Journal of Obstetrics and Gynecology, 193(3 Pt 2), 1270–1273.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghazaryan, L., Smith, L., Parker, M. et al. Hepatitis C Seroprevalence Among HIV-Infected Childbearing Women in New York State in 2006. Matern Child Health J 20, 550–555 (2016). https://doi.org/10.1007/s10995-015-1853-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10995-015-1853-4