Abstract
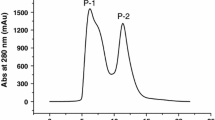
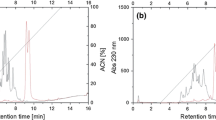
Angiotensin-converting enzyme (ACE) enhances blood pressure by making potential vasoconstrictor, Angiotensin-II from Angiotensin-I. Thus, it is the key target enzyme in cardiovascular disease therapy. Hence, in the present study an attempt has been made for identification and characterization of ACE inhibitory peptide from buffalo milk casein. Buffalo milk casein was isolated and digested with different digestive enzymes (pepsin, trypsin, chymotrypsin and their combination). Pepsin-Trypsin hydrolysates shown highest (72.55 ± 2.23%/50 μg) ACE inhibitory activity, in further ultra-filtration, < 1 kDa fraction shown the highest inhibitory activity among the other fractions. Below 1 kDa filtrate was further fractionated through RP-HPLC, fraction 11 exhibited the highest ACE inhibitory activity. Out of 15 peptides of 11th fraction (LC–MS/MS sequence) VLPVPQK is the novel peptide, which shown significant ACEI activity. Therefore, this study supports the notion that milk is a very good nutraceutical, and the novel peptide sequence can be utilized in commercial peptide synthesis for making functional food preparation.





Similar content being viewed by others
References
Abubakar A, Saito T, Kitazawa H, Kawai T, Itoh T (1998) Structural analysis of new antihypertensive peptides derived from cheese whey protein by proteinse K digestion. J Dairy Sci 81(12):3131–3138
Addeo F, Chobert JM, Ribadeau-Dumas B (1977) Fractionation of whole casein on hydroxyl apatide: Application to a study of buffalo κ–casein. J Dairy Res 44:63–68
Alhaj OA (2017) Identification of potential ACE-inhibitory peptides from dromedary fermented camel milk. CyTA - J Food 15(2):191–195
Cheung HS, Wang FL, Ondetti MA, Sabo EF, Cushman DW (1980) Binding of peptide substrate and inhibitors of angiotensing converting enzyme. J Biol Chem 255:401–407
Church FC, Swaisgood HE, Porter DH, Catignani GL (1983) Spectrophotometric assay using O-phthaldehyde for determination of proteolysis in milk and isolated milk proteins. J Dairy Sci 66:1219–1227
Cushman DW, Cheung HS (1971) Spectrophotometric assay and properties of the angiotensin converting enzyme of rabbit lung. Biochem Pharmacol 20(7):1637–1648
Davies DT, Law AJR (1977) An improved method for the quantitative fractionation of casein mixture using ion-exchange chromatography. J Dairy Res 44:213–221
Deepthi K (2004) Comparative studies on casein derived ACE inhibitory and immunomodulatory peptides from cow and buffalo milk. Ph.D Thesis NDRI (Deemed University), Karnal, India
Ferreia SH, Bartert DC, Greene LJ (1970) Isolation of bradykinin potentiating peptides from Bothrops iararaca venom. Biochemistry 9:2583–2593
FitzGerald RJ, Meisel H (2000) Milk protein-derived peptide inhibitors of angiotensin-I-converting enzyme. Br J Nutr 84(Suppl. 1):S33–S37
García-Moreno PJ, Espejo-Carpio FJ, Guadix A, Guadix EM (2015) Production and identification of angiotensin I-converting enzyme (ACE) inhibitory peptides from Mediterranean fish discards. J Funct Foods 18:95–105
Haque E, Chand R, Kapila S (2009) Biofunctional properties of bioactive peptides of milk origin. Food Reviews International 25(1):28–43
Hata Y, Yamamoto M, Ohni M, Nakajima K, Nakamura Y, Takano T (1996) A placebo-controlled study of the effect of sour milk onblood pressure in hypertensive subjects. Am J Clin Nutr 64(5):767–771
Hernández-Ledesma B, Contreras M, Recio I (2011) Antihypertensive peptides: Production, bioavailability and incorporation into foods. Adv Colloid Interface Sci 165:23–35
Hernandez-Ledesma B, Martin-Alvarez PJ, Pueyo E (2003) Assessment of the spectrophotometric method for determination of angiotensin-converting enzyme activity: Influence of the inhibition type. J Agric Food Chem 51(15):4175–4179
Hernandez-Ledesma B, Recio I, Ramos M, Amigo L (2002) Preparation of ovine and caprine β-lactoglobulin hydrolysates with ACE inhibitory activity: influence of the inhibition type. J Agril Food Chem 51:4175–4179
Huang GJ, Lu TL, Chiu CS, Chen HJ, Wu CH, Lin YC, Hsieh WT, Liao JC, Sheu MJ, Lin YH (2011) Sweet potato storage root defensing and its tryptic hydrolystes exhibited angiotensin converting enzyme inhibitory activity in vitro. Bot Stud 52:257–264
Ibrahim HR, Ahmed AS, Miyata T (2017) Novel angiotensin-converting enzyme inhibitory peptides from caseins and whey proteins of goat milk. J Adv Res. https://doi.org/10.1016/j.jare.2016.12.002
Jenness R, Holt C (1987) Casein and lactose concentrations in milk of 31 species are negatively correlated. Experimentia 43:1015–1018
Guo J, Astrup A, Lovegrove JA, Gijsbers L, Givens DI, Soedamah-Muthu SS (2017) Milk and dairy consumption and risk of cardiovascular diseases and all-cause mortality: dose–response meta-analysis of prospective cohort studies. Eur J Epidemiol 32:269–287
Lin K, Zhang L, Han X, Meng Z, Zhang J, Yifan Wu, Cheng D (2018) Quantitative structure-activity relationship modeling coupled with molecular docking analysis in screening of Angiotensin I-converting enzyme inhibitory peptides from Qula Casein hydrolysates obtained by two-enzyme combination hydrolysis. J Agric Food Chem 66(12):3221–3228
Korhonen H, Pihlanto A (2006) Bioactive peptides: production and functionality—review. Int Dairy J 16:945–960
Laemmli VK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227(15):680–685
Lee SY, Hur SJ (2017) Angiotensin converting enzyme inhibitory and antioxidant activities of enzymatic hydrolysates of Korean Native Cattle (Hanwoo) myofibrillar protein. BioMed Res Int. https://doi.org/10.1155/2017/5274637
Li G, Le G, Shi Y, Shrestha S (2004) Angiotensin I–converting enzyme inhibitory peptides derived from food proteins and their physiological and pharmacological effects. Nutr Res 24:469–486
Lu Z, Karne S, Kolodecik T, Gorelick FS (2002) Alcohols enhance caerulein-induced zymogen activation in pancreatic acinar cells. Am J Physiol Gasterointest Liver Physiol 282:G501–G507
Mada SB, Reddi S, Kumar N, Kumar R, Kapila S, Kapila R, Trivedi R, Karvande A, Ahmad N (2017) Antioxidative peptide from milk exhibits anti-osteopenic effects through inhibition of oxidative damage and bone-resorbing cytokines in ovariectomized rats. Nutrition 43–44:21–31
Mada SB, Reddi S, Kumar N, Vij R, Yadav R, Kapila S, Kapila R (2018) Casein-derived antioxidative peptide prevents oxidative stress-induced dysfunction in osteoblast cells. Pharmanutrition 6:169–179
Maeno M, Yamamoto N, Takano T (1996) Identification of an antihypertensive peptide from casein hydrolysate produced by a proteinase from Lactobacillus helveticus CP790. J Dairy Sci 79(8):1316–1321
Mao X, Cheng X, Wang X, Wu S (2011) Free-radical-scavenging and anti-inflammatory effect of yak milk casein before and after enzymatic hydrolysis. Food Chem 126:484–490
Maolin Tu, Wang C, Chen C, Zhang R, Liu H, Weihong Lu, Jiang L, Ming Du (2018) Identification of a novel ACE-inhibitory peptide from casein and evaluation of the inhibitory mechanisms. Food Chem 256:98–104
Akif M, Masuyer G, Bingham RJ, Sturrock ED, Elwyn Isaac R, Ravi Acharya K (2012) Structural basis of peptide recognition by the angiotensin-1 converting enzyme homologue AnCE from Drosophila melanogaster. FEBS J 279:4525–4534
Mullally MM, Meisel H, FitzGerald RJ (1997) Angiotensin-I-converting enzyme inhibitory activities of gastric and pancreatic proteinase digests of whey proteins. Int Dairy J 7:299–303
Ondetti MA, Williams NJ, Sabo EF, Plusce J, Weaver ER, Kocy O (1971) Angiotensin converting enzyme inhibitors from the venom of Borthrops jararaca, isolation, elucidation of structure and synthesis. Biochemistry 19(22):4033–4039
Pandey M, Kapila R, Kapila S (2018) Evaluation of the osteoprotective potential of whey derived-antioxidative (YVEEL) and angiotensin-converting enzyme inhibitory (YLLF) bioactive peptides in ovariectomised rats. Food Funct 9(9):4791–4801
Pandey M, Kapila R, Kapila S (2018) Osteoanabolic activity of whey-derived anti-oxidative (MHIRL and YVEEL) and angiotensin-converting enzyme inhibitory (YLLF, ALPMHIR, IPA and WLAHK) bioactivepeptides. Peptides 99:1–7
Reddi S, Shanmugam VP, Tanedjeu KS, Kapila S, Kapila R (2016) Effect of buffalo casein-derived novel bioactive peptides on osteoblast differentiation. Eur J Nutr 57:593–605
Segura-Campos MR, Chel-Guerrero LA, Betancur-Ancona DA (2011) Purification of angiotensin I—converting enzyme inhibitory peptides from a cowpea (Vigna unguiculata) enzymatic hydrolysate. Process Biochem 46:864–872
Shanmugam VP, Kapila S, Kemgang Sonfack T, Kapila R (2015) Antioxidative peptide derived from enzymatic digestion of buffalo casein. Int Dairy J 42:1–5
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ, Klenk DC (1985) Measurement of protein using bicinchoninic acid. Anal Biochem 150:76–85
Swaisgood HE (1973) The caseins. CRC Crit Rev Food Technol 3:375–414
Tavano OL (2013) Protein Hydrolysis using Proteases: An Important Tool For Food Biotechnology. J Mol Catal B Enzym 90:1–11
Vij R, Reddi S, Kapila S, Kapila R (2016) Transepithelial transport of milk derived bioactive peptide VLPVPQK. Food Chem 190:681–688
Xu R (2009) Effect of whey protein on the proliferation and differentiation of osteoblasts. J Dairy Sci 92:3012–3018
Yamamoto N, Maeno M, Takano T (1990) Purification and characterisation of anantihypertensive peptid from yoghurt-like product fermented by Lactobacillus helveticus CPN4. J Dairy Sci 82:1388–1393
Acknowledgements
The authors thank the Director of National Dairy Research Institute for providing facilities.
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial or proprietary interests in any material discussed in this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shanmugam, V.P., Kapila, S., Kemgang, T.S. et al. Isolation and Characterization of Angiotensin Converting Enzyme Inhibitory Peptide from Buffalo Casein. Int J Pept Res Ther 27, 1481–1491 (2021). https://doi.org/10.1007/s10989-021-10185-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-021-10185-0