Abstract
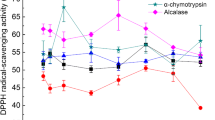
Pecan cake, a by-product of pecan processing, has not been fully developed and utilized. It contains proteins with high nutritional value that can be used as a potential source of bioactive peptides. In this study, the compound protease from the four common proteases was selected as the best protease, and the best hydrolysis was determined via response surface optimization. The optimal hydrolysis conditions were as follows: 55 ℃, pH 7.7, 3900 U/g protease concentration, and 2 h. Under these conditions, peptide yield and total antioxidant capacity (TAC) were 34.1 % ± 0.2 % and 1.1 ± 0.1 mmol/g, respectively. The peptides showed desirable 1,1-diphenyl-2-picrylhydrazyl (DPPH) (IC50: 0.2 mg/mL) and 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) (IC50: 0.4 mg/mL) radical scavenging capacities, which were found to be related to their amino acid compositions, molecular weights, and structures. The molecular weight of the peptides obtained from the pecan cake protein hydrolyzed by the compound protease was < 3 KDa, with their contents reaching 94.0 % ± 0.4 %. The yield of protein hydrolysate from pecan cake was about 7.2 %. HPLC analysis revealed that the content of hydrophobic amino acids in the peptides was relatively high. Fourier transform infrared spectroscopy and X-ray diffraction analysis showed that hydrolysis gradually changed the structure of the proteins from ordered to disordered, thereby exposing more antioxidant groups. α-helix and β-turn angles decreased from 29.5 % and 24.9–20.3 % and 15.2 %, respectively. β-sheet, β-antiparallel, and random coil increased from 24.2 %, 7.7 %, and 13.7–33.5 %, 9.3 %, and 21.8 %, respectively.






Similar content being viewed by others
References
Barth A (2007) Infrared spectroscopy of proteins. Biochim Biophys Acta 1767:1073–1101. https://doi.org/10.1016/j.bbabio.2007.06.004
Bhadane BS, Patil RH (2017) Isolation, purification and characterization of antioxidative steroid derivative from methanolic extract of Carissa carandas (L.) leaves. Biocatal Agric Biotechnol 10:216–223. https://doi.org/10.1016/j.bcab.2017.03.012
Chalamaiah M, Dinesh Kumar B, Hemalatha R, Jyothirmayi T (2012) Fish protein hydrolysates: proximate composition, amino acid composition, antioxidant activities and applications: a. review Food Chem 135:3020–3038. https://doi.org/10.1016/j.foodchem.2012.06.100
Chalamaiah M, Yu W, Wu J (2018) Immunomodulatory and anticancer protein hydrolysates (peptides) from food proteins. A review. Food Chem 245:205–222. https://doi.org/10.1016/j.foodchem.2017.10.087
Cotabarren J, Rosso AM, Tellechea M, Garcia-Pardo J, Rivera JL, Obregon WD, Parisi MG (2019) Adding value to the chia (Salvia hispanica L.) expeller: production of bioactive peptides with antioxidant properties by enzymatic hydrolysis with. Papain Food Chem 274:848–856. https://doi.org/10.1016/j.foodchem.2018.09.061
Feng Y-X, Ruan G-R, Jin F, Xu J, Wang F-J (2018) Purification, identification, and synthesis of five novel antioxidant peptides from Chinese chestnut (Castanea mollissima Blume) protein. hydrolysates Lwt 92:40–46. https://doi.org/10.1016/j.lwt.2018.01.006
Ghassem M, Arihara K, Mohammadi S, Sani NA, Babji AS (2017) Identification of two novel antioxidant peptides from edible bird’s nest (Aerodramus fuciphagus) protein hydrolysates. Food Funct 8:2046–2052. https://doi.org/10.1039/c6fo01615d
Gluvic A, Ulrih NP (2019) Peptides derived from food sources: antioxidative activities and interactions with model lipid membranes. Food Chem 287:324–332. https://doi.org/10.1016/j.foodchem.2019.02.092
Hao L, Gao X, Zhou T, Cao J, Sun Y, Dang Y, Pan D (2020) Angiotensin I-converting enzyme (ACE) inhibitory and antioxidant activity of umami peptides after in vitro gastrointestinal digestion. J Agric Food Chem 68:8232–8241. https://doi.org/10.1021/acs.jafc.0c02797
He S, Shi J, Walid E, Zhang H, Ma Y, Xue SJ (2015) Reverse micellar extraction of lectin from black turtle bean (Phaseolus vulgaris): optimisation of extraction conditions by response surface methodology. Food Chem 166:93–100. https://doi.org/10.1016/j.foodchem.2014.05.156
Hu F, Ci AT, Wang H, Zhang YY, Zhang JG, Thakur K, Wei ZJ (2018) Identification and hydrolysis kinetic of a novel antioxidant peptide from pecan meal using. Alcalase Food Chem 261:301–310. https://doi.org/10.1016/j.foodchem.2018.04.025
Kobbi S et al (2015) Antibacterial activity of novel peptides isolated from protein hydrolysates of RuBisCO purified from green juice alfalfa. J Funct Foods 18:703–713. https://doi.org/10.1016/j.jff.2015.09.007
Li K, Fu L, Zhao Y-Y, Xue S-W, Wang P, Xu X-L, Bai Y-H (2020) Use of high-intensity ultrasound to improve emulsifying properties of chicken myofibrillar protein and enhance the rheological properties and stability of the emulsion. Food Hydrocolloids 1:98. https://doi.org/10.1016/j.foodhyd.2019.105275
Li L-L, Li B, Ji H-F, Ma Q, Wang L-Z (2016) Immunomodulatory activity of small molecular (≤ 3 kDa) Coixglutelin enzymatic hydrolysate CyTA. J Food. https://doi.org/10.1080/19476337.2016.1201147
Li XM, Shi YH, Wang F, Wang HS, Le GW (2007) In vitro free radical scavenging activities and effect of synthetic oligosaccharides on antioxidant enzymes and lipid peroxidation in aged mice. J Pharm Biomed Anal 43:364–370. https://doi.org/10.1016/j.jpba.2006.06.041
Liu D, Chen X, Huang J, Huang M, Zhou G (2017) Generation of bioactive peptides from duck meat during post-mortem. aging Food Chem 237:408–415. https://doi.org/10.1016/j.foodchem.2017.05.094
Mirzapour M, Rezaei K, Sentandreu MA, Moosavi-Movahedi AA (2016) In vitro antioxidant activities of hydrolysates obtained from Iranian wild almond (Amygdalus scoparia) protein by several enzymes International. J Food Sci Technol 51:609–616. https://doi.org/10.1111/ijfs.12996
Mojica L, Gonzalez de Mejia E, Granados-Silvestre M, Menjivar M (2017) Evaluation of the hypoglycemic potential of a black bean hydrolyzed protein isolate and its pure peptides using in silico, in vitro and in vivo approaches. J Funct Foods 31:274–286. https://doi.org/10.1016/j.jff.2017.02.006
Najafian L, Babji AS (2014) Production of bioactive peptides using enzymatic hydrolysis and identification antioxidative peptides from patin (Pangasius sutchi) sarcoplasmic protein hydolysate. J Functi Foods 9:280–289. https://doi.org/10.1016/j.jff.2014.05.003
Neshani A, Sedighian H, Mirhosseini SA, Ghazvini K, Zare H, Jahangiri A (2020) Antimicrobial peptides as a promising treatment option against Acinetobacter baumannii infections. Microbial Pathog 1:46. https://doi.org/10.1016/j.micpath.2020.104238
Nyo MK, Nguyen LT (2017) Value-addition of defatted peanut cake by proteolysis: effects of proteases and degree of hydrolysis on functional properties and antioxidant capacity of peptides . Waste Biomass Valoriz 10:1251–1259. https://doi.org/10.1007/s12649-017-0146-0
Ren D, Zhao F, Liu C, Wang J, Guo Y, Liu J, Min W (2018) Antioxidant hydrolyzed peptides from Manchurian walnut (Juglans mandshurica Maxim.) attenuate scopolamine-induced memory impairment in mice. J Sci Food Agric 98:5142–5152. https://doi.org/10.1002/jsfa.9060
Schmeda-Hirschmann G, de Andrade JP, Jimenez-Aspee F, Mieres-Castro D (2020) A cyclic dipeptide from the Chilean hazelnut cotyledons (Gevuina avellana Mol Proteaceae) . Sci Rep 10:7070. https://doi.org/10.1038/s41598-020-63983-9
Sheng J et al (2019) Antioxidative effects and mechanism study of bioactive peptides from defatted walnut (Juglans regia L.) meal hydrolysate . J Agric Food Chem 67:3305–3312. https://doi.org/10.1021/acs.jafc.8b05722
Song R, Wei R, Zhang B, Wang D (2011) Optimization of the antibacterial activity of half-fin anchovy (Setipinna taty) hydrolysates. Food Bioprocess Technolo 5:1979–1989. https://doi.org/10.1007/s11947-010-0505-3
Sze-Tao KW, C, Sathe SK (2000) Walnuts (Juglans regia L): Proximate composition, protein solubility, protein amino acid composition and protein in vitro digestibility. J Sci Food Agric 80(9):1393–1401
Thaipong K, Boonprakob U, Crosby K, Cisneros-Zevallos L, Hawkins Byrne D (2006) Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J Food Compos Anal 19:669–675. https://doi.org/10.1016/j.jfca.2006.01.003
Tian R, Feng J, Huang G, Tian B, Zhang Y, Jiang L, Sui X (2020) Ultrasound driven conformational and physicochemical changes of soy protein hydrolysates. Ultrason Sonochem 68:105202. https://doi.org/10.1016/j.ultsonch.2020.105202
Toldra F, Reig M, Aristoy MC, Mora L (2018) Generation of bioactive peptides during food processing. Food Chem 267:395–404. https://doi.org/10.1016/j.foodchem.2017.06.119
Wali A, Yanhua G, Ishimov U, Yili A, Aisa HA, Salikhov S (2019) Isolation and identification of three novel antioxidant peptides from the bactrian camel milk hydrolysates. Int J Peptide Res Therapeut 26:641–650. https://doi.org/10.1007/s10989-019-09871-x
Wang B, Li B (2017) Effect of molecular weight on the transepithelial transport and peptidase degradation of casein-derived peptides by using Caco-2 cell model. Food Chem 218:1–8. https://doi.org/10.1016/j.foodchem.2016.08.106
Wang L, Sun J, Ding S, Qi B (2018) Isolation and identification of novel antioxidant and antimicrobial oligopeptides from enzymatically hydrolyzed anchovy fish meal. Process Biochem 74:148–155. https://doi.org/10.1016/j.procbio.2018.08.021
Wen C, Zhang J, Feng Y, Duan Y, Ma H, Zhang H (2020) Purification and identification of novel antioxidant peptides from watermelon seed protein hydrolysates and their cytoprotective effects on H2O2-induced oxidative stress. Food Chem 327:127059. doi:https://doi.org/10.1016/j.foodchem.2020.127059
Wen C, Zhang J, Zhang H, Duan Y, Ma H (2019) Effects of divergent ultrasound pretreatment on the structure of watermelon seed protein and the antioxidant activity of its hydrolysates. Food Chem 299:125165. doi:https://doi.org/10.1016/j.foodchem.2019.125165
Wu Y, Qiang Y, Cao K, Zhang W, Zhang G (2020) Inhibitory effect of the antimicrobial peptide BLP-7 against Propionibacterium acnes and its anti-inflammatory effect on acne vulgaris. Toxicon 184:109–115. https://doi.org/10.1016/j.toxicon.2020.06.006
Xia J, Song H, Huang K, Li S, Guan X (2020) Purification and characterization of antioxidant peptides from enzymatic hydrolysate of mungbean protein. J Food Sci 85:1735–1741. https://doi.org/10.1111/1750-3841.15139
Xu B et al (2020) Effect of multi-frequency power ultrasound (MFPU) treatment on enzyme hydrolysis of casein. Ultrason Sonochem 63:104930. https://doi.org/10.1016/j.ultsonch.2019.104930
You L, Zhao M, Cui C, Zhao H, Yang B (2009) Effect of degree of hydrolysis on the antioxidant activity of loach (Misgurnus anguillicaudatus) protein hydrolysates. Innov Food Sci Emerging Technol 10:235–240. https://doi.org/10.1016/j.ifset.2008.08.007
You L, Zhao M, Liu RH, Regenstein JM (2011) Antioxidant and antiproliferative activities of loach (Misgurnus anguillicaudatus) peptides prepared by papain digestion. J Agric Food Chem 59:7948–7953. https://doi.org/10.1021/jf2016368
Zamora-Sillero J, Gharsallaoui A, Prentice C (2018) Peptides from fish by-product protein hydrolysates and its functional properties: an overview . Mar Biotechnol (NY) 20:118–130. https://doi.org/10.1007/s10126-018-9799-3
Zhang Y, Zhang N, Wang Q, Wang P, Yu Y (2020) A facile and eco-friendly approach for preparation of microkeratin and nanokeratin by ultrasound-assisted enzymatic hydrolysis. Ultrasonics Sonochem. https://doi.org/10.1016/j.ultsonch.2020.105201
Zhao J, He J, Dang Y, Cao J, Sun Y, Pan D (2020) Ultrasound treatment on the structure of goose liver proteins and antioxidant activities of its enzymatic hydrolysate. J Food Biochem 44:e13091. https://doi.org/10.1111/jfbc.13091
Zhou D-Y et al (2012) Antioxidant activity of hydrolysates obtained from scallop (Patinopecten yessoensis) and abalone (Haliotis discus hannai Ino) muscle. Food Chem 132:815–822. https://doi.org/10.1016/j.foodchem.2011.11.041
Zou TB, He TP, Li HB, Tang HW, Xia EQ (2016) The structure-activity relationship of the antioxidant peptides from natural proteins. Molecules 21:72. https://doi.org/10.3390/molecules21010072
Acknowledgements
This research was funded by the key research and development program of Zhejiang Province (No. 2020C02040; 2020C02019), the cooperation projects between the People’s Government of Zhejiang Province and the Chinese Academy of Forestry (2019SY03), and the Public Projects of Zhejiang Province (LGC19C200008).
Author information
Authors and Affiliations
Contributions
Shaozhen Wu designed and performed experiments, analyzed data and co-wrote the paper. Zhiping He and Fenghua Wu co-wrote the paper and review it. Qingqing Wang assisted with the design and perform experiments. Xingquan Liu supervised and edited the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they do not have any conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wu, S., He, Z., Wang, Q. et al. Response Surface Optimization of Enzymatic Hydrolysis of Peptides of Chinese Pecan (Carya cathayensis) and Analysis of Their Antioxidant Capacities and Structures. Int J Pept Res Ther 27, 1239–1251 (2021). https://doi.org/10.1007/s10989-021-10164-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-021-10164-5