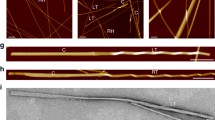
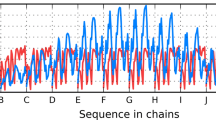
Abstract
Protein deposition as amyloid fibrils underlies more than twenty severely debilitating human disorders. Interestingly, recent studies suggest that all peptides and proteins possess an intrinsic ability to assemble into amyloid fibrils similar to those observed in disease states. The common properties and characteristics of amyloid aggregates thus offer the prospect that simple model systems can be used to systematically assess the factors that predispose a native protein to form amyloid fibrils and understand the origin and progression of fatal disorders associated with amyloid formation. Here, we report the de novo design of a 17-residue peptide model system, referred to as ccβ, which forms a protein-like coiled-coil structure under ambient solution conditions but can be easily converted into amyloid fibrils by raising the temperature. Oxidation of methionine residues at selected hydrophobic positions completely abolished amyloid fibril formation of the peptide while not interfering with its coiled-coil structure. This finding indicates that a small number of site-specific hydrophobic interactions can play a major role in the packing of polypeptide chain segments within amyloid fibrils. The simplicity and characteristics of the ccβ system make it highly suitable for probing molecular details of the assembly of amyloid structures.
Similar content being viewed by others
Abbreviations
- CD:
-
circular dichroism
- Fmoc:
-
9-fluorenylmethoxycarbonyl
- EDT:
-
1,2-ethanedithiol
- GRAVY:
-
Grand average hydrophaty
- HPLC:
-
high-performance liquid chromatography
- MALDI-TOF:
-
matrix-assisted laser-desorption ionization time-of-flight mass spectroscopy
- MBHA-resin:
-
4-methylbenzhydrylamine-resin
- PAL:
-
tris(alkoxy)benzylamide linker
- PBS:
-
phosphate buffered saline
- PEG:
-
polyethylene glycol
- TEM:
-
transmission electron microscope
- TPTU:
-
O-(1,2-dihydro-2-oxo-pyridyl)-N, N, N′,N′-tetramethyluronium tetrafluoroborate
- TFA:
-
trifluoroacetic acid
- UV:
-
ultraviolet
References
A. Aguzzi C. Haass (2003) Science 302 814
F. Albericio N. Kneib-Cordonier S. Biancalana L. Gera R. I. Masada D. Hudson G. Barany (1990) J. Org. Chem 55 3730
M. Balbirnie R. Grothe D. S. Eisenberg (2001) Proc. Natl. Acad. Sci. U.S.A 98 2375
T. L. Benzinger D. M. Gregory T. S. Burkoth H. Miller-Auer D. G. Lynn R. E. Botto S. C. Meredith (1998) Proc. Natl. Acad. Sci. U.S.A 95 13407
D. R. Booth M. Sunde V. Bellotti C. V. Robinson W. L. Hutchinson P. E. Fraser P. N. Hawkins C. M. Dobson S. E. Radford C. C. Blake M. B. Pepys (1997) Nature 385 787 Occurrence Handle10.1038/385787a0
M. Bucciantini E. Giannoni F. Chiti F. Baroni L. Formigli J. Zurdo N. Taddei G. Ramponi C. M. Dobson M. Stefani (2002) Nature 416 507 Occurrence Handle10.1038/416507a Occurrence Handle1:CAS:528:DC%2BD38XivVOgtbg%3D Occurrence Handle11932737
M. G. Burke R. Woscholski S. N. Yaliraki (2003) Proc. Natl. Acad. Sci. U.S.A 100 13928
P. Burkhard M. Meier A. Lustig (2000) Protein Sci 9 2294
P. Burkhard J. Stetefeld S. V. Strelkov (2001) Trends Cell Biol 11 82 Occurrence Handle10.1016/S0962-8924(00)01898-5 Occurrence Handle1:CAS:528:DC%2BD3MXpslyktg%3D%3D Occurrence Handle11166216
R. Cerpa F. E. Cohen I. D. Kuntz (1996) Fold. Des 1 91
A. Chakrabartty R. L. Baldwin (1995) Adv. Protein Chem 46 141
T. Chiba Y. Hagihara T. Higurashi K. Hasegawa H. Naiki Y. Goto (2003) J. Biol. Chem 278 47016
F. Chiti P. Webster N. Taddei A. Clark M. Stefani G. Ramponi C. M. Dobson (1999) Proc. Natl. Acad. Sci. U.S.A 96 3590
F. Chiti N. Taddei M. Bucciantini P. White G. Ramponi C. M. Dobson (2000) EMBO J 19 1441
F. Chiti N. Taddei F. Baroni C. Capanni M. Stefani G. Ramponi C. M. Dobson (2002) Nat. Struct. Biol 9 137
F. Chiti M. Stefani N. Taddei G. Ramponi C.␣M. Dobson (2003) Nature 424 805
B. Ciani E. G. Hutchinson R. B. Sessions D. N. Woolfson (2002) J. Biol. Chem 277 10150
C. Cohen D. A. Parry (1990) Proteins 7 1
A. S. Cohen T. Shirahama M. Skinner (1982) Electron microscopy of amyloid J. R. Harris (Eds) Electron microscopy of proteins Academic Press London, UK
Dado, P. G. and Gellman, S. H.: 1993, J. Am. Chem. Soc. , 12609.
W. F. Grado ParticleDe C. M. Summa V. Pavone F. Nastri A. Lombardi (1999) Annu. Rev. Biochem 68 779
M. Fandrich M. A. Fletcher C. M. Dobson (2001) Nature 410 165
Y. Fezoui D. M. Hartley D. M. Walsh D. J. Selkoe J. J. Osterhout D. B. Teplow (2000) Nat. Struct. Biol 7 1095
S. Frank A. Lustig T. Schulthess J. Engel R.A. Kammerer (2000) J. Biol. Chem 275 11672
C. García-Echeverría (1996) Bioorg. Med. Chem. Lett 6 229
P. Hammarstrom X. Jiang A. R. Hurshman E. T. Powers J. W. Kelly (2002) Proc. Natl. Acad. Sci. U.S.A 99 IssueID4 16427
P. B. Harbury T. Zhang P. S. Kim T. Alber (1993) Science 262 1401
M. H. Hecht A. Das A. Go L. H. Bradley Y. Wei (2004) Protein Sci 13 1711
M. R. Hurle L. R. Helms L. Li W. Chan R. Wetzel (1994) Proc. Natl. Acad. Sci. U.S.A 91 5446
K. Janek J. Behlke J. Zipper H. Fabian Y. Georgalis M. Beyermann M. Bienert E. Krause (1999) Biochemistry 38 8246
R. A. Kammerer (1997) Matrix Biol 15 555
R. A. Kammerer T. Schulthess R. Landwehr A. Lustig J. Engel U. Aebi M. O. Steinmetz (1998) Proc. Natl. Acad. Sci. U.S.A 95 13419
R. A. Kammerer V. A. Jaravine S. Frank T. Schulthess R. Landwehr A. Lustig C. Garcia-Echeverria A. T. Alexandrescu J. Engel M. O. Steinmetz (2001) J Biol. Chem 276 13685
R. A. Kammerer D. Kostrewa J. Zurdo A. Detken C. Garcia-Echeverria J. D. Green S. A. Muller B. H. Meier F. K. Winkler C. M. Dobson M. O. Steinmetz (2004) Proc. Natl. Acad. Sci. U.S.A 101 4435
J. W. Kelly W. E. Balch (2003) J. Cell Biol 161 461
W. D. Kohn R. S. Hodges (1998) Trends Biotechnol 16 379
J. Kyte R. F. Doolittle (1982) J Mol. Biol 157 105
P. T. Lansbury SuffixJr. (1999) Proc. Natl. Acad. Sci. U.S.A 96 3342
S. Liemann R. Glockshuber (1999) Biochemistry 38 3258 Occurrence Handle10.1021/bi982714g Occurrence Handle1:CAS:528:DyaK1MXht12hu7w%3D Occurrence Handle10079068
P. M. Lopez ParticleDe La K. Goldie J. Zurdo E. Lacroix C.M. Dobson A. Hoenger L. Serrano (2002) Proc. Natl. Acad. Sci. U.S.A 99 16052
M. Lu W. Shu H. Ji E. Spek L. Wang N. R. Kallenbach (1999) J Mol. Biol 288 743
A. Lupas (1996) Trends Biochem. Sci 21 375
S. L. McCutchen Z. Lai G. J. Miroy J. W. Kelly W. Colon (1995) Biochemistry 34 13527
D. L. Minor SuffixJr. P. S. Kim (1994) Nature 367 660 Occurrence Handle10.1038/367660a0 Occurrence Handle1:CAS:528:DyaK2cXitFWjtLo%3D Occurrence Handle8107853
A. D. Miranker (2004) Proc. Natl. Acad. Sci. U.S.A 101 4335
L. Nielsen S. Frokjaer J. Brange V. N. Uversky A.L. Fink (2001) Biochemistry 40 8397
D. E. Otzen O. Kristensen M. Oliveberg (2000) Proc. Natl. Acad. Sci. U.S.A 97 9907
M. F. Perutz (1999) Trends Biochem. Sci 24 58
M. F. Perutz T. Johnson M. Suzuki J. T. Finch (1994) Proc. Natl. Acad. Sci. U.S.A 91 5355
S. B. Prusiner (2001) N. Engl. J. Med 344 1516
M. Ramirez-Alvarado J. S. Merkel L. Regan (2000) Proc. Natl. Acad. Sci. U.S.A 97 8979
J. S. Richardson D. C. Richardson (1998) Science 240 1648
J. C. Rochet P. T. Lansbury SuffixJr (2000) Curr. Opin. Struct. Biol 10 60
J. C. Sacchettini J. W. Kelly (2002) Nat. Rev. Drug Discov 1 267
L. C. Serpell M. Sunde C. C. Blake (1997) Cell Mol. Life Sci 53 871
D. P. Smith S. Jones L. C. Serpell M. Sunde S. E. Radford (2003) J. Mol. Biol 330 943
M. Stefani C. M. Dobson (2003) J. Mol. Med 81 678
M. O. Steinmetz A. Stock T. Schulthess R. Landwehr A. Lustig J. Faix G. Gerisch U. Aebi R. A. Kammerer (1998) EMBO J 17 1883
J. Y. Su R. S. Hodges C. M. Kay (1994) Biochemistry 33 15501
M. Sunde L. C. Serpell M. Bartlam P. E. Fraser M. B. Pepys C. C. Blake (1997) J. Mol. Biol 273 729
Y. Takahashi A. Ueno H. Mihara (2000) Structure. Fold. Des 8 915
J. P. Taylor J. Hardy K. H. Fischbeck (2002) Science 296 1991
K. Tenidis M. Waldner J. Bernhagen W. Fischle M. Bergmann M. Weber M. L. Merkle W. Voelter H. Brunner A. Kapurniotu (2000) J. Mol. Biol 295 1055
L. O. Tjernberg D. J. Callaway A. Tjernberg S. Hahne C. Lilliehook L. Terenius J. Thyberg C. Nordstedt (1999) J. Biol. Chem 274 12619
L. Tjernberg W. Hosia N. Bark J. Thyberg J. Johansson (2002a) J. Biol. Chem 277 43243
L. O. Tjernberg A. Tjernberg N. Bark Y. Shi B. P. Ruzsicska Z. Bu J. Thyberg D. J. Callaway (2002b) Biochem. J 366 343
V. Villegas J. Zurdo V. V. Filimonov F. X. Aviles C. M. Dobson L. Serrano (2000) Protein Sci 9 1700
D. M. Walsh D. M. Hartley Y. Kusumoto Y. Fezoui M. M. Condron A. Lomakin G. B. Benedek D. J. Selkoe D. B. Teplow (1999) J. Biol. Chem 274 25945
M. W. West W. Wang J. Patterson J. D. Mancias J. R. Beasley M. H. Hecht (1999) Proc. Natl. Acad. Sci. U.S.A 96 11211
S. Zhang (2003) Nat. Biotechnol 21 1171
S. Zhang A. Rich (1997) Proc. Natl. Acad. Sci. U.S.A 94 23
L. Zhu X. J. Zhang L. Y. Wang J. M. Zhou S. Perrett (2003) J. Mol. Biol 328 235
Author information
Authors and Affiliations
Corresponding author
Additional information
Abbreviations used for amino acids follow the recommendations of the IUPAC-IUB Commission of Biochemical Nomenclature [Eur. J. Biochem., 138 (1984) 9].
Rights and permissions
About this article
Cite this article
Steinmetz, M.O., García-Echeverría, C. & Kammerer, R.A. Design of a Coiled-Coil-based Model Peptide System to Explore the Fundamentals of Amyloid Fibril Formation. Int J Pept Res Ther 11, 43–52 (2005). https://doi.org/10.1007/s10989-004-1720-4
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10989-004-1720-4