Abstract
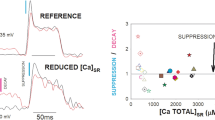
MagFluo-4 fluorescence (Ca2+) transients associated with action potentials were measured in intact muscle fibres, manually dissected from toads (Leptodactylus insularis) or enzymatically dissociated from mice. In toads, the decay phase of the Ca2+ transients is described by a single exponential with a time constant (τ) of about 7 ms. In mice, a double exponential function with τ's of 1.5 and 15.5 ms, respectively gives a better fit. In both species the amplitude of Ca2+ transients diminished during repetitive stimulation: in amphibian muscle fibres, the decrease was about 20% with 1 Hz stimulation and 55% at 10 Hz. In mammalian fibres, repetitive stimulation causes a less conspicuous decrease of the transient amplitude: 10% at 1 Hz and 15 % at 10 Hz. During tetanic stimulation at 100 Hz the transient amplitude decays to 20 % in toad fibres and 40 % in mouse fibres. This decrease could be associated with the phenomenon of inactivation of Ca2+ release, described by other authors. Recovery from inactivation, studied by a double stimuli protocol also indicates that in toad fibres the ability to release Ca2+ is abolished to a greater extent than in mouse fibres. In fact the ratio between the amplitudes of the second and first transient, when they are separated by a 10 ms interval, is 0.29 for toad and 0.58 for mouse fibres. In toad fibres, recovery from inactivation, to about 80 % of the initial value, occurs with a τ of 32 ms at 22 °C; while in mouse fibres recovery from inactivation is almost complete and occurs with a τ of 36 ms under the same conditions. The results indicate that Ca2+ release in enzymatically dissociated mammalian muscle fibres inactivates to a smaller extent than in intact amphibian muscle fibres.
Similar content being viewed by others
References
Baylor SM, Chandler WS and Marshall MW (1983) Sarcoplasmic reticulum calcium release in frog skeletal muscle fibres estimated from Arsenazo III calcium transients. J Physiol (Lond) 344: 625–666.
Baylor SM and Hollingworth S (1988) Fura-2 calcium transients in frog skeletal muscle fibres [published erratum appears in J Physiol (Lond) 1988 Dec;407:616]. J Physiol (Lond) 403: 151–192.
Baylor SM and Hollingworth S (2003) Sarcoplasmic reticulum calcium release compared in slow-twitch and fast-twitch fibres of mouse muscle. J Physiol (Lond) 551: 125–138.
Block BA, Imagawa T, Campbell KP and Franzini-Armstrong C (1988) Structural evidence for direct interaction between the molecular components of the transverse tubule/sarcoplasmic reticulum junction in skeletal muscle. J Cell Biol 107: 2587–2600.
Brum G, Rios E and Stefani E (1988) Effects of extracellular calcium on calcium movements of excitation-contraction coupling in frog skeletal muscle fibres. J Physiol (Lond) 398: 441–473.
Caputo C and Bolaños P (1994) Fluo-3 signals associated with potassium contractures in single amphibian muscle fibres. J Physiol (Lond) 481: 119–128.
Caputo C and Bolaños P (2002) Inactivation of Ca2+ release in amphibian and mammalian skeletal muscle fibres. Biophys J 82: 641a.
Carroll SL, Klein M and Schneider MF (1995) Calcium transients in intact rat skeletal muscle fibres in agarose gel. Am J Physiol 269: C28–34.
Carroll SL, Klein MG and Schneider MF (1997) Decay of calcium transients after electrical stimulation in rat fast-and slow-twitch skeletal muscle fibres. J Physiol (Lond) 501: 573–588.
Endo M (1977) Calcium release from the Sarcoplasmic Reticulum. Physiol Rev 57: 71–108.
Felder E and Franzini-Armstrong C (2002) Type 3 ryanodine receptors of skeletal muscle are segregated in a parajunctional position. Proc Natl Acad Sci USA 99: 1695–1700.
Franzini-Armstrong C and Protasi F (1997) Ryanodine receptors of striated muscles: a complex channel capable of multiple interactions. Physiol Rev 77: 699–729.
Garcia J and Schneider MF (1993) Calcium transients and calcium release in rat fast-twitch skeletal muscle fibres. J Physiol (Lond) 463: 709–728.
Gonzalez A and Rios E (1993) Perchlorate enhances transmission in skeletal muscle excitation–contraction coupling. J Gen Physiol 102: 373–421.
Harkins AB, Kurebayashi N and Baylor SM (1993) Resting myoplasmic free calcium in frog skeletal muscle fibres estimated with fluo-3 [see comments]. Biophys J 65: 865–881.
Hollingworth S, Zhao M and Baylor SM (1996) The amplitude and time course of the myoplasmic free [Ca2+] transient in fast-twitch fibres of mouse muscle. J Gen Physiol 108: 455–469.
Jacquemond V, Csernoch L, Klein MG and Schneider MF (1991) Voltage-gated and calcium-gated calcium release during depolarization of skeletal muscle fibres. Biophys J 60: 867–873.
Jong DS, Pape PC, Baylor SM and Chandler WK (1995) Calcium inactivation of calcium release in frog cut muscle fibres that contain millimolar EGTA or Fura-2. J Gen Physiol 106: 337–388.
Jong DS, Pape PC, Chandler WK and Baylor SM (1993) Reduction of calcium inactivation of sarcoplasmic reticulum calcium release by fura-2 in voltage-clamped cut twitch fibres from frog muscle. J Gen Physiol 102: 333–370.
Klein MG, Simon BJ, Szucs G and Schneider MF (1988) Simultaneous recording of calcium transients in skeletal muscle using high-and low-affinity calcium indicators. Biophys J 53: 971–988.
Konishi M, Hollingworth S, Harkins AB and Baylor SM (1991) Myoplasmic calcium transients in intact frog skeletal muscle fibres monitored with the fluorescent indicator furaptra. J Gen Physiol 97: 271–301.
Lamb GD (1992) DHP receptors and excitation-contraction coupling. J Muscle Res Cell Motil 13: 394–405.
Meissner G (1994) Ryanodine receptor/Ca2+ release channels and their regulation by endogenous effectors. Ann Rev Physiol 56: 485–508.
Melzer W, Rios E and Schneider MF (1984) Time course of calcium release and removal in skeletal muscle fibres. Biophys J 45: 637–641.
Melzer W, Rios E and Schneider MF (1986) The removal of myoplasmic free calcium following calcium release in frog skeletal muscle. J Physiol (Lond) 372: 261–292.
Melzer W, Rios E and Schneider MF (1987) A general procedure for determining the rate of calcium release from the sarcoplasmic reticulum in skeletal muscle fibres. Biophys J 51: 849–863.
Murayama T, Kurebayashi N and Ogawa Y (2000) Role of Mg2+ in Ca2+ induced Ca2+ release through ryanodine receptors of frog skeletal muscle: modulation by adenine nucleotides and caffeine. Biophys J 78: 1810–1824.
Murayama T and Ogawa Y (1992) Purification and characterization of two ryanodine-binding protein isoforms from sarcoplasmic reticulum of bullfrog skeletal muscle. J Biochem 112: 514–522.
Murayama T and Ogawa Y (2001) Selectively suppressed Ca2+-induced Ca2+ release activity of a-Ryanodine receptor (a-RyR) in frog skeletal muscle sarcoplasmic reticulum. J Biol Chem 276: 2953–2960.
Murayama T and Ogawa Y (2002) Roles of two ryanodine receptor isoforms coexisting in skeletal muscle. Trends Cardiovasc Med 12: 305–311.
Ogawa Y, Kurebayashi N and Murayama T (2000) Putative roles of the type 3 ryanodine receptor isoforms (RyR3). Trends Cardiovasc Med 10: 65–70.
Pape PC, Fenelon K and Carrier N (2002) Extra activation component of calcium release in frog muscle fibres. J Physiol (Lond) 542: 867–886.
Pape PC, Jong DS, Chandler WK and Baylor SM (1993) Effect of fura-2 on action potential-stimulated calcium release in cut twitch fibres from frog muscle. J Gen Physiol 102: 295–332.
Posterino GS and Lamb GD (2003) Effect of sarcoplasmic reticulum Ca2+ content on action potential-induced Ca+2 release in rat skeletal muscle fibres. J Physiol (Lond) 551: 219–237.
Rios E and Brum G (1987) Involvement of dihydropyridine receptors in excitation-contraction coupling in skeletal muscle. Nature 325: 717–720.
Rios E and Pizarro G (1988) Voltage sensors and calcium channels of excitation–contraction coupling. News Physiol Sci 3: 223–227.
Rios E and Pizarro G (1991) Voltage sensor of excitation-contraction coupling in skeletal muscle. Physiol Rev 71: 849–908.
Rios E and Stern MD (1997) Calcium in close quarters: microdomain feedback in excitation–contraction coupling and other cell biological phenomena. Ann Rev Biophys Biomol Struct 26: 47–82.
Schneider MF and Chandler WK (1973) Voltage dependent charge movement of skeletal muscle: a possible step in excitationcontraction coupling. Nature 242: 244–246.
Schneider MF and Simon BJ (1988) Inactivation of calcium release from the sarcoplasmic reticulum in frog skeletal muscle. J Physiol (Lond) 405: 727–745.
Schneider MF, Simon BJ and Szucs G (1987) Depletion of calcium from the sarcoplasmic reticulum during calcium release in frog skeletal muscle. J Physiol (Lond) 392: 167–192.
Shirokova N, Garcia J, Pizarro G and Rios E (1996) Ca2+ release from the sarcoplasmic reticulum compared in amphibian and mammalian skeletal muscle. J Gen Physiol 107: 1–18.
Simon BJ, Klein MG and Schneider MF (1991) Calcium dependence of inactivation of calcium release from the sarcoplasmic reticulum in skeletal muscle fibres. J Gen Physiol 97: 437–471.
Simon BJ and Schneider MF (1988) Time course of activation of calcium release from sarcoplasmic reticulum in skeletal muscle. Biophys J 54: 1159–1163.
Simon BJ, Schneider MF and Szucs G (1985) Inactivation of sarcoplasmic reticulum calcium release in frog skeletal muscle is mediated by calcium. J Gen Physiol 86: 36A.
Sonnleitner A, Conti A, Bertocchini F, Schindler H and Sorrentino V (1998) Functional properties of the ryanodine receptor type 3 (RyR3) Ca2+ release channel. EMBO J 17: 2790–2798.
Sorrentino V and Reggiani C (1999) Expression of the ryanodine receptor type 3 in skeletal muscle. A new partner in excitation– contraction coupling? Trends Cardiovasc Med 9: 54–61.
Sun X, Protasi F, Takahasi M, Takeshima H, Ferguson DG and Franzini-Armstrong C (1995) Molecular architecture of membranes involved in excitation–contraction coupling of cardiac muscle. J Cell Biol 129: 659–671.
Tanabe T, Beam KG, Powell JA and Numa S (1988) Restoration of excitation-contraction coupling and slow calcium current in dysgenic muscle by dihydropyridine receptor complementary DNA. Nature 336: 134–139.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Caputo, C., Bolaños, P. & Gonzalez, A. Inactivation of Ca2+ transients in amphibian and mammalian muscle fibres. J Muscle Res Cell Motil 25, 315–328 (2004). https://doi.org/10.1007/s10974-004-4071-z
Issue Date:
DOI: https://doi.org/10.1007/s10974-004-4071-z