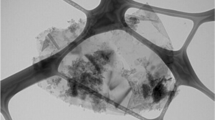
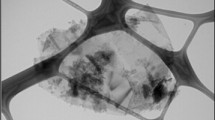
Abstract
The aim of the research work reports the thermal performance of graphene-water-based nanofluid phase change material (NFPCM) with different types of surfactants for a cold thermal storage (CTS) system. The graphene nanoplatelets (GnPs) are dispersed in DI water with the addition of commonly used surfactants, such as cetyltrimethylammonium bromide, sodium dodecylbenzene sulfonate (SDBS), sodium dodecylsulfate (SDS), polysorbate (Tween80), polyvinylpyrrolidone (PVP), and gum arabic (GA). The effect on the stability of different surfactants on the colloidal solution of GnP nanofluids is analyzed by zeta potential and particle size distribution. The average zeta potential values indicate that the SDBS-GnP, SDS-GnP, and GA-GnP combinations have good stability after 72 h. The freezing experiment is performed at − 7 °C cooling bath temperature in a spherical container and reveals that the surfactants have a significant influence on the supercooling rate and freezing time. The maximum reduction of 92.16% and 84.20% is observed for GA-GnP and SDBS-GnP in the supercooling rate, respectively. The prepared NFPCM can be incorporated in the CTS system owing to its enhanced thermal transport properties to increase the energy-saving potential. Also, the reduction of supercooling facilitates the chiller to operate at higher temperatures.















Similar content being viewed by others
Data availability
The authors declare that all the data supporting the findings of this study are available within the article.
Abbreviations
- T :
-
Temperature/°C
- t :
-
Time/s
- f :
-
Freezing
- n :
-
Supercooling
- \(\dot{\mathrm{q }}\) :
-
Average surface heat flux/W m−2
- m :
-
Mass of PCM/kg
- λ :
-
Latent heat/KJ kg−1
- S :
-
Uncertainty
- logg:
-
Data logger
- ini:
-
Initial
- fin:
-
Final
- DI :
-
Deionized
- BF:
-
Base fluid
- GA:
-
Gum arabic
- PCM :
-
Phase change material
- DSC :
-
Differential scanning calorimetry
- RTD :
-
Resistance temperature detector
- PUF:
-
Polyurethane foam
- TEM :
-
Transmission electron microscope
- HTF :
-
Heat transfer fluid
- GnP :
-
Graphene nanoplatelet
- SDS:
-
Sodium dodecyl sulfate
- NPE:
-
Nonylphenolethoxylate
- PVP:
-
Polyvinylpyrrolidone
- HRSEM:
-
High-resolution scanning electron microscopy
- MWCNT:
-
Multi-wall carbon nanotubes
- CTES :
-
Cool thermal energy storage
- LTES:
-
Latent heat thermal energy storage
- LDPE :
-
Low-density polyethylene
- PTDC :
-
Proportionate temperature differential controller
- NFPCM :
-
Nanofluid phase change material
- SDBS:
-
Sodium dodecylbenzene sulfonate
- CTAB:
-
Cetyltrimethylammonium bromide
References
Souayfane F, Fardoun F, Biwole PH. Phase change materials (PCM) for cooling applications in buildings: a review. Energy Build. 2016;129:396–431. https://doi.org/10.1016/j.enbuild.2016.04.006.
Velraj R, Cheralathan M, Renganarayanan S. Energy management through encapsulated PCM based storage system for large building air conditioning application. Int Energy J. 2006;7:253–9.
Sidik NAC, Kean TH, Chow HK, Rajaandra A, Rahman S, Kaur J. Performance enhancement of cold thermal energy storage system using nanofluid phase change materials: a review. Int Commun Heat Mass Transf. 2018;94:85–95. https://doi.org/10.1016/j.icheatmasstransfer.2018.03.024.
Salunkhe PB, Shembekar PS. A review on effect of phase change material encapsulation on the thermal performance of a system. Renew Sustain Energy Rev. 2012. https://doi.org/10.1016/j.rser.2012.05.037.
Nazir H, Batool M, Bolivar Osorio FJ, Isaza-Ruiz M, Xu X, Vignarooban K, et al. Recent developments in phase change materials for energy storage applications: a review. Int J Heat Mass Transf. 2019;129:491–523. https://doi.org/10.1016/j.ijheatmasstransfer.2018.09.126.
Li G, Hwang Y, Radermacher R, Chun HH. Review of cold storage materials for subzero applications. Energy. 2013;51:1–17. https://doi.org/10.1016/j.energy.2012.12.002.
Chandrasekaran P, Cheralathan M, Velraj R. Effect of fill volume on solidification characteristics of DI (deionized) water in a spherical capsule - An experimental study. Energy. 2015;90:508–15. https://doi.org/10.1016/j.energy.2015.07.086.
Zou T, Fu W, Liang X, Wang S, Gao X, Zhang Z, Fang Y. Preparation and performance of modified calcium chloride hexahydrate composite phase change material for air-conditioning cold storage. Int J Refrig. 2018;95:175–81. https://doi.org/10.1016/j.ijrefrig.2018.08.001.
Vikram MP, Kumaresan V, Christopher S, Velraj R. Experimental studies on solidification and subcooling characteristics of water-based phase change material (PCM) in a spherical encapsulation for cool thermal energy storage applications. Int J Refrig. 2019;100:454–62. https://doi.org/10.1016/j.ijrefrig.2018.11.025.
Nematpour Keshteli A, Sheikholeslami M. Nanoparticle enhanced PCM applications for intensification of thermal performance in building: a review. J Mol Liq. 2019;274:516–33. https://doi.org/10.1016/j.molliq.2018.10.151.
Prabakaran R, Sidney S, Lal DM, Selvam C, Harish S. Solidification of graphene-assisted phase change nanocomposites inside a sphere for cold storage applications. Energies. 2019. https://doi.org/10.3390/en12183473.
Sathishkumar A, Cheralathan M. Influence of thermal transport properties of NEPCM for cool thermal energy storage system. J Therm Anal Calorim. 2020. https://doi.org/10.1007/s10973-020-10339-0.
Altohamy AA, Abd Rabbo MF, Sakr RY, Attia AAA. Effect of water based Al2O3 nanoparticle PCM on cool storage performance. Appl Therm Eng. 2015;84:331–8. https://doi.org/10.1016/j.applthermaleng.2015.03.066.
Shah KW. A review on enhancement of phase change materials—A nanomaterials perspective. Energy Build. 2018;175:57–68. https://doi.org/10.1016/j.enbuild.2018.06.043.
Kumaresan V, Chandrasekaran P, Nanda M, Maini AK, Velraj R. Role of PCM based nanofluids for energy efficient cool thermal storage system. Int J Refrig. 2013;36:1641–7. https://doi.org/10.1016/j.ijrefrig.2013.04.010.
Munyalo JM, Zhang X, Xu X. Experimental investigation on supercooling, thermal conductivity and stability of nanofluid based composite phase change material. J Energy Storage. 2018;17:47–55. https://doi.org/10.1016/j.est.2018.02.006.
Kim SC, Prabakaran R, Sakthivadivel D, Thangapandian N, Bhatia A, Kumar PG. Thermal transport properties of carbon-assisted phase change nanocomposite. Fullerenes Nanotub Carbon Nanostruct. 2020. https://doi.org/10.1080/1536383X.2020.1786814.
Sathishkumar A, Kumaresan V, Velraj R. Solidification characteristics of water based graphene nanofluid PCM in a spherical capsule for cool thermal energy storage applications. Int J Refrig. 2016;66:73–83. https://doi.org/10.1016/j.ijrefrig.2016.01.014.
Zahir MH, Mohamed SA, Saidur R, Al-Sulaiman FA. Supercooling of phase-change materials and the techniques used to mitigate the phenomenon. Appl Energy. 2019;240:793–817. https://doi.org/10.1016/j.apenergy.2019.02.045.
Texter J. Graphene dispersions. Curr Opin Colloid Interface Sci. 2014;19:163–74. https://doi.org/10.1016/j.cocis.2014.04.004.
Mingzheng Z, Guodong X, Jian L, Lei C, Lijun Z. Analysis of factors influencing thermal conductivity and viscosity in different kinds of surfactant solutions. Exp Therm Fluid Sci. 2012;36:22–9. https://doi.org/10.1016/j.expthermflusci.2011.07.014.
Xian HW, Sidik NAC, Saidur R. Impact of different surfactants and ultrasonication time on the stability and thermophysical properties of hybrid nanofluids. Int Commun Heat Mass Transf. 2020. https://doi.org/10.1016/j.icheatmasstransfer.2019.104389.
Le T, Omid B, Somchai M, Szilágyi IM. Review on the recent progress in the preparation and stability of graphene-based nanofluids modified hummers method. J Therm Anal Calorim. 2020;142:1145–72. https://doi.org/10.1007/s10973-020-09365-9.
Nazari B, Ranjbar Z, Hashjin RR, Rezvani Moghaddam A, Momen G, Ranjbar B. Dispersing graphene in aqueous media: Investigating the effect of different surfactants. Colloid Surfaces A Physicochem Eng Asp. 2019. https://doi.org/10.1016/j.colsurfa.2019.123870.
Gallego A, Cacua K, Herrera B, Cabaleiro D, Piñeiro MM, Lugo L. Experimental evaluation of the effect in the stability and thermophysical properties of water-Al2O3 based nanofluids using SDBS as dispersant agent. Adv Powder Technol. 2020;31:560–70. https://doi.org/10.1016/j.apt.2019.11.012.
Wang J, Li G, Li T, Zeng M, Sundén B. Effect of various surfactants on stability and thermophysical properties of nanofluids. J Therm Anal Calorim. 2020. https://doi.org/10.1007/s10973-020-09381-9.
Calderón A, Barreneche C, Hernández-Valle K, Galindo E, Segarra M, Fernández AI. Where is Thermal energy storage (TES) research going? – A bibliometric analysis. Sol Energy. 2020;200:37–50. https://doi.org/10.1016/j.solener.2019.01.050.
Jia L, Peng L, Chen Y, Mo S, Li X. Improving the supercooling degree of titanium dioxide nanofluids with sodium dodecylsulfate. Appl Energy. 2014;124:248–55. https://doi.org/10.1016/j.apenergy.2014.03.019.
Yoon S, In I. Role of poly(N-vinyl-2-pyrrolidone) as stabilizer for dispersion of graphene via hydrophobic interaction. J Mater Sci. 2011;46:1316–21. https://doi.org/10.1007/s10853-010-4917-2.
Rashmi W, Ismail AF, Sopyan I, Jameel AT, Khalid M, Mubarak NM. Stability and thermal conductivity enhancement of carbon nanotube nanofluid using gum arabic. J Exp Nanosci. 2011. https://doi.org/10.1080/17458080.2010.487229.
Askari S, Lotfi R, Seifkordi A, Rashidi AM, Koolivand H. A novel approach for energy and water conservation in wet cooling towers by using MWNTs and nanoporous graphene nanofluids. Energy Convers Manag. 2016;109:10–8. https://doi.org/10.1016/j.enconman.2015.11.053.
Sarsam WS, Amiri A, Kazi SN, Badarudin A. Stability and thermophysical properties of non-covalently functionalized graphene nanoplatelets nanofluids. Energy Convers Manag. 2016;116:101–11. https://doi.org/10.1016/j.enconman.2016.02.082.
Moffat RJ. Describing the uncertainties in experimental results. Exp Therm Fluid Sci. 1988;1:3–17.
Godson L, Deepak K, Enoch C, Jefferson Raja BR, Raja B. Heat transfer characteristics of silver/water nanofluids in a shell and tube heat exchanger. Arch Civ Mech Eng. 2014;14:489–96. https://doi.org/10.1016/j.acme.2013.08.002.
Prabakaran R, Naveen JP, Dhasan K, Lal M. Constrained melting of graphene-based phase change nanocomposites inside a sphere. J Therm Anal Calorim. 2020;139:941–52. https://doi.org/10.1007/s10973-019-08458-4.
Ilyas SU, Ridha S, Abdul Kareem FA. Dispersion stability and surface tension of SDS-Stabilized saline nanofluids with graphene nanoplatelets. Colloids Surfaces A Physicochem Eng Asp. 2020;592: 124584. https://doi.org/10.1016/j.colsurfa.2020.124584.
Prabakaran R, Sidney S, Lal DM, Harish S, Kim SC. Experimental performance of a mobile air conditioning unit with small thermal energy storage for idle stop/start vehicles. J Therm Anal Calorim. 2021. https://doi.org/10.1007/s10973-021-10863-7.
Akyurt M, Zaki G, Habeebullah B. Freezing phenomena in ice-water systems. Energy Convers Manag. 2002;43:1773–89. https://doi.org/10.1016/S0196-8904(01)00129-7.
Chandrasekaran P, Cheralathan M, Kumaresan V, Velraj R. Enhanced heat transfer characteristics of water based copper oxide nanofluid PCM (phase change material) in a spherical capsule during solidification for energy efficient cool thermal storage system. Energy. 2014;72:636–42. https://doi.org/10.1016/j.energy.2014.05.089.
Braga SL, Milón Guzmán JJ, Jiménez Pacheco HG. A study of cooling rate of the supercooled water inside of cylindrical capsules. Int J Refrig. 2009;32:953–9. https://doi.org/10.1016/j.ijrefrig.2008.10.014.
Güémez J, Fiolhais C, Fiolhais M. Revisiting Black’s experiments on the latent heats of water. Phys Teach. 2002;40:26–31. https://doi.org/10.1119/1.1457825.
Acknowledgements
The authors wish to thank the Department of Mechanical Engineering, Pondicherry Engineering College, Puducherry for the support and encouragement for carrying out this research work. The authors also like to thank the Department of Mechanical Engineering, SRM Institute of Science and Technology, Kattankulathur, Chennai for their motivation and support.
Funding
The authors state that they did not receive any specific grant from the funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
PS conducted the experiments in various surrounding conditions and consolidate the results. AK and PS interrupt the physical scenario behind the improvement of the thermophysical properties of the Phase Change Materials (PCM).
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no conflicts of interest in publishing this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sundaram, P., Kalaisselvane, A. Effect of different additives on freezing characteristics and stability of GnP-aqueous-based PCM for cold thermal storage. J Therm Anal Calorim 147, 8033–8045 (2022). https://doi.org/10.1007/s10973-021-11056-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-021-11056-y