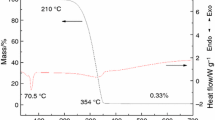
Abstract
Thermal reaction process of paraffin/stearate mixtures was studied by thermogravimetric–differential scanning calorimetry–Fourier transform infrared spectroscopy (TG–DSC–FTIR) simultaneous experiments under argon and oxygen atmospheres, and the thermal reaction kinetics were estimated through the non-isothermal measurements and model-free isoconversional methods. The results show that all stearates except for magnesium stearate have a positive effect on the oxidation of paraffin at lower temperature, but only cobalt stearate plays a positive effect during the whole oxidation process; the oxidation of paraffin/cobalt stearate mixture could be divided into three stages, and cobalt stearate could decrease the oxidation reaction temperature of paraffin obviously concerning the first stage; however, the catalytic effects could not be improved markedly by adding more cobalt stearate; the increase in activation energy (Ea) with the increase of cobalt stearate may be attributed to the adsorption catalysis system of the oxidation process.










Similar content being viewed by others
References
Altman D. Overview and History of Hybrid Rocket Propulsion. Fundamentals of hybrid rocket combustion and propulsion. Progress Astronaut Aeronaut Am Inst Aeronaut Astronaut; 2007. p. 1–36.
Arves J, Jones H, Kline K, Smith K, Slack T, Bales T et al. Development of a N2O/HTPB hybrid rocket motor. In 33rd Joint Propulsion Conference and Exhibit. Joint Propulsion Conferences: American Institute of Aeronautics and Astronautics; 1997.
Yuasa S, Yamamoto K, Hachiya H, Kitagawa K, Oowada Y, editors. Development of a small sounding hybrid rocket with a swirling-oxidizer-type engine. Joint Propulsion Conference and Exhibit; 2001.
Zhao S, Cai G, Tian H, Yu N, Zeng P. Experimental Tests of Throttleable H<sub>2</sub>O<sub>2</sub>/PE Hybrid Rocket Motors. 51st AIAA/SAE/ASEE Joint Propulsion Conference. AIAA Propulsion and Energy Forum: American Institute of Aeronautics and Astronautics; 2015.
Chiaverini MJ, Serin N, Johnson DK, Lu Y-C, Kuo KK, Risha GA. Regression rate behavior of hybrid rocket solid fuels. J Propul Power. 2000;16(1):125–32. https://doi.org/10.2514/2.5541.
Karabeyoglu MA, Altman D, Cantwell BJ. Combustion of liquefying hybrid propellants: part 1, general theory. J Propul Power. 2002;18(3):610–20. https://doi.org/10.2514/2.5975.
Karabeyoglu MA, Cantwell BJ. Combustion of liquefying hybrid propellants: part 2, stability of liquid films. J Propul Power. 2002;18(3):621–30. https://doi.org/10.2514/2.5976.
Karabeyoglu A, Zilliac G, Cantwell BJ, Dezilwa S, Castellucci P. Scale-up tests of high regression rate paraffin-based hybrid rocket fuels. J Propul Power. 2012;20(6):1037–45.
Chiaverini M, Harting G, Lu Y-C, Kuo K, Serin N, Johnson D. Fuel decomposition and boundary-layer combustion processes of hybrid rocket motors. In: 31st Joint Propulsion Conference and Exhibit. Joint Propulsion Conferences: American Institute of Aeronautics and Astronautics; 1995.
Coronetti A, Sirignano WA. Numerical analysis of hybrid rocket combustion. J Propul Power. 2013;29(2):371–84. https://doi.org/10.2514/1.B34760.
Arena Z, Athougies A, Rodulfo A, DeTurris D. Swirl Injection Hybrid Rocket Motor Design and Testing. 47th AIAA/ASME/SAE/ASEE joint propulsion conference & exhibit. In: Joint Propulsion Conferences: American Institute of Aeronautics and Astronautics; 2011.
Li X, Tian H, Yu N, Cai G. Experimental investigation of combustion in axial-injection end-burning hybrid rocket motor. J Propul Power. 2015;31(3):930–6. https://doi.org/10.2514/1.B35480.
Hitt MA, Frederick RA. Testing and modeling of a porous polyethylene axial-injection, end-burning hybrid rocket motor. In: 51st AIAA/SAE/ASEE Joint Propulsion Conference. AIAA Propulsion and Energy Forum: American Institute of Aeronautics and Astronautics; 2015.
Hu S, Wu G, Hua Y, Rashid NF, Hu H. Study on thermal degradation characteristics and regression rate measurement of paraffin-based fuel. Energies. 2015;8(9):10058–81.
He S, Li J, Wang B, Dai X, Sun C, Bai Z, et al. Metals modification of the paraffin dehydrogenation catalyst regenerated by coke combustion. Appl Catal A. 2016;513:82–8.
Pal Y, Kumar VR. Thermal decomposition study of paraffin based hybrid rocket fuel containing Aluminum and Boron additives. Thermochim Acta. 2017;655:63–75. https://doi.org/10.1016/j.tca.2017.06.002.
Gönen M, Balköse D, İnal F, Ülkü S. The effect of zinc stearate on thermal degradation of paraffin wax. J Therm Anal Calorim. 2008;94(3):737–42.
Flynn JH, Wall LA. General treatment of the thermogravimetry of polymers. J Res Natl Bureau Stand Phys Chem. 1966;70A(6).
Ozawa T. A new method of analyzing thermogravimetric data. Bull Chem Soc Jpn. 1965;38(11):1881–6.
Kissinger HE. Reaction kinetics in differential thermal analysis. Anal Chem. 1957;29(11):1702–6. https://doi.org/10.1021/ac60131a045.
Akahira T, Sunose T. Joint convention of four electrical institutes. Res Rep Chiba Inst Technol. 1971;16:22–31.
Starink MJ. A new method for the derivation of activation energies from experiments performed at constant heating rate. Thermochim Acta. 1996;288(288):97–104.
Liu L-l, Hu S-q, Liu P-j, He G-q, Wu G-f. Thermal oxidation reaction of paraffin with O2 and N2O under different pressures. J Therm Anal Calorim. 2018;131(2):1817–20. https://doi.org/10.1007/s10973-017-6615-7.
Acknowledgements
This work is supported by National Natural Science Foundation of China (Grant No. 51976175 and No. 51606157), Natural Science Basic Research Plan in Shaanxi Province of China (Grant No. 606150897031) and Fundamental Research Funds for the Central Universities (Grant No. 3102017zy007).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, Ll., Li, Bb., Kang, B. et al. Effect of stearates on the thermal oxidation process of paraffin. J Therm Anal Calorim 146, 2233–2240 (2021). https://doi.org/10.1007/s10973-021-10577-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-021-10577-w