Abstract
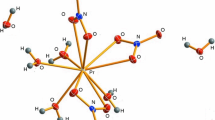
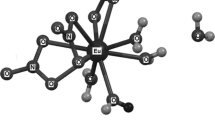
The hexahydrate Nd(NO3)3·6H2O melts in its water of crystallization at 328 K. There are no phase transitions at least above 233 K. Similar to other nitrates in the lanthanide series, its thermal decomposition is a gradual process, which entails the formation of a cluster 6[Nd(NO3)3·6H2O] after the condensation of 6 mol of the initial monomer. During this process, a multicomponent multiphase system exists far from equilibrium and is continuously maintained in this state until normal neodymium oxide is formed. The main volatile products of thermolysis are water, nitric acid, azeotrope 68% HNO3–32% H2O, nitrogen dioxide and oxygen. The proposed mechanism satisfactorily explains the mass losses in accordance with the experimental results.






Similar content being viewed by others
References
Lacerda S, Toth E. Lanthanide complexes in molecular magnetic resonance imaging and theranostics. ChemMedChem. 2017;12:883–94.
Rare Earth Coordination Chemistry. Chunui Haung. Asian ed. Singapore: Wiley; 2011.
Hijazi AK, Taha ZF, Ajlouni AM, Al-Momani WM, Idris IM, Hamra TA. Synthesis and biological activities of lanthanide(III) nitrate complexes with N-(2-hydroxynaphthalen-1-yl) methylene) nicotinohydrazide Schiff base. J Med Chem. 2017;13:77–84.
Rogers DJ, Taylor NJ, Toogood GE. Tetraaquatrinitratoneodymium(III) dihydrate, [Nd(NO3)3(H2O)4]·2H2O. Acta Cryst. 1983;39:939–41.
Balboul B, Myhoub YZ. The characterization of the formation course of neodymium oxide from different precursors: a study of thermal decomposition and combustion process. J Anal Appl Pyrol. 2010;89:95–101.
Melnikov P, Nascimento VA, Consolo LZZ. Thermal decomposition of gallium nitrate hydrate and modeling of thermolysis products. J Therm Anal Calorim. 2012;107:1117–21.
Melnikov P, Nascimento VA, Consolo LZZ. Computerized modeling of intermediate compounds formed during thermal decomposition of gadolinium nitrate hydrate. Russ J Phys Chem. 2012;86:1659–63.
Melnikov P, Nascimento VA, Consolo LZZ, Silva AF. Mechanism of thermal decomposition of yttrium nitrate hexahydrate Y(NO3)3·6H2O and modeling of intermediate oxynitrates. J Therm Anal Calorim. 2013;111:115–9.
Melnikov P, Nascimento VA, Arkhangelsky IV, Consolo LZZ. Thermal decomposition mechanism of aluminum nitrate octahydrate and characterization of intermediate products by the technique of computerized modeling. J Therm Anal Calorim. 2013;111:543–8.
Melnikov P, Nascimento VA, Arkhangelsky IV, Consolo LZZ, de Oliveira LCS. Thermolysis mechanism of chromium nitrate nonahydrate and computerized modeling of intermediate products. J Therm Anal Calorim. 2013;114:1021–7.
Melnikov P, Nascimento VA, Arkhangelsky IV, Consolo LZZ, de Oliveira LCS. Thermal decomposition mechanism of iron(III) nitrate and characterization of intermediate products by the technique of computerized modeling. J Therm Anal Calorim. 2014;115:145–51.
Melnikov P, Arkhangelsky IV, Nascimento VA, Silva AF, Consolo LZZ, de Oliveira LCS, Herrero AS. Thermolysis mechanism of dysprosium hexahydrate nitrate Dy(NO3)3·6H2O and modeling of intermediate decomposition products. J Therm Anal Calorim. 2015;122:571–8.
Melnikov P, Arkhangelsky IV, Nascimento VA, Silva AF, Consolo LZZ. Thermolysis mechanism of samarium nitrate hexahydrate. J Therm Anal Calorim. 2014;118:1537–41.
Melnikov P, Nascimento VA, Arkhangelsky IV, Silva AF, Zanoni Consolo LZ. Thermogravimetric study of the scandium nitrate hexahydrate thermolysis and computer modeling of intermediate oxynitrates. J Therm Anal Calorim. 2015;119:1073–9.
Melnikov P, Arkhangelsky IV, Nascimento VA, de Oliveira LCS, Silva AF, Zanoni LZ. Thermal analysis of europium nitrate hexahydrate Eu(NO3)3·6H2O. J Therm Anal Calorim. 2016;128:12–6.
Melnikov P, Arkhangelsky IV, Nascimento VA, Oliveira LCS, Guimarães WR, Zanoni LZ. Thermal decomposition of praseodymium nitrate hexahydrate Pr(NO3)3·6H2O. J Therm Anal Calorim. 2018;133:929–34.
Strydom CA, Van Vuuren CPJ. The thermal decomposition of lanthanum(III), praseodymium(III) and europium(III) nitrates. Thermochim Acta. 1988;124:277–83.
NIST Chemistry WebBook. NIST standard reference database number 69. www.http//webbook.nist/chemistry. Accessed 22 Sept 2018.
Horsley LH. Azeotrope data: tables of azeotropes and nonazeotropes. Washington, DC: American Chemical Society; 1952.
Liu Y, Bluck D, Brana-Melero F. Static and dynamic simulation of NOx absorption tower based on a hybrid-kinetic equilibrium reaction model. In: Eden MR, Siirola JD, Towler GP, editors. Proceedings of the 8th international conference on foundations of computer-aided process design. Amsterdam: Elsevier; 2014. p. 363.
S.C. Atkinson. Crystal structures and phase transitions in the rare earth oxides. Ph.D. thesis, University of Salford; 2013.
Tian H, Guo YN, Zhao L, Tang J, Liu Z. Hexanuclear dysprosium(III) compound incorporating vertex- and edge-sharing Dy3 triangles exhibiting single-molecule-magnet behavior. Inorg Chem. 2011;50:8688–90.
Giester G, Unfried P, Zak Z. Synthesis and crystal structure of some new rare earth basic nitrates II: [Ln6O(OH)8(H2O)12(NO3)6](NO3)2·xH2O, Ln = Sm, Dy, Er; x(Sm) = 6, x(Dy) = 5, x(Er) = 4. J Alloys Compd. 1997;257:175–81.
Acknowledgements
This study was financed by Conselho Nacional de Pesquisa (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Melnikov, P., Arkhangelsky, I.V., Nascimento, V.A. et al. Thermoanalytical behavior of neodymium nitrate hexahydrate Nd(NO3)3·6H2O. J Therm Anal Calorim 139, 3493–3497 (2020). https://doi.org/10.1007/s10973-019-08748-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-019-08748-x