Abstract
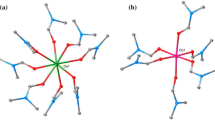
Three-dimensional MOFs, {[Cu3(tz)4Cl2] 2CH3OH}n (Htz = 1H-1,2,3,4-tetrazole), have been synthesized in methanol. It was characterized by chemical analysis, element analysis, IR spectroscopy, single-crystal X-ray diffraction and thermal analysis. Thermogravimetric analysis demonstrated that the MOFs after removing methanol molecules possessed good thermostability with decomposition temperature up to 533 K. The enthalpy change of liquid-phase formation reaction was determined by a RD496–2000 microcalorimeter at 298.15 K with the value of (−46.63 ± 0.26) kJ mol−1. The enthalpy change of solid-phase formation reaction was calculated as (−573.89 ± 0.89) kJ mol−1 on the basis of a designed thermochemical cycle. The thermodynamics of formation reaction of the MOFs was investigated by changing the temperature of liquid-phase reaction. Based on the experimental results, fundamental kinetic and thermodynamic parameters k, n, E, \(\Delta H_{ \ne }^{\theta }\), \(\Delta S_{ \ne }^{\theta }\) and \(\Delta G_{ \ne }^{\theta }\) were obtained. The specific heat capacity at 298.15 K was determined to be (1.87 ± 0.08) J K−1 g−1 by a RD496-2000 calorimeter. In addition, the constant volume combustion energy of title MOFs was determined by a RBC-II rotating-bomb calorimeter at 298.15 K. The standard molar enthalpy of combustion and standard molar enthalpy of formation were calculated to be (−4736.43 ± 4.03) kJ mol−1 and (293.42 ± 4.11) kJ mol−1, respectively.




Similar content being viewed by others
References
Čejka J. Metal-organic frameworks. applications from catalysis to gas storage. Angew Chem Int Ed. 2012;51:4782–3.
Logvinenko VA, Sapchenko SA, Fedin VP. Thermal decomposition of inclusion compounds on the base of the metal–organic framework [Zn4(DMF)(ur)2(ndc)4]. J Therm Anal Calorim. 2016;123:697–702.
Cheetham AK, Rao CNR, Feller RK. Structural diversity and chemical trends in hybrid inorganic–organic framework materials. Chem Commun. 2006;46:4780–95.
Allendorf MD, Bauer CA, Bhakta RK, Houk RJT. Luminescent metal–organic frameworks. Chem Soc Rev. 2009;38:1330–52.
Kurmoo M. Magnetic metal–organic frameworks. Chem Soc Rev. 2009;38:1353–79.
Morozan A, Jaouen F. Metal organic frameworks for electrochemical applications. Energy Environ Sci. 2012;5:9269–90.
Hosny NM. Solvothermal synthesis, thermal and adsorption properties of metal-organic frameworks Zn and CoZn(DPB). J Therm Anal Calorim. 2015;122:89–95.
Hu ZC, Deiberta BJ, Li J. Luminescent metal–organic frameworks for chemical sensing and explosive detection. Chem Soc Rev. 2014;43:5815–40.
Li SH, Wang Y, Qi C, Zhao XX, Zhang JH, Zhang SW, Pang SP. 3D energetic metal–organic frameworks: synthesis and properties of high energy material. Angew Chem Int Ed. 2013;52:14031–5.
He X, Wu CD, Li MX, Batten SR. Synthesis, structure and properties of two new 3D frameworks with primitive cubic topology. Inorg Chem Commun. 2008;11:1378–81.
Zhao XJ, Yang EC, Hou ZJ, Liu ZY, Liu N. Four tetrazolate-based 3D frameworks with diverse subunits directed by inorganic anions and azido coligand: hydro/solvothermal syntheses, crystal structures, and magnetic properties. Dalton Trans. 2015;44:2223–33.
Song Q, Kang QY, Li SY, Yu QY, Liu ZH. Thermochemical properties of three isotypic polyborate cluster compounds: Na5[MB24O34(OH)12] nH2O (M = Cr 3+, Al 3+, Ga 3+). J Chem Thermodyn. 2014;72:65–70.
Wu J, Chen SP, Di YY, Gao SL. Low-temperature thermodynamics of Ln(Me2dtc)3(C12H8N2) (Me2dtc = dimethyldithiocarbamate, Ln = La, Pr, Nd, Sm). J Therm Anal Calorim. 2010;100:1091–8.
Olkhovich MV, Blokhina SV, Sharapova AV, Perlovich GL, Proshin AN. Thermodynamics of sublimation and solvation for bicyclo-derivatives of 1,3-thiazine. Thermochim Acta. 2013;569:61–5.
Guan W, Xue WF, Chen SP, Fang DW, Huang Y, Gao SL. Enthalpy of dilution of aqueous [C4mim][Gly] at 298.15 K. J Chem Eng Data. 2009;54:2871–3.
Kilday MV. The enthalpy of solution of SRM 1665 (KCl). J Res Natl Inst Stand. 1980;85:467–81.
Yang XW, Chen SP, Gao SL, Li H, Shi QZ. Construction of a rotating-bomb combustion calorimeter and measurement of thermal effects. Instrum Sci Technol. 2002;30:311–21.
Sheldrick GM. SHELXS-97 Program for X-ray crystal structure determination. Göttingen: Göttingen University; 1997.
Sheldrick GM. SHELXL-97. Göttingen: Göttingen University Program for X-ray crystal structure refinement; 1997.
Lyakhov AS, Gaponik PN, Degtyarik MM, Matulis VE, Ivashkevich LS. The layered structure of poly-[[di-µ-chloro-bis-[chloro(2-ethyl-tetrazole-κN4)copper(II)]]-di-µ-2-ethyl-tetrazole-κ 2N1:N4]. Acta Cryst. 2003;C59:m204–6.
Gao SL, Ji M, Chen SP, Hu RZ, Shi QZ. The thermokinetics of the formation reaction of cobalt histidine complex. J Therm Anal Calorim. 2001;66:423–9.
Zhang Y, Wu H, Xu KZ, Qiu QQ, An T, Song JR, Zhao FQ. Nonisothermal decomposition kinetics, specific heat capacity, and adiabatic time-to-explosion of Zn(NH3)2(FOX-7)2. J Therm Anal Calorim. 2014;116:817–23.
Wang SX, Tan ZC, Di YY. Heat capacity and thermodynamic properties of 2, 4-dichlorobenzaldehyde (C7H4Cl2O). J Chem Thermodyn. 2004;36:393–9.
Aaron RA, Eulogio OG. Thermochemistry of methyl-D-glucopyranosides and methyl-D-galactopyranosides. J Chem Thermodyn. 2000;32:767–5.
NIST Chemistry WebBook. http://webbook.nist.gov/chemistry/. Accessed May 2016.
Chen SP, Xie G, Fan G, Gao SL, Shi QZ. Thermochemistry of the ternary complex Eu(Et2dtc)3(phen). J Chem Thermodyn. 2005;37:397–404.
Acknowledgements
We gratefully acknowledge the financial support from the National Natural Science Foundation of China (Grant Nos. 21373162, 21473135, 21673180, 21673181).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, Q., Yang, G., Ge, J. et al. Thermodynamic properties of 3D copper(II)-MOFs assembled by 1H-tetrazole. J Therm Anal Calorim 128, 1175–1182 (2017). https://doi.org/10.1007/s10973-016-6014-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-016-6014-5