Abstract
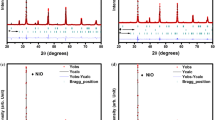
Yttrium-doped strontium titanate is a perovskite with a great potential to act as an anode material for solid oxide fuel cells. Two different synthesis procedures have been used to modify and optimize perovskite morphology. First way involved usage of surfactant TX-100 and led to obtainment of irregular structure with pore diameter of 150–200 nm, whereas in the second one, spheres of poly (methyl methacrylate) acted as soft templates and led to attainment of highly regular hollow perovskite network with pore diameter of around 110 nm. The discussion concerning the influence of yttrium on thermal properties of material, thermal expansion coefficients and chemical stability (in water–carbon dioxide rich atmosphere) were presented. It was found that introduction of yttrium into the strontium titanate leads to the decrease of SrTiO3 shrinkage temperature. Moreover, coupling TG measurements with mass spectrometry allowed to determinate behavior of pure and yttrium-doped strontium titanate in H2O and CO2 containing atmosphere. It was observed that addition of yttrium into SrTiO3 has no influence on materials stability.







Similar content being viewed by others
References
Tanaka H, Misono M. Advances in designing perovskite catalysts. Curr Opin Solid State Mat Sci. 2001;5:381–7.
Suthirakun S, Ammal SCh, Xiao G, Chen F, Huang K, Loye H-C, Heyden A. Obtaining mixed ionic/electronic conductivity in perovskite oxides in reducing environment: a computational prediction for doped SrTiO3. Solid State Ion. 2012;228:37–45.
Simwonis D, Tietz F, Stover D. Nickel coarsening in annealed Ni/8YSZ anode substrates for solid oxide fuel cells. Solid State Ion. 2000;132:241–51.
Haile SM, Staneff G, Ryu KH. Non-stoichiometry, grain boundary transport and chemical stability of proton conducting perovskites. J Mater Sci. 2011;36:1149–60.
Verbraeken MC, Ramos T, Agersted K, Ma Q, Savaniu CD, Sudireddy BR, Irvine JTS, Holtappels P, Tietz F. Modified strontium titanates: from defect chemistry to SOFC anodes. RSC Adv. 2015;5:1168–80.
Medvedev D, Murashkina A, Pikalova E, Demin A, Podias A. Tsiakaras. BaCeO3: materials development, properties and application. Prog Mater Sci. 2014;60:72–129.
Ryu KH, Haile SM. Chemical stability and proton conductivity of doped BaCeO3–BaZrO3 solid solutions. Solid State Ion. 1999;125:355–67.
Pasierb P, Gajerski R, Osiadły M, Łącz A. Application of DTA-TG-MS for determination of chemical stability of BaCeO3-δ-based protonic conductors. J Therm Anal Calorim. 2014;117:683–91.
Pasierb P, Drozdz-Ciesla E, Gajerski R, Labus S, Komornicki S, Rekas M. Chemical stability of Ba(Ce1−xTix)1−yYyO3 proton conducting solid electrolytes. J Therm Anal Calorim. 2009;96:475–80.
Lytle FW. X-ray diffractometry of low-temperature phase transformations in strontium titanate. J Appl Phys. 1964;35:2212–5.
Ruffa AR. Calculation of thermal expansion in insulating and ceramic materials. In: Larsen DC, editor. thermal expansion 7. West Lafayette: Purdue Research Foundation; 1982.
Tietz F. Thermal expansion of SOFC materials. Ionics. 1999;5:129–39.
Drożdż-Cieśla E, Wyrwa J, Broś J, Rękas M. Structural, microstructural, thermal and electrical properties of Ni/YSZ cermet materials. J Therm Anal Calorim. 2012;108:1051–7.
Ren TZ, Yuan ZY, Su BL. A novel macroporous structure of mesoporous titanias: synthesis and characterization. Colloid Surf A Physicochem Eng Asp. 2004;241:67–73.
Fang JF, Xuan YM, Li Q. Preparation of three-dimensionally ordered macroporous perovskite materials. Chin Sci Bull. 2011;56:2156–61.
Sadakane M, Asanuma T, Kubo J, Ueda W. Facile procedure to prepare three-dimensionally ordered macroporous (3DOM) perovskite-type mixed metal oxides by colloidal crystal templating method. Chem Mater. 2005;17:3546–51.
Dufaud V, Lefebvre F. Inorganic hybrid materials with encapsulated polyoxometalates. Materials. 2010;3:682–703.
Liu C, Zhang L, Deng J, Mu Q, Dai H, He H. Surfactant-aided hydrothermal synthesis and carbon dioxide adsorption behavior of three-dimensionally mesoporous calcium oxide single-crystallites with tri-, tetra-, and hexagonal morphologies. J Phys Chem C. 2008;112:19248–56.
Athanasiou A, Mitsionis A, Skouras G, Todorova N, Trapalis C, Vaimakis T. Thermogravimetric study of the surfactant–diethanolamine–titanium isopropoxide system behavior. J Therm Anal Calorim. 2014;116:15–25.
Ionita D, Cristea M, Banabic D. Viscoelastic behavior of PMMA in relation to deformation mode. J Therm Anal Calorim. 2015;120:1775–83.
Hui S, Petric A. Evaluation of yttrium-doped SrTiO3 as an anode for solid oxide fuel cells. J Eur Ceram Soc. 2002;22:1673–81.
Lee J, Orilall MC, Warren SC, Kamperman M, Disalvo FJ, Wiesner U. Direct access to thermally stable and highly crystalline mesoporous transition-metal oxides with uniform pores. Nat Mater. 2008;7:222–8.
Ptáček P, Bartoníčková E, Švec J, Opravil T, Šoukal F, Frajkorová F. The kinetics and mechanism of thermal decomposition of SrCO3 polymorphs. Ceram Int. 2015;14:115–26.
Robbins SA, Rupard RG, Weddle BJ, Maull TR, Gallagher PK. Some observations on the use of strontium carbonate as a temperature standard for DTA. Thermochim Acta. 1995;269(270):43–9.
Acknowledgements
This work was financially supported by the National Science Center of the Republic of Poland, Grant No. 2014/14/EST5/00763.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Drożdż, E., Łańcucki, Ł. & Łącz, A. Synthesis, microstructural properties and chemical stability of 3DOM structures of Sr1−xYxTiO3 . J Therm Anal Calorim 125, 1225–1231 (2016). https://doi.org/10.1007/s10973-016-5463-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-016-5463-1