Abstract
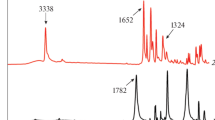
The reaction of cyclization of diphenylalanine in solid phase under heating was studied, which is a stage in formation of various nanostructures from this dipeptide. The temperature ranges of the reaction as well as of dehydration of clathrate of diphenylalanine with water were determined. Kinetic parameters of cyclization were estimated within the approaches of the non-isothermal kinetics (“model-free” kinetics and linear regression methods for detection of topochemical equation). The product of diphenylalanine cyclization was characterized by X-ray powder diffractometry, FTIR spectroscopy and TG/DSC analysis. Crystallization of diphenylalanine and cyclo(diphenylalanine) from methanol solutions was studied using atomic force microscopy. The results obtained may be useful for the design of new nanomaterials based on diphenylalanine at high temperatures.








Similar content being viewed by others
References
Busseron E, Ruff Y, Moulin E, Giuseppone N. Supramolecular self-assemblies as functional nanomaterials. Nanoscale. 2013;5:7098–140.
Hamley IW. Peptide nanotubes. Angew Chem Int Ed. 2014;53:6866–81.
Ma H, Fei J, Li Q, Li J. Photo-induced reversible structural transition of cationic diphenylalanine peptide self-assembly. Small. 2015;11:1787–91.
Guo C, Luo Y, Zhou R, Wei G. Triphenylalanine peptides self-assemble into nanospheres and nanorods that are different from the nanovesicles and nanotubes formed by diphenylalanine peptides. Nanoscale. 2014;6:2800–11.
Afonso R, Mendes A, Gales L. Peptide-based solids: porosity and zeolitic behavior. J Mater Chem. 2012;22:1709–23.
Soldatov DV, Moudrakovski IL, Grachev EV, Ripmeester JA. Micropores in crystalline dipeptides as seen from the crystal structure, He pycnometry, and 129Xe NMR spectroscopy. J Am Chem Soc. 2006;128:6737–44.
Comotti A, Bracco S, Distefano G, Sozzani P. Methane, carbon dioxide and hydrogen storage in nanoporous dipeptide-based materials. Chem Commun. 2009;3:284–6.
Soldatov DV, Moudrakovski IL, Ripmeester JA. Dipeptides as microporous materials. Angew Chem Int Ed. 2004;116:6468–71.
Reches M, Gazit E. Casting metal nanowires within discrete self-assembled peptide nanotubes. Science. 2003;300:625–7.
Ryu J, Kim S-W, Kang K, Park CB. Synthesis of diphenylalanine/cobalt oxide hybrid nanowires and their application to energy storage. ACS Nano. 2010;4:159–64.
Adler-Abramovich L, Badihi-Mossberg M, Gazit E, Rishpon J. Characterization of peptide-nanostructure-modified electrodes and their application for ultrasensitive environmental monitoring. Small. 2010;6:825–31.
Gorbitz CH. Microporous organic materials from hydrophobic dipeptides. Chem Eur J. 2007;13:1022–31.
Kim S, Kim JH, Lee JS, Park CB. Beta-sheet-forming, self-assembled peptide nanomaterials towards optical, energy, and healthcare applications. Small. 2015;11:3623–40.
Li Q, Ma H, Jia Y, Li J, Zhu B. Facile fabrication of diphenylalanine peptide hollow spheres using ultrasound-assisted emulsion templates. Chem Commun. 2015;51:7219–21.
Gleichmann AJ, Wolff JM, Sheldrick WS. η5-Pentamethylcyclopentadienylruthenium(II) complexes containing η6-co-ordinated dipeptides with aromatic side chains. J Chem Soc Dalton Trans. 1995;1:1549–54.
Walchshofer N, Sarciron ME, Garnier F, Delatour P, Petavy AF, Paris J. Anthelmintic activity of 3,6-dibenzyl-2,5-dioxopiperazine, cyclo(L-Phe-L-Phe). Amino Acids. 1997;12:41–7.
Hill RJA, Sedman VL, Allen S, Williams PM, Paoli M, Adler-Abramovich L, Gazit E, Eaves L, Tendler SJB. Alignment of aromatic peptide tubes in strong magnetic fields. Adv Mater. 2007;19:4474–9.
Wang M, Du L, Wu X, Xiong S, Chu PK. Charged diphenylalanine nanotubes and controlled hierarchical self-assembly. ACS Nano. 2011;5:4448–54.
Mason TO, Chirgadze DY, Levin A, Adler-Abramovich L, Gazit E, Knowles TPJ, Buell AK. Expanding the solvent chemical space for self-assembly of dipeptide nanostructures. ACS Nano. 2014;8:1243–53.
Li Q, Ma H, Wang A, Jia Y, Dai L, Li J. Self-assembly of cationic dipeptides forming rectangular microtubes and microrods with optical waveguiding properties. Adv Opt Mater. 2015;3:194–8.
Lee JS, Yoon I, Kim J, Ihee H, Kim B, Park CB. Self-assembly of semiconducting photoluminescent peptide nanowires in the vapor phase. Angew Chem Int Ed. 2011;50:1164–7.
Adler-Abramovich L, Aronov D, Beker P, Yevnin M, Stempler S, Buzhansky L, Rosenman G, Gazit E. Self-assembled arrays of peptide nanotubes by vapour deposition. Nat Nanotechnol. 2009;4:849–54.
Adler-Abramovich L, Reches M, Sedman VL, Allen S, Tendler SJB, Gazit E. Thermal and chemical stability of diphenylalanine peptide nanotubes: implications for nanotechnological applications. Langmuir. 2006;22:1313–20.
Ryu J, Park CB. High stability of self-assembled peptide nanowires against thermal, chemical, and proteolytic attacks. Biotechnol Bioeng. 2010;105:221–30.
Ryu J, Park CB. High-temperature self-assembly of peptides into vertically well-aligned nanowires by aniline vapor. Adv Mater. 2008;20:3754–8.
Huang R, Wang Y, Qi W, Su R, He Z. Temperature-induced reversible self-assembly of diphenylalanine peptide and the structural transition from organogel to crystalline nanowires. Nanoscale Res Lett. 2014;9:653–62.
Ziganshin MA, Gerasimov AV, Gorbatchuk VV, Gubaidullin AT. Thermal analysis of clathrates of tripeptide LLL with organic compounds and water. J Therm Anal Calorim. 2015;119:1811–6.
Ziganshin MA, Gubina NS, Gerasimov AV, Gorbatchuk VV, Ziganshina SA, Chuklanov AP, Bukharaev AA. Interaction of l-alanyl-l-valine and l-valyl-l-alanine with organic vapors: thermal stability of clathrates, sorption capacity and the change in the morphology of dipeptide films. Phys Chem Chem Phys. 2015;17:20168–77.
Vyazovkin S, Burnham AK, Criado JM, Luis A, Perez-Maqueda LA, Popescu C, Sbirrazzuoli N. ICTAC kinetics committee recommendations for performing kinetic computations on thermal analysis data. Thermochim Acta. 2011;520:1–19.
Vyazovkin S, Chrissafis K, Di Lorenzo M-R, Koga N, Pijolat M, Roduit B, Sbirrazzuoli N, Suñol J-J. ICTAC kinetics committee recommendations for collecting experimental thermal analysis data for kinetic computations. Thermochim Acta. 2014;590:1–23.
Kissinger HE. Variation of peak temperature with heating rate in differential thermal analysis. J Res Natl Bur Stand. 1956;57:217–21.
Friedman HL. Kinetics of thermal degradation of charforming plastics from thermogravimetry. Application to a phenolic plastic. J Polym Sci. 1964;6:183–95.
Ozawa T. A new method of analyzing thermogravimetric data. Bull Chem Soc Jpn. 1965;38:1881–6.
Ozawa T. Estimation of activation energy by isoconversion methods. Thermochim Acta. 1992;203:159–65.
Flynn JH, Wall LA. General treatment of the thermogravimetry of polymers. J Res Natl Bur Stand. 1966;70:478–523.
Logvinenko V, Drebushchak V, Pinakov D, Chekhova G. Thermodynamic and kinetic stability of inclusion compounds under heating. J Therm Anal Calorim. 2007;90:23–30.
Logvinenko VA, Dybtsev DN, Bolotov VA, Fedin VP. Thermal decomposition of inclusion compounds on the base of the metal–organic framework [Zn2(bdc)2(dabco)]. J Therm Anal Calorim. 2015;121:491–7.
Logvinenko VA, Aliev SB, Fedin VP. Thermal (kinetic) stability of the inclusion compound on the base of Li-contain MOF [Li2(H2btc)]·dioxane. J Therm Anal Calorim. 2015;120:53–8.
Logvinenko V. Stability of supramolecular compounds under heating thermodynamic and kinetic aspects. J Therm Anal Calorim. 2010;101:577–83.
Ziganshin MA, Efimova IG, Gorbatchuk VV, Ziganshina SA, Chuklanov AP, Bukharaev AA, Soldatov DV. Interaction of l-leucyl-l-leucyl-l-leucine thin film with water and organic vapors: receptor properties and related morphology. J Pept Sci. 2012;18:209–14.
Gdaniec M, Liberek B. Structure of cyclo(-l-phenylalanyI-l-phenylalanyl-). Acta Cryst. 1986;C42:1343–5.
Jeon J, Scott M. Shell self-assembly of cyclo-diphenylalanine peptides in vacuum. J Phys Chem B. 2014;118:6644–52.
Azuri I, Adler-Abramovich L, Gazit E, Hod O, Kronik L. Why are diphenylalanine-based peptide nanostructures so rigid? Insights from first principles calculations. J Am Chem Soc. 2014;136:963–9.
Gorbitz CH. Nanotube formation by hydrophobic dipeptides. Chem Eur J. 2001;7:5153–9.
Opfermann J. Kinetic analysis using multivariate non-linear regression. I. Basic concepts. J Therm Anal Calorim. 2000;60:641–58.
Logvinenko VA, Sapchenko SA, Fedin VP. Thermal decomposition of inclusion compounds on the base of the metal–organic framework [Zn4(dmf)(ur)2(ndc)4]. Part I. J Therm Anal Calorim. 2014;117:747–53.
Acknowledgements
This study was supported by Russian Government Program of Competitive Growth of Kazan Federal University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ziganshin, M.A., Gerasimov, A.V., Ziganshina, S.A. et al. Thermally induced diphenylalanine cyclization in solid phase. J Therm Anal Calorim 125, 905–912 (2016). https://doi.org/10.1007/s10973-016-5458-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-016-5458-y