Abstract
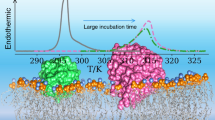
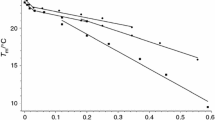
In this paper, the effect of increasing amounts of lysozyme (Lyso) and bovine serum albumin (BSA) on the behaviour of lecithin (DMPC) and cephalin (DMPE) liposomes was investigated by means of Raman and DSC techniques. The results showed that both proteins affected, but in a different way, both lecithin and cephalin liposomes. In the samples with lower Lyso concentrations (up to 2 % w/w), a small decrease on the main transition temperature (T m) was observed, whereas T m increased by further addition of Lyso (up to 15.0 % w/w). At the same time, an increase of about 20 % in the ΔH of the transition was observed. Pre-transition was also affected in a greater extent by protein presence. When BSA interacted with liposomes, a smaller increase in the T m values was observed with a contemporary increase of about 8 % in the associated ΔH. The data suggested that the BSA–liposomes interaction involves only the external surface of the bilayer, excluding thus any penetration into the liposomal hydrophobic core. On the contrary, a partial penetration into the bilayer is suggested when Lyso is added to liposomes. Both considered proteins strengthened the overall bilayer structure of DMPC liposomes, suggesting a decrease in the membrane permeability. Moreover, Lyso secondary structure changed by interaction with liposomes, as demonstrated by the Raman spectra behaviour, in particular in the case of DMPE.





Similar content being viewed by others
References
Ioffe V, Gorbenko GP. Lysozyme effect on structural state of model membranes as revealed by pyrene excimerization studies. Biophys Chem. 2005;114:199–204.
Hirano H, Yoshikawa H, Matsushita S, Yamada Y, Shiraki K. Adsorption and disruption of lipid bilayers by nanoscale protein aggregates. Langmuir. 2012;28:3887–95.
Al Kayal T, Nappini S, Russo E, Berti D, Bucciantini M, Stefani M, Baglioni P. Lysozyme interaction with negatively charged lipid bilayers: protein aggregation and membrane fusion. Soft Matter. 2012;8:4524–34.
Feng W, Zhu SP, Ishihara K, Brash JL. Adsorption of fibrinogen and lysozyme on silicon grafted with poly(2-methacryloyloxyethyl phosphorylcholine) via surface-initiated atom transfer radical polymerization. Langmuir. 2005;21:5980–7.
Roach P, Farrar D, Perry CC. Interpretation of protein adsorption: surface-induced conformational changes. J Am Chem Soc. 2005;127:8168–73.
Husband FA, Garrood MJ, Mackie AR, Burnett GR, Wilde PJ. Adsorbed protein secondary and tertiary structures by circular dichroism and infrared spectroscopy with refractive index matched emulsions. J Agric Food Chem. 2001;49:859–66.
Mudgil P, Torres M, Millar TJ. Adsorption of lysozyme to phospholipid and meibomian lipid monolayer films. Colloid Surf B. 2006;48:128–37.
Matsumura H, Dimitrova M. A comparative study of the sorption of serum albumin, lysozyme, and cytochrome C at phospholipid membranes using surface tensiometry, electrophoresis, and leakage of probe molecules. Colloid Surf B. 1996;6:165–72.
Meng GT, Chan JCK, Rousseau D, Li-Chan ECY. Study of protein–lipid interactions at the bovine serum albumin/oil interface by Raman microspectroscopy. J Agric Food Chem. 2005;53:845–52.
Torreggiani A, Di Foggia M, Manco I, De Maio A, Markarian SA, Bonora S. Effect of sulfoxides on the thermal denaturation of hen lysozyme: a calorimetric and Raman study. J Mol Struct. 2008;891:115–22.
Bonora S, Di Foggia M, Markarian SA, Tugnoli V. Vibrational and calorimetric study on the effect of di-n-propylsulfoxide (DPSO) on DMPC, DPPC and DMPE liposomes. J Mol Struct. 2009;935:115–22.
Dimitrova MN, Matsumura H, Terezova N, Neytchev V. Binding of globular proteins to lipid membranes studied by isothermal titration calorimetry and fluorescence. Colloid Surf B. 2002;24:53–61.
Howell NK, Herman H, Li-Chan ECY. Elucidation of protein–lipid interactions in a lysozyme-corn oil system by Fourier transform Raman spectroscopy. J Agric Food Chem. 2001;49:1529–33.
Swaminathan R, Ravi VK, Kumar S, Kumar MVS, Chandra N. Lysozyme: a model protein for amyloid research. In: Donev R, editor. Advances in protein chemistry and structural biology, vol. 84. San Diego: Elsevier Academic Press; 2011. p. 63–111.
Yuan B, Xing LL, Zhang YD, Lu Y, Luo YY, Mai ZH, Li M. Penetration and saturation of lysozyme in phospholipid bilayers. J Phys Chem B. 2007;111:6151–5.
Takeda K, Wada A, Yamamoto K, Moriyama Y, Aoki K. Conformational change of bovine serum albumin by heat treatment. J Protein Chem. 1989;8:653–9.
Tsunoda T, Imura T, Kadota M, Yamazaki T, Yamauchi H, Kwon OK, Yokoyama S, Sakai H, Abe M. Effects of lysozyme and bovine serum albumin on membrane characteristics of dipalmitoylphosphatidylglycerol liposomes. Colloids Surf B. 2001;20:155–63.
Gardikis K, Hatziantoniou S, Viras K, Wagner M, Demetzos C. A DSC and Raman spectroscopy study on the effect of PAMAM dendrimer on DPPC model lipid membranes. Int J Pharm. 2006;318:118–23.
Iafisco M, Foltran I, Di Foggia M, Bonora S, Roveri N. Calorimetric and Raman investigation of cow’s milk lactoferrin. J Therm Anal Calorim. 2011;103:41–7.
Bonora S, Torreggiani A, Fini G. DSC and Raman study on the interaction between polychlorinated biphenyls (PCB) and phospholipid liposomes. Thermochim Acta. 2003;408:55–65.
Biltonen RL, Lichtenberg D. The use of differential scanning calorimetry as a tool to characterize liposome preparations. Chem Phys Lipids. 1993;64:129–42.
Gaber BP, Peticolas WL. On the quantitative interpretation of biomembrane structure by Raman spectroscopy. Biochim Biophys Acta. 1977;465:260–74.
Tu AT. Raman spectroscopy in biology: principles and applications. New York: Wiley; 1982. p. 65–116.
Takeuchi H. Raman structural markers of tryptophan and histidine side chains in proteins. Biopolymers. 2003;72:305–17.
Yin C, Huo F, Yang P. UV–vis spectroscopic study directly detecting inorganic phosphorus in urine and our reagent kit. Anal Bioanal Chem. 2006;384:774–9.
Tenchov B. On the reversibility of the phase-transitions in lipid–water systems. Chem Phys Lipids. 1991;57:165–77.
Marsh D. Handbook of lipids bilayers. Boca Raton: CRC Press; 1990. p. 135.
Picquart M, Lefèvre T. Raman and Fourier transform infrared study of phytol effects on saturated and unsaturated lipid multibilayers. J Raman Spectrosc. 2003;34:4–12.
Tinti A, Di Foggia M, Taddei P, Torreggiani A, Dettin M, Fagnano C. Vibrational study of auto-assembling oligopeptides for biomedical applications. J Raman Spectrosc. 2008;39:250–9.
Alix AJP, Pedanou G, Berjot M. Fast determination of the quantitative secondary structure of proteins by using some parameters of the raman amide 1-band. J Mol Struct. 1988;174:159–64.
Maddams WF. The scope and limitations of curve fitting. Appl Spectrosc. 1980;34:245–67.
Wojdyr M. Fityk: a general-purpose peak fitting program. J Appl Cryst. 2010;43:1126–8.
Van Osdol WW, Ye Q, Johnson ML, Biltonen RL. Effects of the anesthetic dibucaine on the kinetics of the gel-liquid crystalline transition of dipalmitoylphosphatidylcholine multilamellar vesicles. Biophys J. 1992;63:1011–7.
Jorgensen K, Ipsen JH, Mouritsen OG, Bennett D, Zuckermann M. The effects of density-fluctuations on the partitioning of foreign molecules into lipid bilayers—application to anesthetics and insecticides. J Biochim Biophys Acta. 1991;1062:227–38.
Mouritsen OG, Jorgensen K. Dynamical order and disorder in lipid bilayers. Chem Phys Lipids. 1994;73:3–25.
Ermakov YA, Makhmudova SS, Averbakh AZ. Two components of boundary potentials at the lipid membrane surface: electrokinetic and complementary methods studies. Colloid Surf A. 1998;140:13–22.
Jendrasiak GL, Smith R, Ribeiro AA. Chaotropic anion phosphatidylcholine membrane interactions—an ultra high-field nmr-study. Biochim Biophys Acta. 1993;1145:25–32.
Bertoluzza A, Bonora S, Fini G, Morelli MA. Spectroscopic and calorimetric studies of phospholipid polyamine molecular-interactions. J Raman Spectrosc. 1988;19:369–73.
Kuzuhara A. Protein structural changes in keratin fibers induced by chemical modification using 2-iminothiolane hydrochloride: a Raman spectroscopic investigation. Biopolymers. 2005;79:173–84.
Sabín J, Prieto G, Ruso JM, Messina PV, Salgado FJ, Nogueira M, Costas M, Sarmiento F. Interactions between DMPC liposomes and the serum blood proteins HSA and IgG. J Phys Chem B. 2009;113:1655–61.
Acknowledgements
This work was supported by grants from Bologna University (Ricerca fondamentale orientata—ex 60 %).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Di Foggia, M., Bonora, S. & Tugnoli, V. DSC and Raman study on the effect of lysozyme and bovine serum albumin on phospholipids liposomes. J Therm Anal Calorim 111, 1871–1880 (2013). https://doi.org/10.1007/s10973-012-2842-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-012-2842-0