Abstract
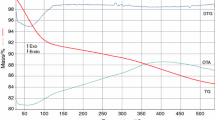
In this article, we present a new method for the obtaining of ZnCr2O4 and MgCr2O4 embedded in silica matrix. This method consists in the formation of Cr(III), Zn(II) and Cr(III), Mg(II) hydroxycarboxylate/carboxylate compounds, during the redox reaction between the nitrate ion and diol (1,3-propanediol), uniformly dispersed in the pores of hybrid gels. The thermal decomposition of these precursors leads to a mixture of corresponding metal oxides. The gels were synthesized starting from mixtures of Cr(NO3)3·9H2O, Zn(NO3)2·6H2O and Cr(NO3)3·9H2O, Mg(NO3)2·6H2O with tetraethyl orthosilicate and 1,3-propanediol for final compositions 50% ZnCr2O4/50% SiO2 and 50% MgCr2O4/50% SiO2. The obtained gels have been thermally treated at 140 °C, when the redox reaction nitrates-diol took place with formation of the precursors within the xerogels pores. The thermal decomposition of all precursors took place up to 300 °C, with formation of oxides mixtures (Cr2O3 + x and ZnO) and (Cr2O3 + x and MgO), respectively. At 400 °C, Cr2O3 + x turn to Cr2O3 which reacts with ZnO forming ZnCr2O4/SiO2. Starting with 400 °C, Cr2O3 reacts with MgO to an intermediary phase MgCrO4, which decomposes with the formation of MgCr2O4/SiO2. The formation of the precursors inside the silica matrix and the evolution of the crystalline phases were studied by thermal analysis, FT-IR spectrometry, XRD, and TEM.









Similar content being viewed by others
Abbreviations
- 1,3PG:
-
1,3-Propanediol
- TEOS:
-
Tetraethyl orthosilicate
References
Morozova LV, Popov VP. Synthesis and investigation of magnesium chromium spinel. Glass Phys Chem. 2010;36(1):86–91.
Mančić L, Marinković ZV, Vulić P, Milošević O. The synthesis-structure relationship in the ZnO-Cr2O3 system. Sci Sinter. 2004;36:189–96.
Szczygiel I, Winiarska K. Low-temperature synthesis and characterization of the Mn–Zn ferrite. J Therm Anal Calorim. 2011;104:577–83.
Souaya ER, Ismail EH, Mohamed AA, Milad NE. Preparation, characterization and thermal studies of some transition metal ternary complexes. J Therm Anal Calorim. 2009;95:253–8.
Suryanarayana C, Grant Norton M. X-ray diffraction. A practical approach. New York: Plenum Press; 1998.
Stefanescu M, Sasca V, Birzescu M. Thermal behaviour of the homopolynuclear glyoxylate complex combinations with Cu(II) and Cr(III). J Therm Anal Calorim. 2003;72(2):515–24.
Stefanescu M, Sasca V, Birzescu M. Studies on the thermal decompositions of heteropolynuclear glyoxylates of Cr(III) and Cu(II). J Therm Anal Calorim. 1999;56(2):569–78.
Stefanescu M, Stoia M, Stefanescu O, Popa A, Simon M, Ionescu C. The interaction between teos and some polyols. Thermal analysis and FT-IR. J Therm Anal Calorim. 2007;88:19–26.
Stefanescu M, Stoia M, Stefanescu O. Thermal and FT-IR study of the hybrid ethylene–glycol–silica matrix. J Sol Gel Sci Techn. 2007;41:71–8.
Brinker CJ, Scherrer GW. Sol–gel science: the physics and chemistry of sol-gel Processing. New York: Academic Press; 1990.
Stefanescu M. Consideration on the formation of the mixed oxides starting from substances with high reactivity. PhD Thesis, University of Timisoara, Romania, 1993.
Barbu M, Stoia M, Stefanescu O, Stefanescu M. Thermal and FT-IR studies on the interaction between Cr(NO3)3·9H2O and some diols. Chem Bull. 2010;55(69):180–5.
Bernazzani P, Sanchez RF. Structural and thermal behavior of polystyrene thin films using ATR–FTIR–NanoDSC measurements. J Therm Anal Calorim. 2009;96:727–32.
Purnendu P, Manivannan V. Microwave metathetic approach for the synthesis and characterization of ZnCr2O4. J Eur Ceram Soc. 2008;28:1665–70.
Pocol V, Patron L, Carp O, Brezeanu M, Segal E, Stanica N. Some polynuclear coordination compounds precursor of chromites synthesis, physicochemical characterization and thermal stability. J Therm Anal Calorim. 1999;55:143–54.
Bielanski A, Deren J, Duczyminska E. Elektrische Leitfahigkeit von MgO–Cr2O3 Gemischen verschiedener Zusammensetzung und verschiedenen Sinterungsgrades. Z Anorg Allg Chim. 1962;316(1–2):75–88.
Rida K, Benabbas A, Bouremmad F, Pen MA, Martinez-Arias A. Influence of the synthesis method on structural properties and catalytic activity for oxidation of CO and C3H6 of pirochromite MgCr2O4. Appl Catal A. 2010;375:101–6.
Joint Committee on Powder Diffraction Standards-International Center for Diffraction Data. Swarthmore, 1993.
Aknowledgements
This study was partially supported by the strategic grant POSDRU/88/1.5/S/50783, Project ID50783 (2009), co-financed by the European Social Fund—Investing in People, within the Sectoral Operational Programme Human Resources Development 2007-2013 and by the strategic grant POSDRU/21/1.5/G/13798 inside POSDRU Romania 2007-2013, co-financed by the European Social Fund—Investing in People.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barbu, M., Stefanescu, M., Stoia, M. et al. New synthesis method for M(II) chromites/silica nanocomposites by thermal decomposition of some precursors formed inside the silica gels. J Therm Anal Calorim 108, 1059–1066 (2012). https://doi.org/10.1007/s10973-011-1933-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-011-1933-7