Abstract
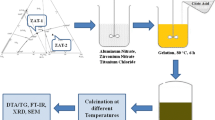
GEL combustion technique was applied to obtain oxides of thorium and cerium from their respective nitrate solutions using citric acid as the gelating agent. The dried samples were characterized by IR and TG studies. Intermediate and final products during TG studies have been isolated and characterized by XRD studies. All the TG runs during heating of thorium and cerium nitrate with citric acid dried Gels showed a two step process. The weight loss at each step and the X-ray data of the product at each step, helped in suggesting a possible mechanism. Kinetic study was carried out independently for each step. The reaction mechanism as observed during interactive procedure was found to be diffusion controlled. The kinetic parameters (activation energy and pre-exponential factor) for each step in all reactions have been calculated. Observations from XRD studies show that with increase in cerium concentration in the oxides, the lattice parameter values have shown a decreasing trend for all the five compositions studied. It was observed that in TG studies with increase in cerium concentration, the final temperature of the reactions have shown a decreasing trend. SEM studies of the powders reveal that synthesized oxides have a tendency to form agglomerate of varying size ranging from 50 to 100 μm in case of mixed oxides but the size of thorium oxide powder so synthesized have pore size 10–100 μm. SEM images shows that GEL combustion may result in agglomeration, if the temperature is not properly controlled to the desired value. SEM studies also reveal that each agglomerate contains approximately 10–100 individual particles. Surface area of the mixed oxide powders were determined using Gas adsorption technique. The surface area was found to be in the range of 3–17 m2/g in all cases. Specific surface area of thorium oxide was found to be lesser than cerium oxide but in case of mixed oxides surface area decreases with increase in cerium content. Majority of pores, indicating the particle size are in the range of 0.01–0.04 cm3/g.





Similar content being viewed by others
References
Pepin JG, McCarthy GJ. J Am Ceram Soc. 1918;64(9):511.
Pope JML, Redford KC. Physical properties of some thoria powders and their influence on sinterability. J Nucl Mater. 1974;52:241.
Das RN. Nanocrystalline ceramics from sucrose process’. Mater Lett. 2001;47:344–50.
Wu Y, Bandyopadhyay A, Bose S. J Matter Sci Eng A. 2004;380:349–55.
Saberi A, Alinejad B, Nagahdari Z, Kazemi F, Almasi A. A novel method to low temperature synthesis of nanocrystalline forsterite. Mater Res Bull. 2007;42–4:666–73.
Kovasheva D, Gadjov H, Petro K, Mandal S, Lazarraga MG, Pascual L, Amarilla JM, Rojas RN, Herrero P, Rojo JM. J Mater Chem. 2002;12:1184–8.
Alinejad B, Sarpoolaky H, Beittollahi A, Saberi A, Afshar S. Synthesis and characterization of nanocrystalline MgAl2O4 spinel via sucrose process. Mater Res Bull. 2008;43:1188–94.
Chandramouli V, Anthonysamy S, Vasudeva Rao PR, Divakar R, Sundaraman D. PVA aided microwave synthesis: a novel route for the roduction of nanocrystalline thoria powder’. J Nucl Mater. 1996;231:213.
Mathews D, Ambekar BR, Tyagi AK. Bulk thermal expansion studies of Th1−x Ce x O2 in the complete solid solution range. J Nuclear Mater. 2001;288:83–5.
Mothé MG, Leni FMLeite, Mothé CheilaG. Kinetic parameters of different asphalt binders by thermal analysis. J Therm Anal Calorim. 2011;103:1–6.
Madhurambal G, Mariappan M, Ravindran B, Mojumdar SC. Thermal and FTIR spectral studies in various proportions of urea thiourea mixed crystal. J Therm Anal Calorim. 2011;103:1–7.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Naik, Y.P., Ramarao, G.A., Banthiya, A. et al. Synthesis and characterization of nano-structured Th1−x Ce x O2 mixed oxide. J Therm Anal Calorim 107, 105–110 (2012). https://doi.org/10.1007/s10973-011-1533-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-011-1533-6