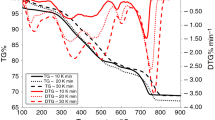
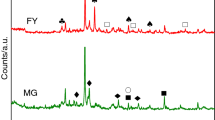
Abstract
The thermooxidative decomposition of four oil shale samples from Estonia, Jordan, Israel and Morocco and one sample of Estonian oil shale derivative, semicoke, was studied with the aim to determine the characteristics of the process and the differences of it related to the origin of oil shale. The experiments with a Setaram Setsys 1750 thermoanalyzer coupled to a Nicolet 380 FTIR Spectrometer were carried out under non-isothermal conditions up to 1000 °C at the heating rates of 1, 2, 5, 10 and 20 °C min−1 in an oxidizing atmosphere. A model-free kinetic analysis approach based on the differential isoconversional method of Friedman was used to calculate the kinetic parameters. The results of TG–DTA–FTIR analyses and the variation of activation energy E along the reaction progress α indicated the complex character of thermooxidative decomposition of oil shale and semicoke, being at that the most complicated for Estonian and Jordanian oil shale characterized by higher content of organic matter as compared to the other samples studied.





Similar content being viewed by others
References
Johannes I, Zaidentsal A. Kinetics of low-temperature retorting of kukersite oil shale. Oil Shale. 2008;25:412–25.
Olivella MA, de las Heras FXC. Nonisothermal thermogravimetry of Spanish fossil fuels. Oil Shale. 2006;23:340–55.
Berkovich AJ, Young BR, Levy JH, Schmidt SJ, Ray A. Thermal characterization of Australian oil shales. J Therm Anal Calorim. 1997;49:737–43.
Torrente MC, Galán MA. Kinetics of the thermal decomposition of oil shale from Puertollano (Spain). Fuel. 2001;80:327–34.
Aboulkas A, El Harfi K. Study of the kinetics and mechanisms of thermal decomposition of Morrocan Tarfaya oil shale and its kerogen. Oil Shale. 2008;25:426–43.
Qing W, Hongpeng L, Baizhong S, Shaohua L. Study on pyrolysis characteristics of Huadian oil shale with isoconversional method. Oil Shale. 2009;26:148–62.
Kök MV, Pamir R. Pyrolysis kinetics of oil shales determined by DSC and TG/DTG. Oil Shale. 2003;20:57–68.
Lisboa ACL, Watkinson AP. Operating conditions for oil shale thermogravimetry. Powder Technol. 1999;101:151–6.
Kök MV, Pokol G, Keskin C, Madarász J, Bagci S. Combustion characteristics of lignite and oil shale samples by thermal analysis techniques. J Therm Anal Calorim. 2004;76:247–54.
Kök MV, Iscan AG. Oil shale kinetics by differential methods. J Therm Anal Calorim. 2007;88:657–61.
Kök MV, Pamir MR. Comparative pyrolysis and combustion kinetics of oil shales. J Anal Appl Pyrol. 2000;55:185–94.
Kök MV. Heating rate effect on the DSC kinetics of oil shale. J Therm Anal Calorim. 2007;90:817–21.
Kök MV, Pamir MR. ASTM kinetics of oil shales. J Therm Anal Calorim. 1998;53:567–75.
Han XX, Jiang XM, Cui ZG. Thermal analysis studies on combustion mechanism of oil shale. J Therm Anal Calorim. 2006;84:631–6.
Qing W, Baizhong S, Xiahua W, Jingru B, Jian S. Influence of retorting temperature on combustion characteristics and kinetic parameters of oil shale semicoke. Oil Shale. 2006;23:328–39.
Qing W, Baizhong S, Xiahua W, Jingru B, Jian S. Study on combustion characteristics of Huadian oil shale and semicoke. Oil Shale. 2007;24:135–45.
Friedman HL. Kinetics of thermal degradation of char-forming plastics from thermogravimetry. Application to phenolic plastic. J Polym Sci. 1965;6C:183–95.
AKTS Softwear Setaram Instruments. A global solution for kinetic analysis and determination of the thermal stability of materials. Switzerland: AKTS AG; 2006. p. 88.
Kaljuvee T, Kuusik R. Emission of sulphur dioxide during thermal treatment of fossil fuels. J Therm Anal Calorim. 1999;56:1243–51.
Kaljuvee T, Kuusik R, Radin M, Bender V. Carbon dioxide binding in the heterogeneous systems formed at combustion of oil shale. 4. Reactivity of ashes towards acid gases in the system fly ash-flue gases. Oil Shale. 2004;21:13–26.
Xie W, Liu K, Pan WP, Riley JT. Interaction between emissions of SO2 and HCl in fluidized bed combustors. Fuel. 1999;78:1425–36.
Partanen J, Backman P, Backman R, Hupa M. Adsorption of HCl by limestone in hot flue gases. Part III: simultaneous adsorption with SO2. Fuel. 2005;84:1685–94.
Lille Ü. Current views on the origin of Estonian kukersite kerogen. Oil Shale. 2002;19:3–18.
Kuusik R, Martins A, Pihu T, Pesur A, Kaljuvee T, Prikk A, Trikkel A, Arro H. Fluidized bed combustion of oil shale retorting solid waste. Oil Shale. 2004;21:237–48.
Trikkel A, Kuusik R, Martins A, Pihu T, Stencel JM. Utilization of Estonian oil shale semicoke. Fuel Proc Technol. 2008;89:756–63.
Kaljuvee T, Pelt J, Radin M. TG-FTIR study of gaseous compounds evolved at thermooxidation of oil shale. J Therm Anal Calorim. 2004;78:399–414.
Brown ME, Maciejewski M, Vyazovkin S, et al. Computational aspects of kinetic analysis. Part A: the ICTAC kinetics project-data, methods and results. Thermochim Acta. 2000;355:125–43.
Acknowledgements
This work was partly supported by the Estonian Ministry of Education and Research (SF0140082s08).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kaljuvee, T., Keelmann, M., Trikkel, A. et al. Thermooxidative decomposition of oil shales. J Therm Anal Calorim 105, 395–403 (2011). https://doi.org/10.1007/s10973-010-1033-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-010-1033-0